Orgo Final (Ethers and Epoxides)
1/10
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
11 Terms
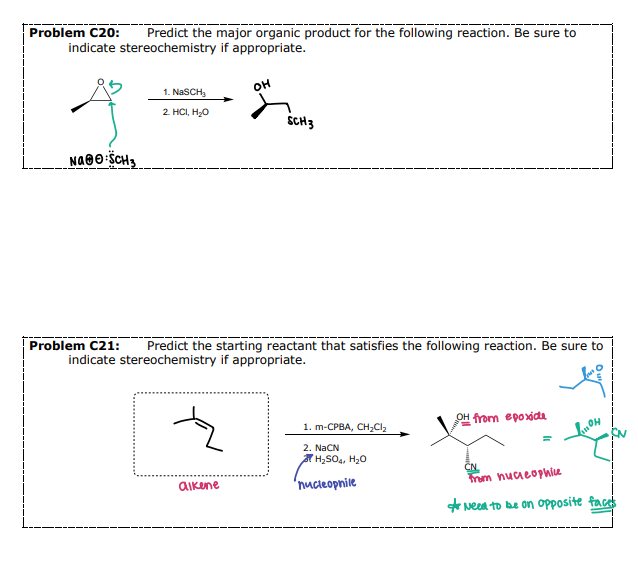
Williamson-Ether Synthesis
#1 Preparation of Ethers - ___________
Step 1: Acid-Base Reaction
Step 2: SN2 Reaction (fails with tertiary alkyl halides)
Original stereochemistry of the alcohol is MAINTAINED
Acid-Catalyzed Addition of Alcohols to Alkenes
#2 Preparation of Ethers - _____________
An alcohol can add to an alkene in the presence of an acid-catalyst
Carbocation Intermediate so shifts are possible.
Acid-Catalyzed Cleavage by HX
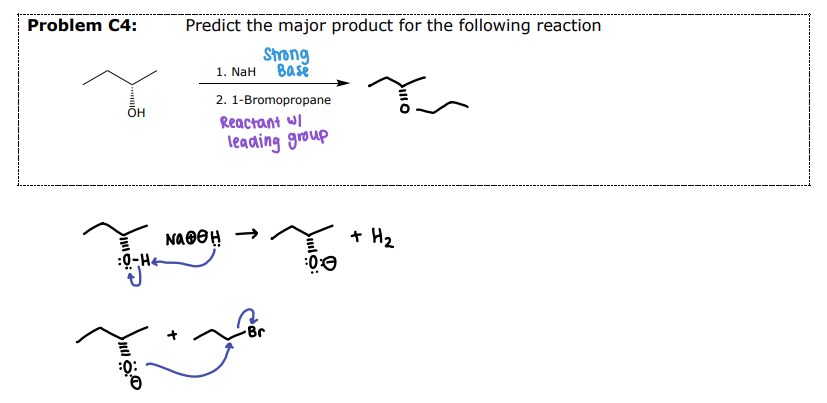
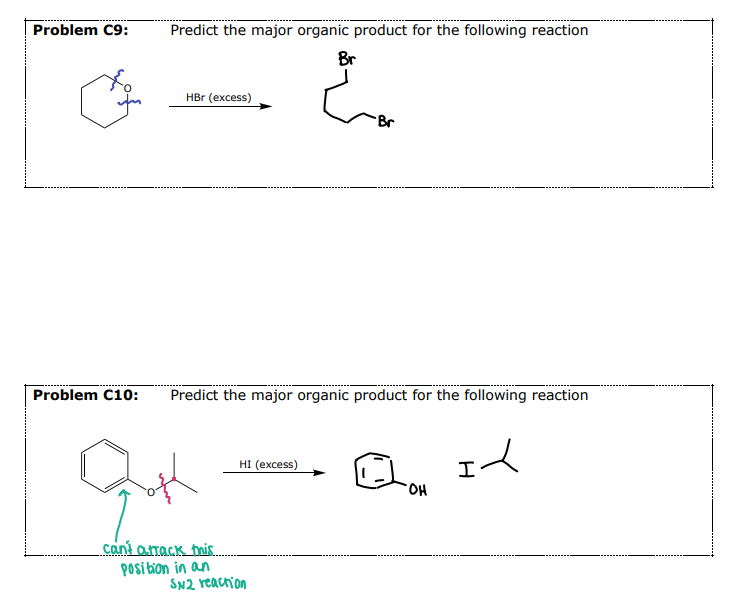
#3 Reactions of Ethers- __________
Reaction requires both a strong acid and a strong nucleophile
HBr and HI are used
Mechanism (Sn1 or Sn2) depends on the substitution of the alkyl group
Aryl and Vinylic ethers will NOT be cleaved
Split across the O and insert the halide
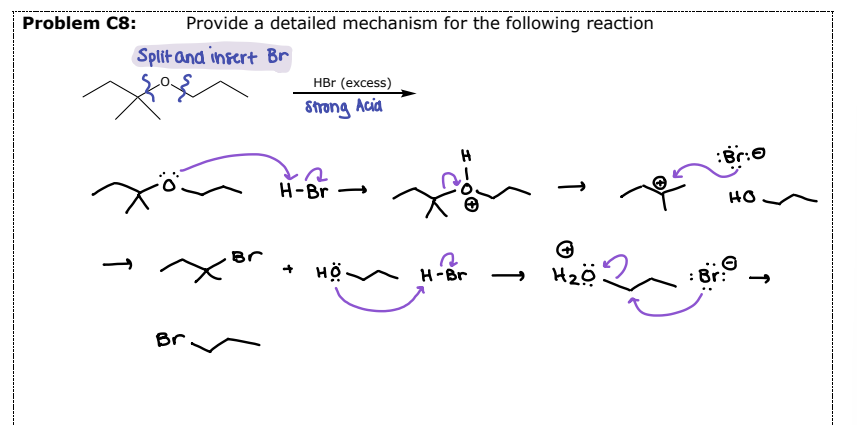
Acid-Catalyzed Cleavage by HX
#3 Example 2
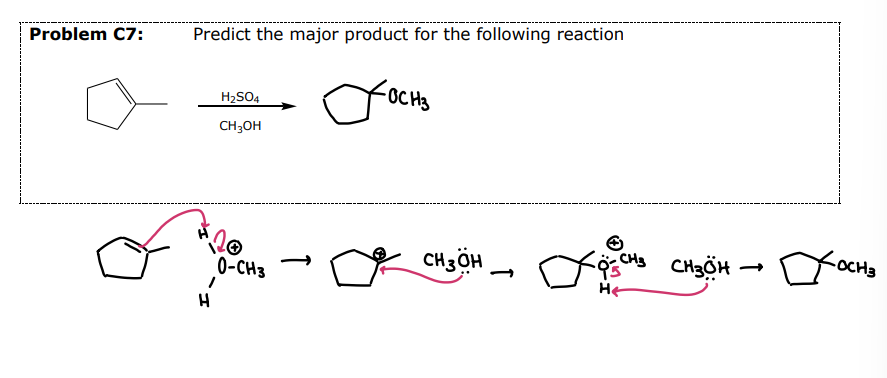
Peroxycarboxylic Acids
#4 Epoxide Formation: __________
mCPBA
Mechanism is concerted
The reaction is stereospecific - Syn Addition
The original stereochemistry of the alkene is maintained
A cis alkene will yield a cis epoxide
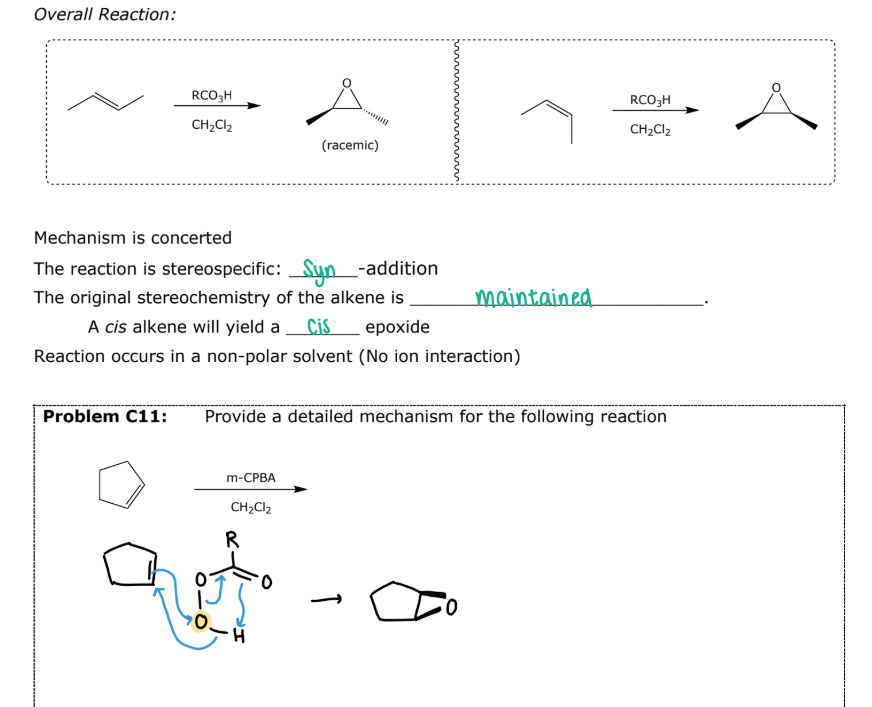
Peroxycarboxylic Acids
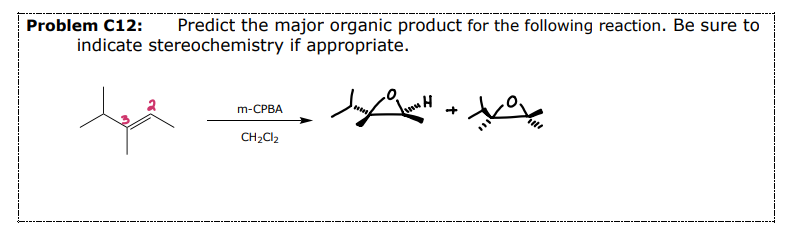
#4 Example 2
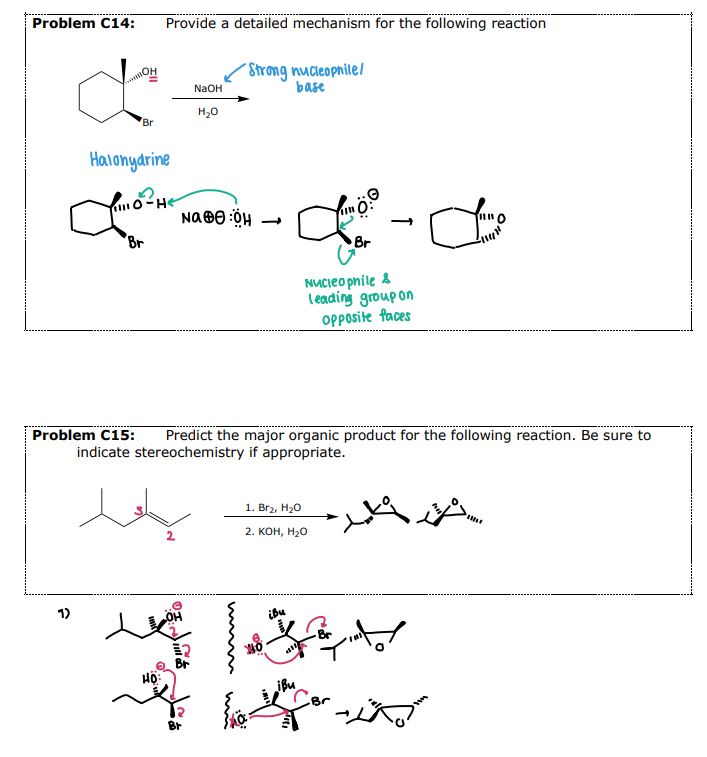
Internal Nucleophilic Substitution in Halohydrins
#5 Epoxide Formation: __________
A base is used to deprotonate the alcohols, which allows for the intramolecular SN2 reaction
Intramolecular variation of Williamson Ether Synthesis
Acid-Catalyzed Ring Opening
#6 Reactions of Epoxides: __________
An acid catalyst is used when the nucleophile is weak
Opens the epoxide on the MORE substituted side (regioselective)
Selectivity: ANTI ADDITION
Protonate
Attack
Deprotonate
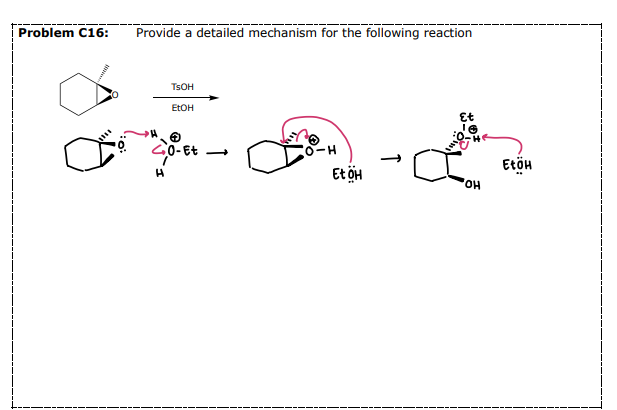
Acid-Catalyzed Ring Opening
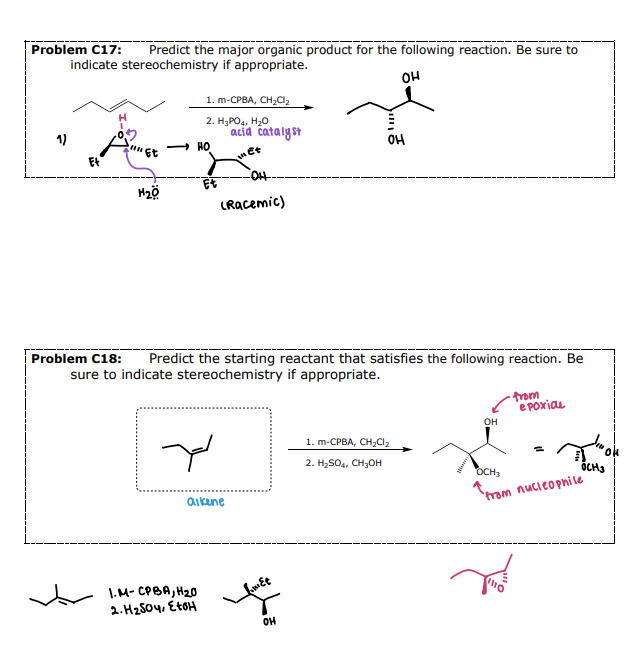
#6 Example 2
Nucleophilic Ring Opening
#7) Reactions of Epoxides: __________
Strong Nucleophiles do not require an acid catalyst
Opens the epoxide on the less-substituted side (regioselective)
Stereoselectivity: Anti-Addition
Attack
Protonate
Some instances require a workup step
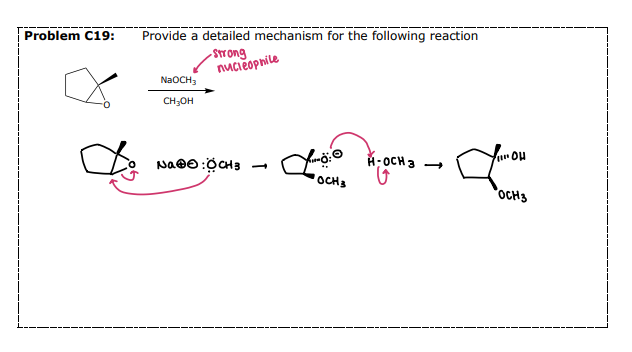
Nucleophilic Ring Opening
#7 Example 2