Lab 10: Synthesis of Azo Dyes
1/16
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
17 Terms
what is the azo group designated as
R-N=N-R’
how is the azo group prepared?
the coupling of aryl diazonium cation with a coupling reagent via electrophilic substitution
what are the reagents
Sodium carbonate (structure, MW, MP)
Water (structure, MW, BP, density)
Sulfanilic acid (structure, MW, MP)
Sodium nitrite (structure, MW, MP)
Hydrochloric acid (structure, MW, density)
N,N-dimethylaniline (structure, MW, MP)
N,N-diethylaniline (structure, MW, MP)
Phenol (structure, MW, MP)
Glacial acetic acid (structure, MW, BP, density)
Sodium hydroxide (structure, MW, concentration)
Sodium chloride (structure, MW, MP)
Ethanol (structure, MW, BP, density)
broadly explain the procedure
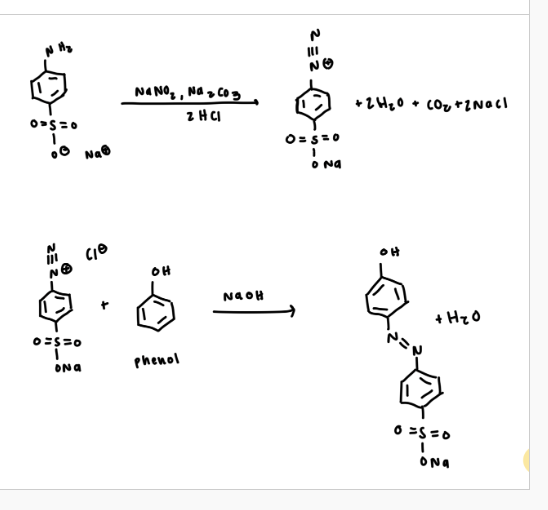
First: preparing the diazotized sulfanilic acid
dissolve anhydrous sodium carbonate in water
add sulfanilic acid monohydrate to the solution and let it dissolve then add sodium nitrite
then add a solution of cold water and hydrochloric acid to the mixture in a dropwise manner to form the diazonium salt (should be a precipitate)
Second: preparation of the azo dye
get you unknown coupling reagent and mix with glacial acetic acid
add dropwise over the diazotized sulfanilic acid solution and a precipatate should form. let the reaction progress and cool for about 15 minutes
add 10% aqueous sodium hydrozide to the solution slowly and as you are stirring. make sure the solution is basic. heat the mixture to make sure the newly formed dye dissolves
when all of it dissolves, add sodium chloride and cool mixture in ice bath (recrystallization should occur)
purify product with hot ethanol (not all dye will dissolve)
let cool then filter through Buchner funnel
what techniques were used
IR and HNMR
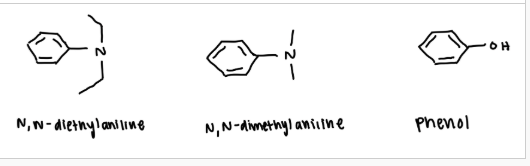
what are the possible unknown coupling materials
N, N-diethylaniline
N, N-dimethylaniline
phenol
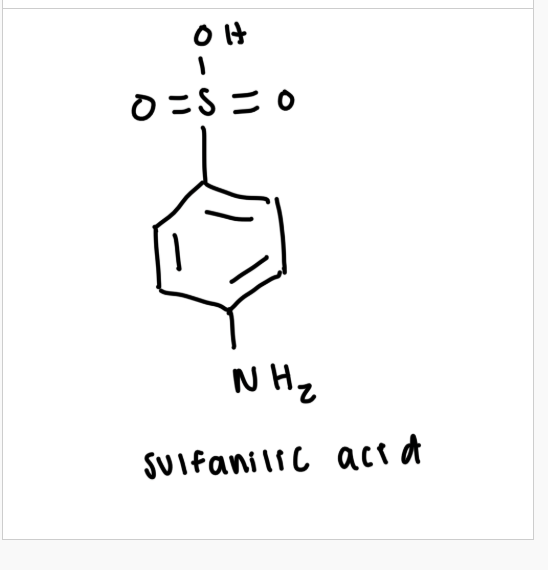
draw the structure of sulfanilic acid
what will be used to purify your product
ethanol
balanced equation
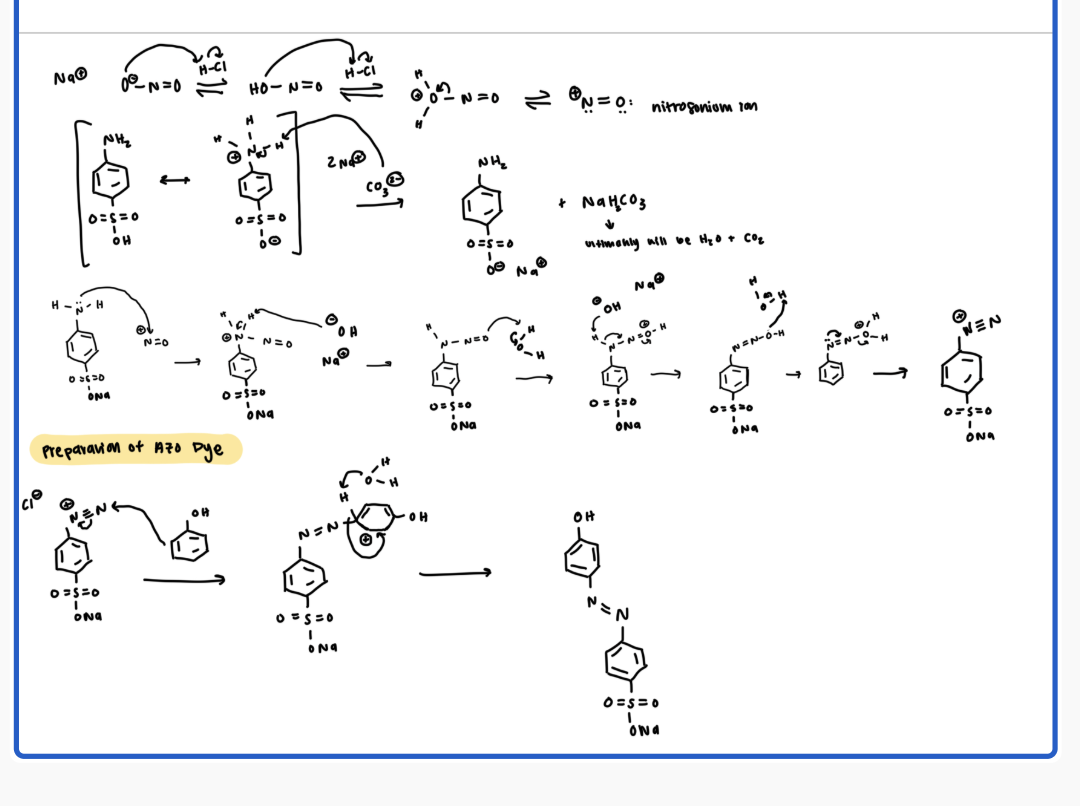
what is the mechanism
Nitrosonium ion
NO2 ion will get protonated by HCl twice to ultimately have H2O leave and have a nitrosonium ion
Diazotized sulfanilic acid formation
deprotonate the NH3 to get NH2 (makes it the soluble version)
then nitrosonium ion will firm a bond with the N in the sulfanilic acid, the central N will get deprotonated, the O in nitrosonium will get protonated, central N will get deprotonated again by hydroxide, other O will get protonated, and lose H, and then you will get your triple bond N
preparation of azo dye
pi bond in phenol will attack the non charged N, and ultimately the adjacent H will get eliminated by H2O to restore aromaticity. the electrons from the triple bonded N get pushed to the positively charged N to neutralize the charge
were there any IR changes
first the amino group is gone in the product IR
the primary amine peak found in sulfanilic acid (should have a broad peak that will have two peak around 3000-3300 cm) will be gone
azo bridge will form (1400-1500)
and a broad peak around 3200-3500 representing the OH group
how do you determine if your reaction went to completion with HNMR
To determine if your reaction went to completion, one prominent change is the absence of the broad signal for the primary amino group on sulfanilic acid. It usually is around 5-6 ppm, but this disappears because it is converted into the diazonium salt and then the azo bridge. Additionally, there should be a broad singlet in my product HNMR. This represents the phenolic proton from the phenol group.
what color of light did your dye absorb
reflected orange so absorbed blue (450-520 nm)
What would happen to the color of the dye if we increased the conjugation in the molecule?
if you increase the conjugation, less energy is needed for electorn excitation. the molecules will absorb light at a longer wavelength, so it will shift from an orange dye to a more red color
what would happen to the color of the dye if we decrease the conjugation?
there is a larger energy gap between energy levels, so the molecule will need more energy for electron excitaiton. it will absorb at higher energy wavelengths (shorter wavelength), and the color will shift from orange to more of a green color
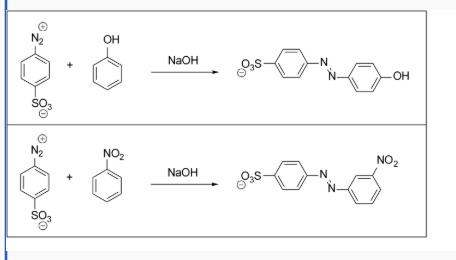
Which of the following azo dye syntheses would be faster? Explain your reasoning.
the one with an electron donating group because it will make the nucleophile more nucleophilic (electron rich). the ring will be more activated. NO2 is an electron withdrawing group and will decrease the electron density around the aromatic ring. this will deactivate the ring and make it less susceptible to attacking the electrophilic diazonium ion (the ring will be deactivated)