Chem 1111 final review
1/106
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
107 Terms
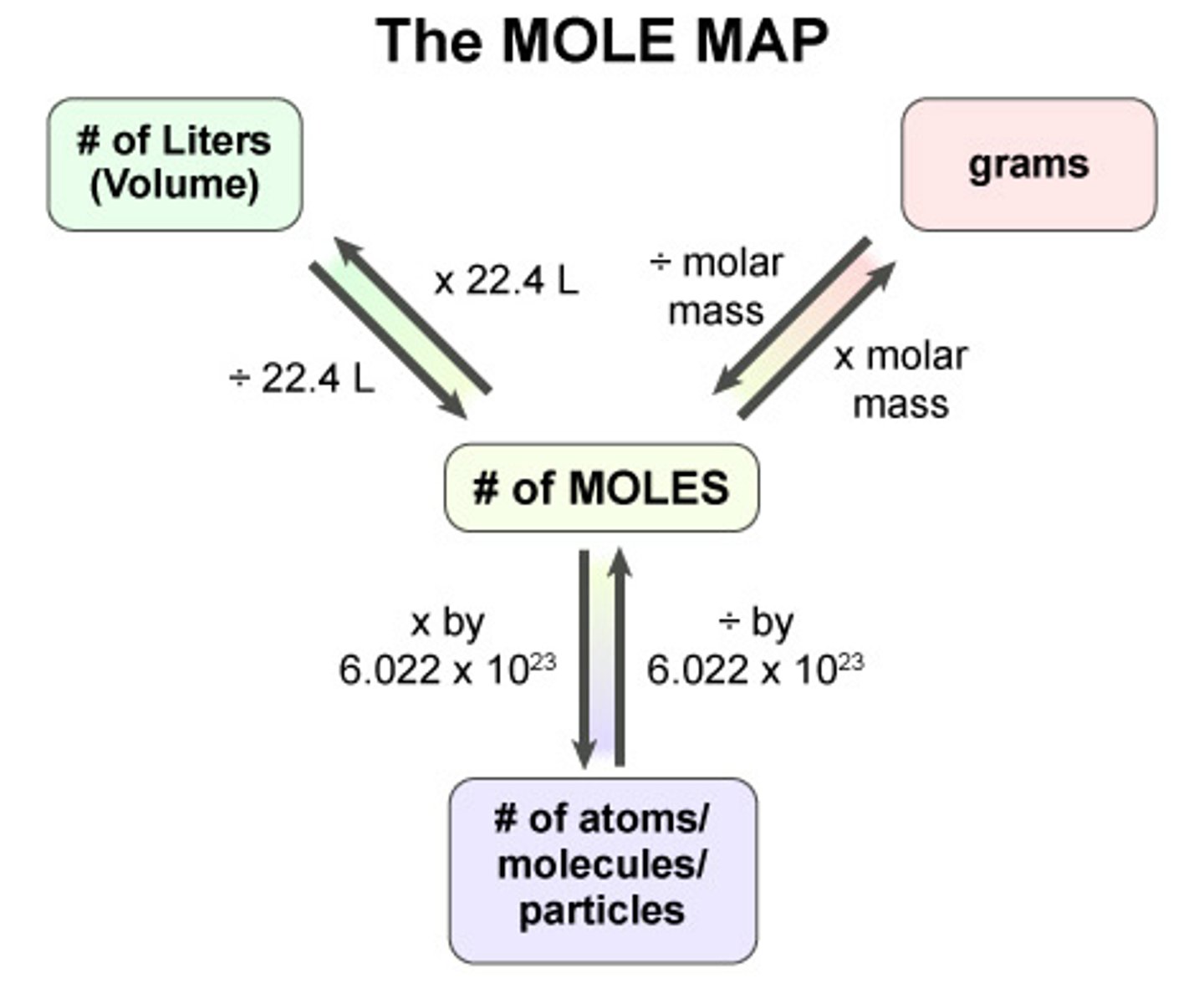
How do you calculate density.
Density = mass/volume

significant figures
All the digits that can be known precisely in a measurement, plus a last estimated digit
How many significant figures are in the measurement 811.40 grams?
5
3 multiple choice options
How many significant figures are in the measurement 0.0034 kg?
2
3 multiple choice options
Are leading zeros significant figures. EX: 0.00
no
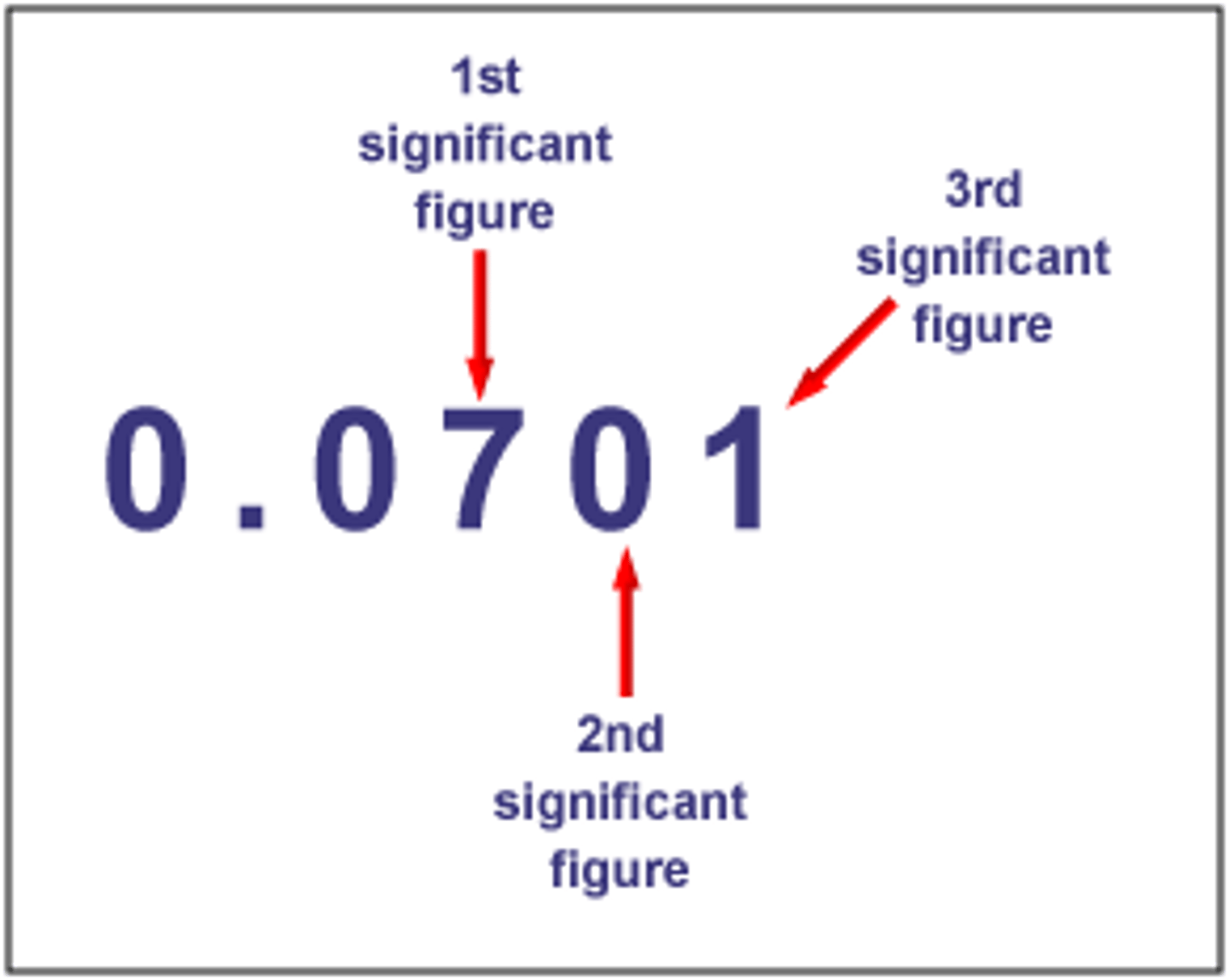
trailing zeros with a decimal (2.300) OR (50.0)
significant
trailing zeros with NO decimal (2300) OR (5000)
NON SIG FIG
Accuracy
how close a measurement is to the true value

Precision
a measure of how close a series of measurements are to one another
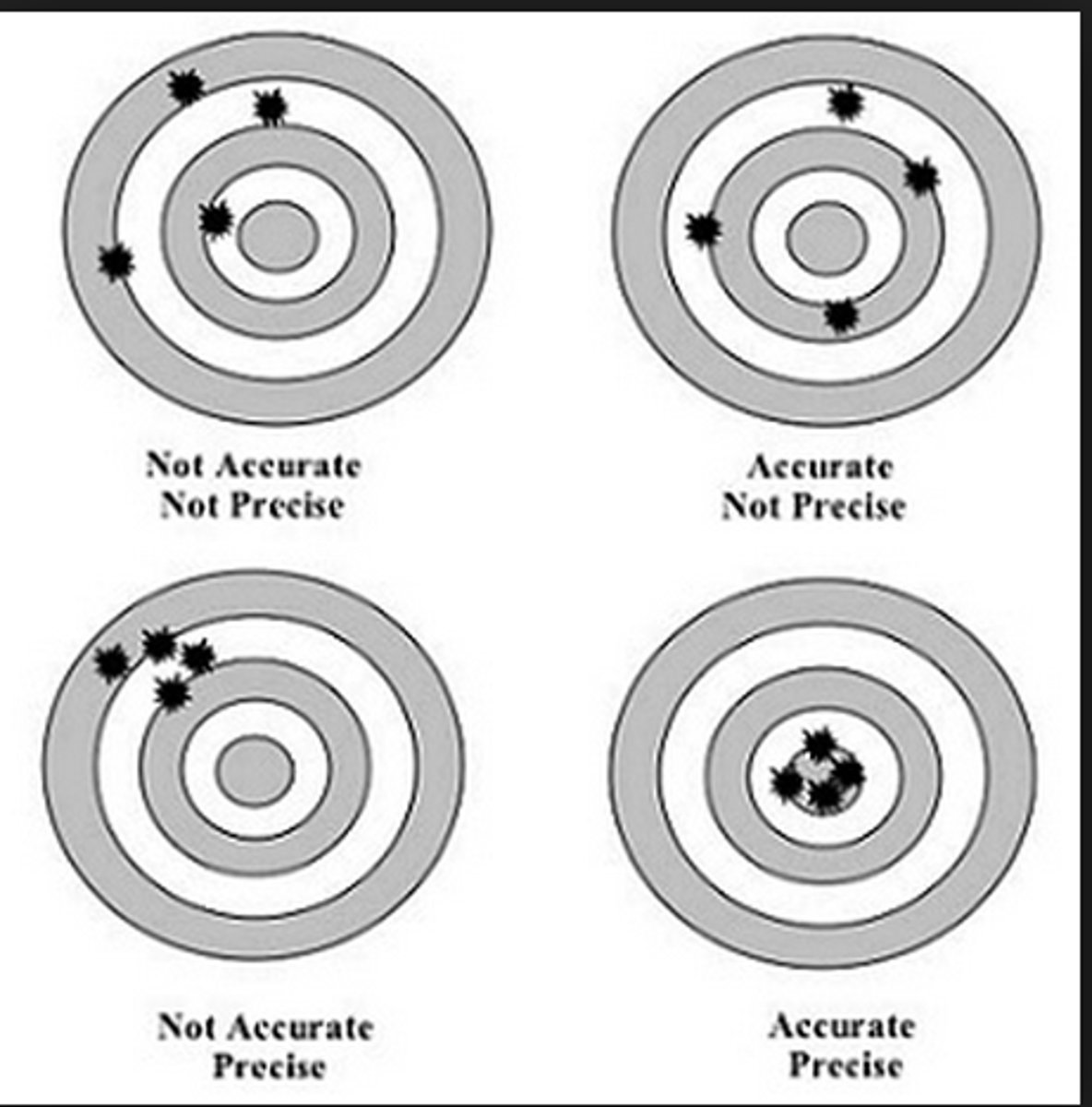
Precision vs. Accuracy
precision is consistency of output whereas accuracy is alignment with the targeted value or goal
formula of a hydrate
n= moles of water / moles of anhydrous salt
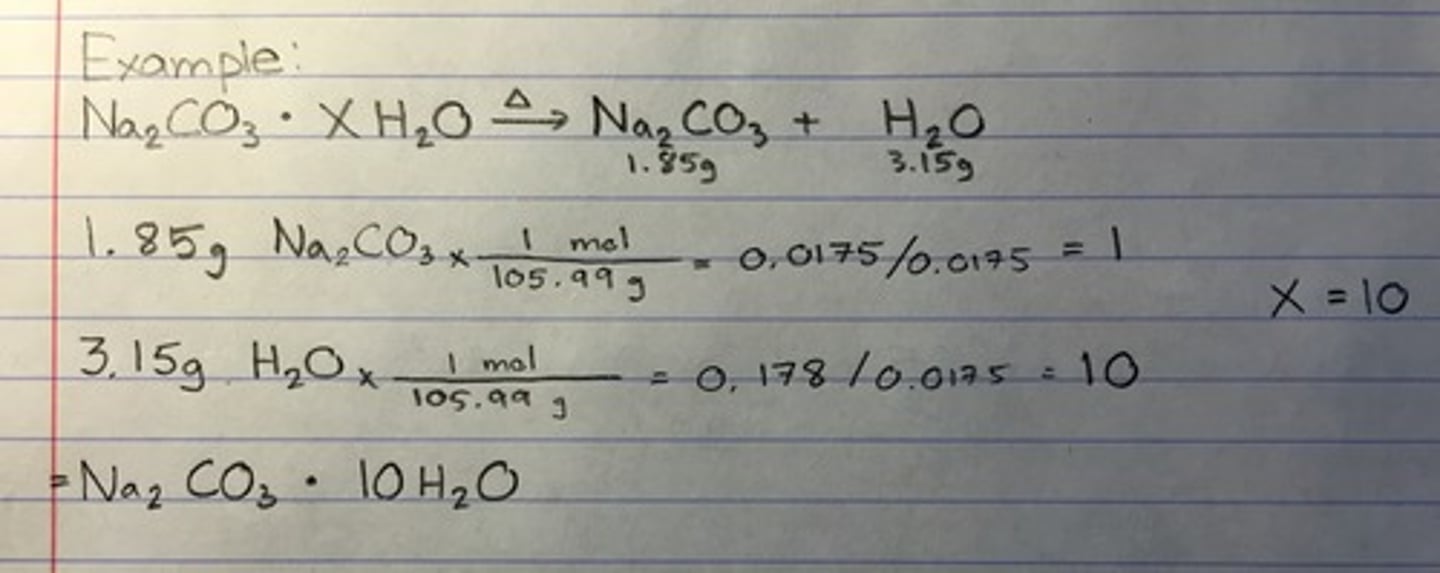
Choose the correct option regarding Lab: Determining formula of hydrate:
'Weighing to constant mass'
Was achieved by making sure that successive weight measurements of the anhydrate were very close to each other

What is a hydrate?
a compound that contains water of hydration

what do we do to remove water from a hydrate
we use heat
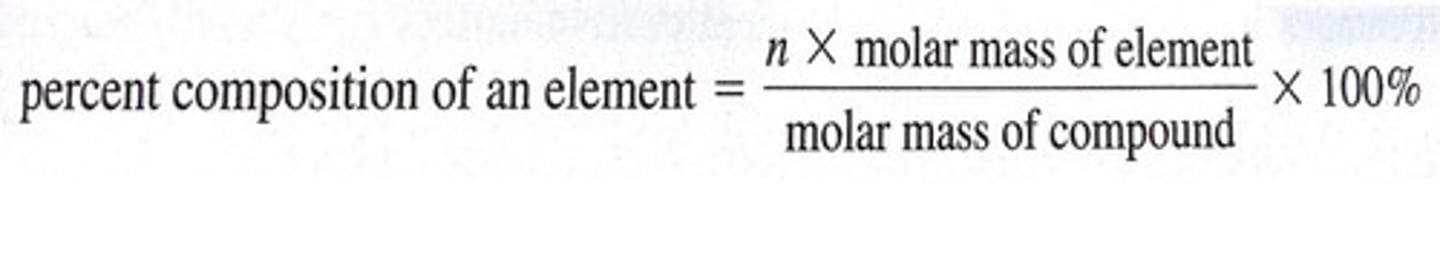
What is an anhydrate?
the substance that remains after the water is removed from a hydrate

empirical formula
The simplest whole number ratio of atoms of each element present in a compound

limiting reactant
the reactant that limits the amount of the other reactant that can combine and the amount of product that can form in a chemical reaction
What is theoretical yield?
the maximum amount of product that can be produced from a given amount of reactant

with limiting reactant/ theoretical yield what do i need to know
Convert grams-> moles
compare mole ratios
smaller amount= limiting reagent
use limiting reagent for yield
What is a redox reaction?
A chemical reaction involving the transfer of one or more electrons from one reactant to another.
What is another name for a redox reaction?
Oxidation-reduction reaction.
Oxidation
loss of electrons

reduction
gain of electrons
In lab what element was oxidized
Al

What element was reduced
Cu2+ -> Cu(s)

If there are three moles of H2O for every 1 mole of LiClO4, then what is the hydrate formula
LiClO4 x 3H2O
3 multiple choice options
Hydrate stoichiometry
MgSO4 x ? H2O
What do you need to find moles of H2O
1. find the water lost (hydrate mass- anhydrous mass)
2. Convert water to moles.
(grams of water loss / molar mass of H2O (18.02))
3. Convert given salt mass to moles.
(Given mass of salt / molar mass of given salt = moles)
4. divide both moles by the smallest mole to get ratio.
A 5.018 gram sample of a certain hydrate of magnesium sulfate, MgSO4•xH2O, is heated until all the water is driven off.
The resulting anhydrous compound weighs 2.449 grams.
How many moles of water does the hydrate contain?
(Atomic weight: Mg: 24.305 g/mol, S: 32.065 g/mol, O: 15.9994 g/mol, H: 1.0079 g/mol)
MgSO4 x 7 H2O (1:7 ratio)

What happened to the copper metal during the recovery experiment?
Copper metal began dissolving.
What color did the solution turn during the copper recovery experiment?
The solution turned blue due to the formation of Cu²⁺ ions (copper nitrate).
What toxic gas was produced during the copper recovery experiment?
Brown toxic gas (NO2) was produced.
What was observed during the copper recovery experiment?
Fumes were observed.
What change in temperature was noted during the copper recovery experiment?
The beaker may have become warm.
What formula do you need to have recovery yield of copper
%yield = (final recovered Cu/ initial Cu) x 100

CuSO4 (aq) + Zn (s) -> Cu (s) + ZnSO4 (aq)
What describes this reaction
copper is reduced and zinc is oxidized
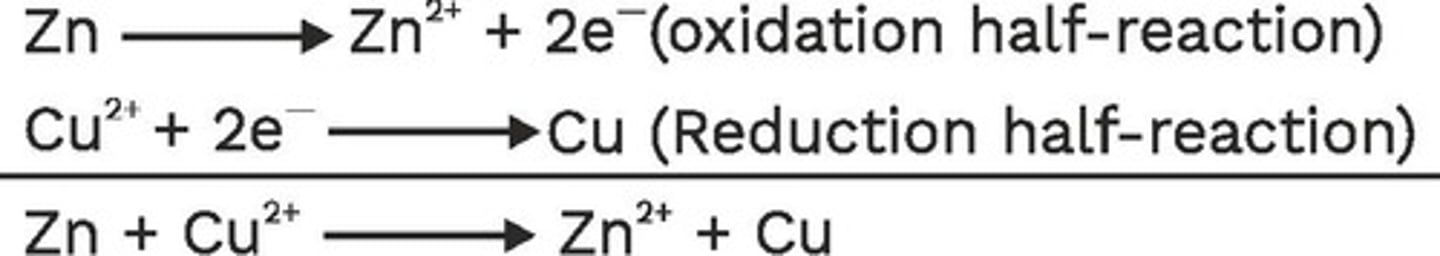
Cu+2 -> Cu(s)
what is this?
reduction
gains electrons
Zn(s) -> Zn+2
Oxidation
loses electrons
OIL RIG
oxidation is loss, reduction is gain
What is distillation?
separates liquids with different boiling points
double replacement
a chemical reaction where two elements in different compounds trade places
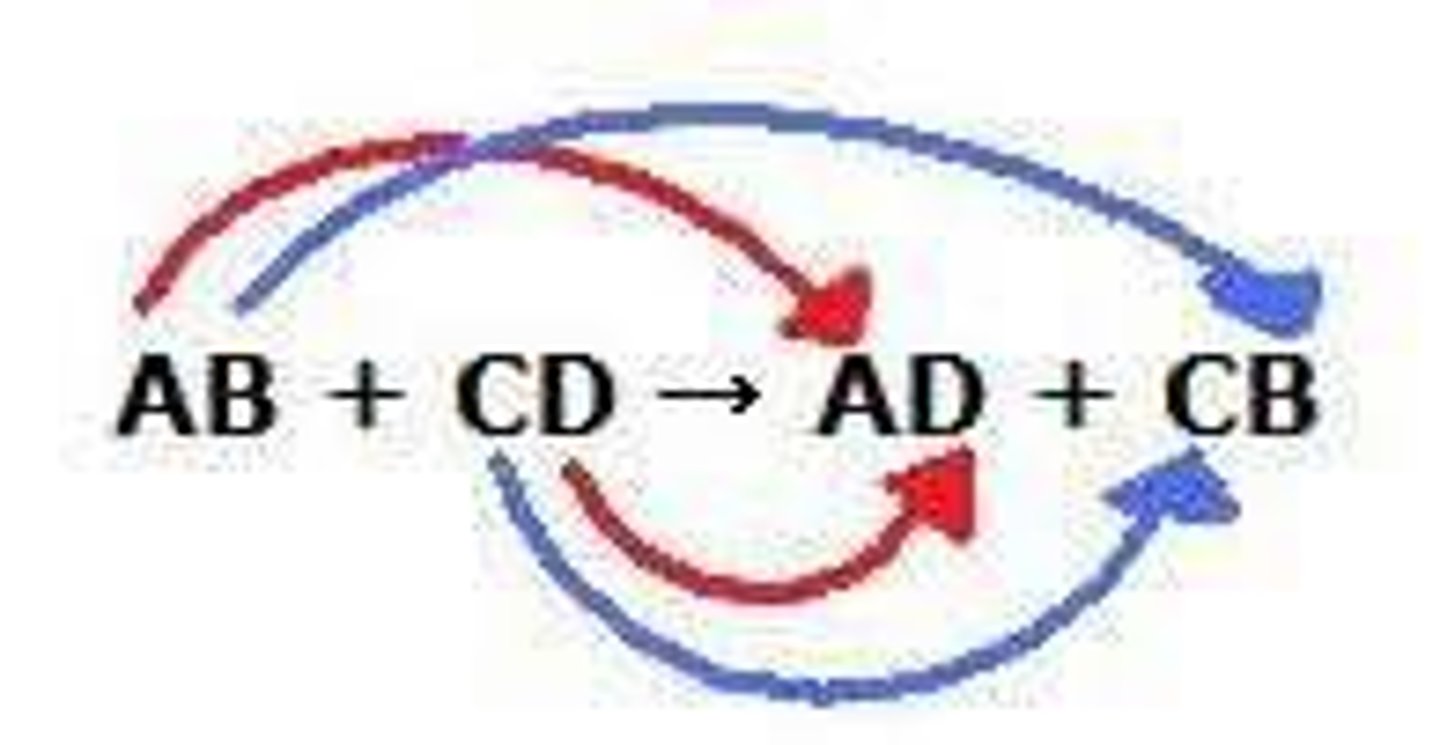
double replacement reaction (metathesis)
AB + CD --> AD + CB
precipitation reaction
a reaction in which an insoluble substance forms and separates from the solution

Single Replacement (Displacement)
one element replaces another element in a compound

synthesis reaction
a reaction in which two or more substances combine to form a new compound
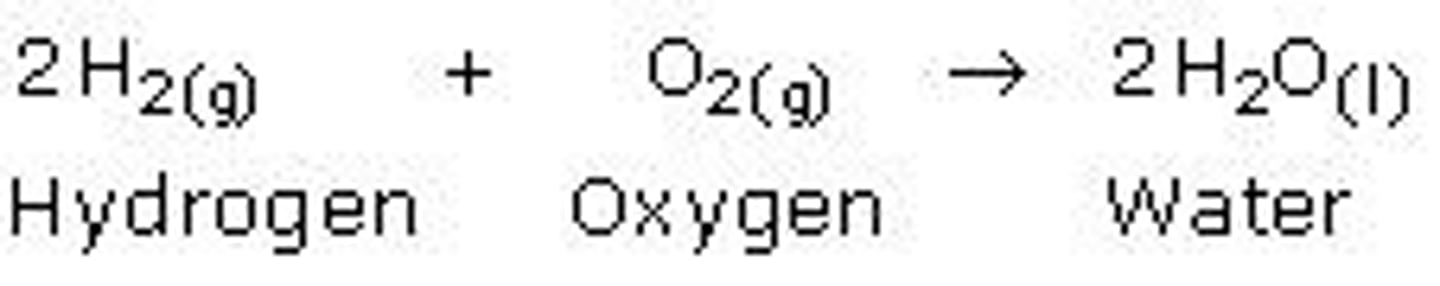
decomposition reaction
AB --> A + B
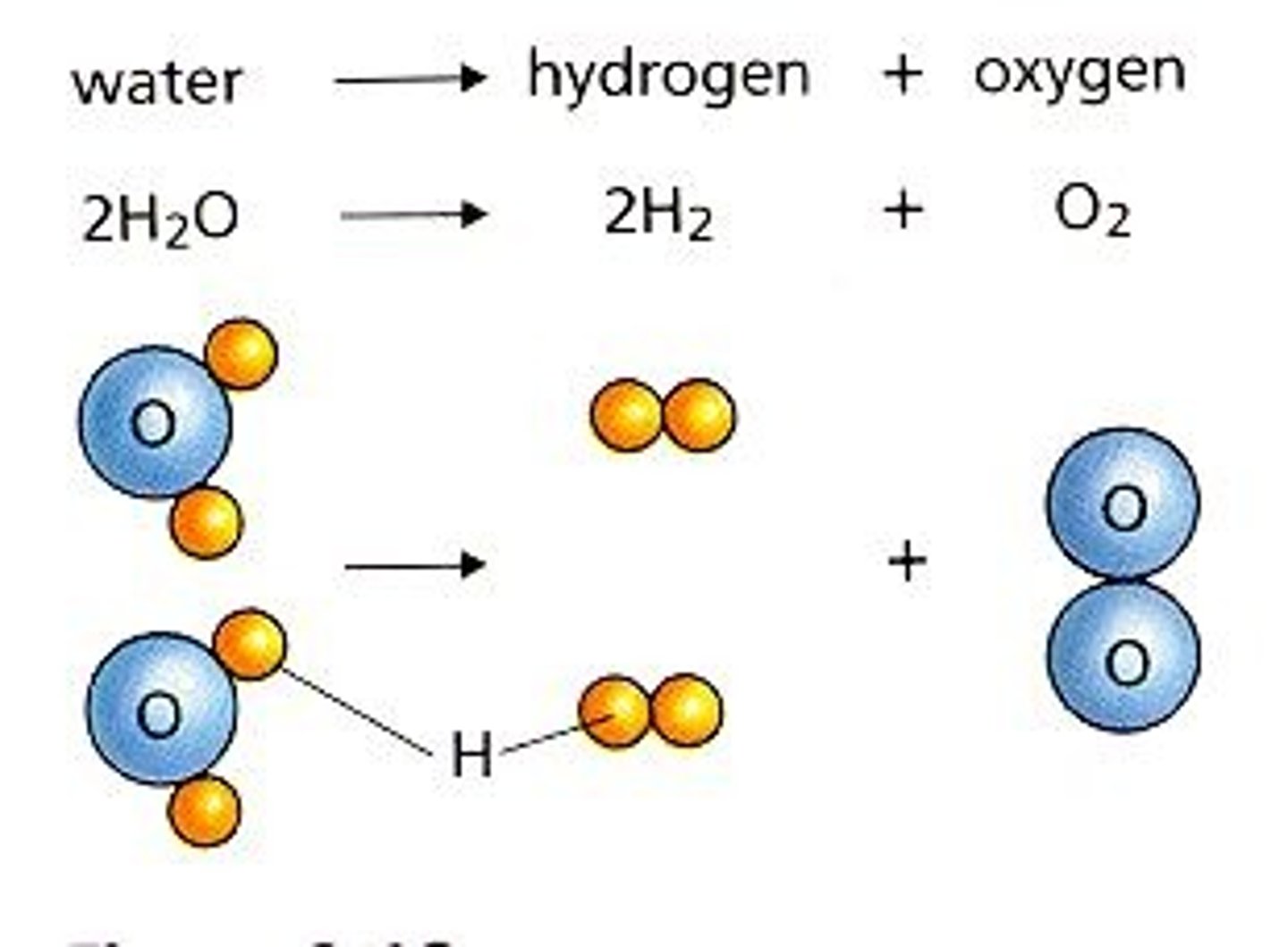
Decomposition
A chemical reaction that breaks down compounds into simpler products.
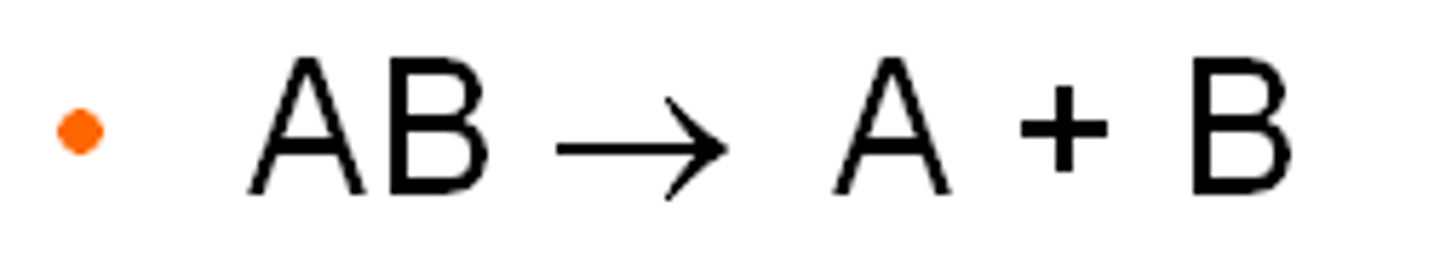
What is a combustion reaction?
A chemical reaction that occurs when a substance reacts with oxygen.
What energy is released during a combustion reaction?
Energy in the form of heat and light.

What is the organic compound in the mixture of oleic acid, iron, sand, salt, and zinc oxide?
Oleic acid
Why would we have to remove propanol first from the mixture?
Because only oleic acid is an organic compound and soluble in propanol.
TRUE OR FALSE
iron, sand, salt, and zinc oxide do not dissolve well in propanol
TRUE
Experimentally, you can separate two liquids from each other by
distillation
Methods to separate a solid from a liquid
filtration, decantation, evaporation
what equipment was used to separate solvent from oleic acid?
hot plate
What is acid-base titration?
A laboratory procedure to determine the concentration of an unknown acidic or basic solution.
What type of solution is used in acid-base titration to determine concentration?
A basic (or acidic) solution of known concentration.
What is the purpose of acid-base titration?
To determine the concentration of an unknown solution.
What is the equivalence point?
The point in a titration when neutralisation is reached (i.e. when moles H+ = moles OH-)
What is an acid-base indicator?
a chemical dye whose color is affected by acidic and basic solutions
What is an end point?
the point at which the indicator that is used in a titration changes color
equivalence point
when the acid and the base neutralized each other
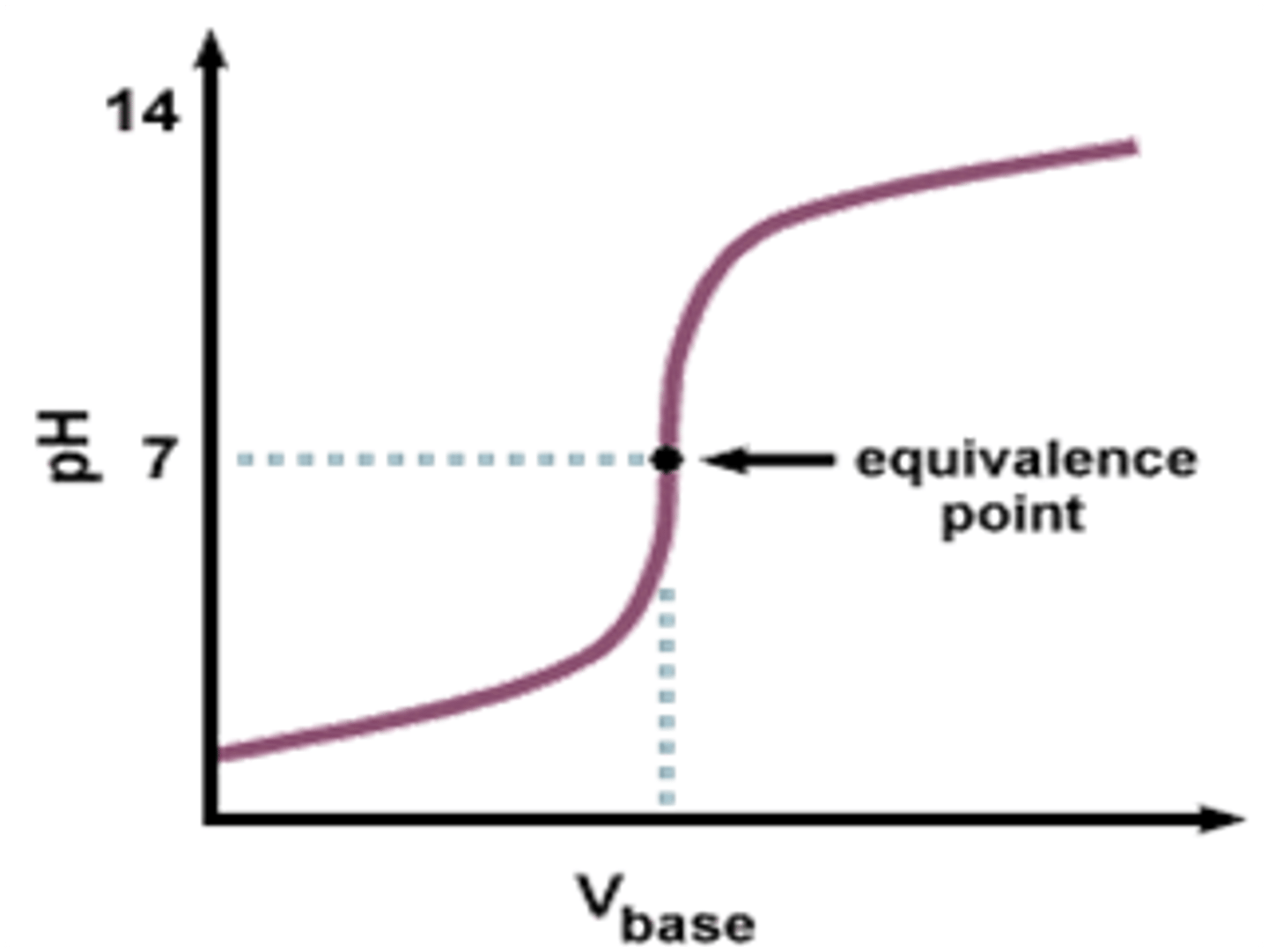
what does it mean when its a faint pink
its at the endpoint

What is the formula to calculate moles in a titration?
moles = M x L
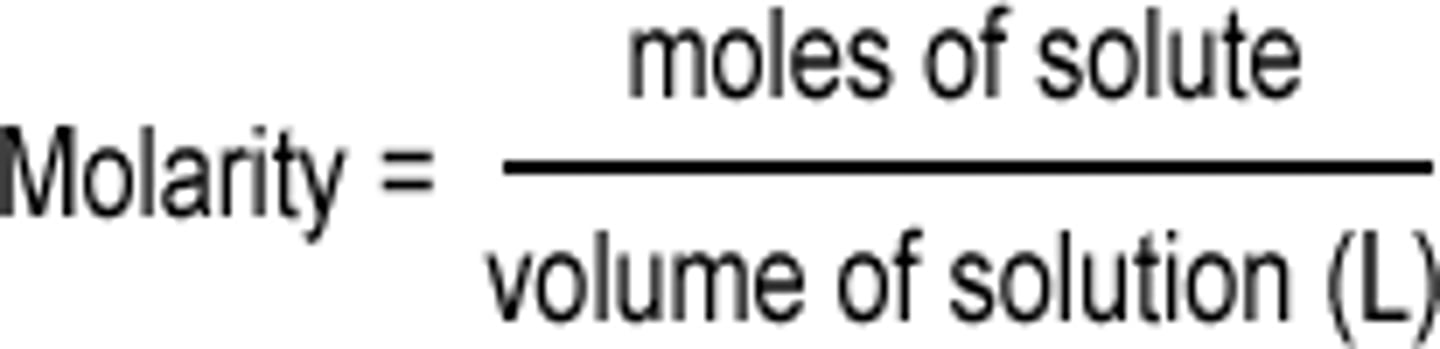
What does M stand for in the moles formula?
Molarity
What does L stand for in the moles formula?
Liters
How do you convert mL to L?
Move 3 decimal places to the left
What do you multiply to find moles in a titration?
Molarity (M) and Liters (L)
Titration Indicator (Phenolphthalein)
colorless in acidic solution. pink in basic solution

titration molarity calculation
The equivalence point in a titration of 20.0 mL of HCl with 0.500 M NaOH was determined to be at 13.5 mL. What is the molarity of HCl and the number of moles of NaOH at equivalence point?
The molarity of HCl is 0.337M and the moles of NaOH is at equivalence point is 0.00675
WHAT IS GIVEN?
HCl vol- 20.0 mL
NaOH - 0.500 M
EQP REACHED- 13.5 mL NaOH
HCl + NaOH -> NaCl + H2O
*look at the ratios. Only 1 HCl and Only 1 NaOH ( ratio 1:1)
*find moles of NaOH.
- convert 13.5 mL --> xL --> 0.0135 L
*Now do the formula moles= molarity x Liters
- 0.500 x 0.0135 = 0.00675 mols
*Find molarity of HCl M= mol/L
- 0.00675 mol / 0.0200 L= .3375 molarity
HCL molarity - 0.337M
NaOH moles - 0.00675
Gas law
PV=nRT
If a gas's Kelvins Tempature doubles and volume remains constant, what happens to pressure
The pressure doubles
Gay-Lussac's Law
the pressure of a gas is directly proportional to the Kelvin temperature if the volume is constant
Gay-Lussac's Law Equation
P1/T1 = P2/T2
according to the ideal gas law, what are the units for the gas constant R
volume x pressure/ (moles x Kelvin
how was hydrogen gas generated in our lab experiment on gases (part ii)
Reaction of Mg with HCl
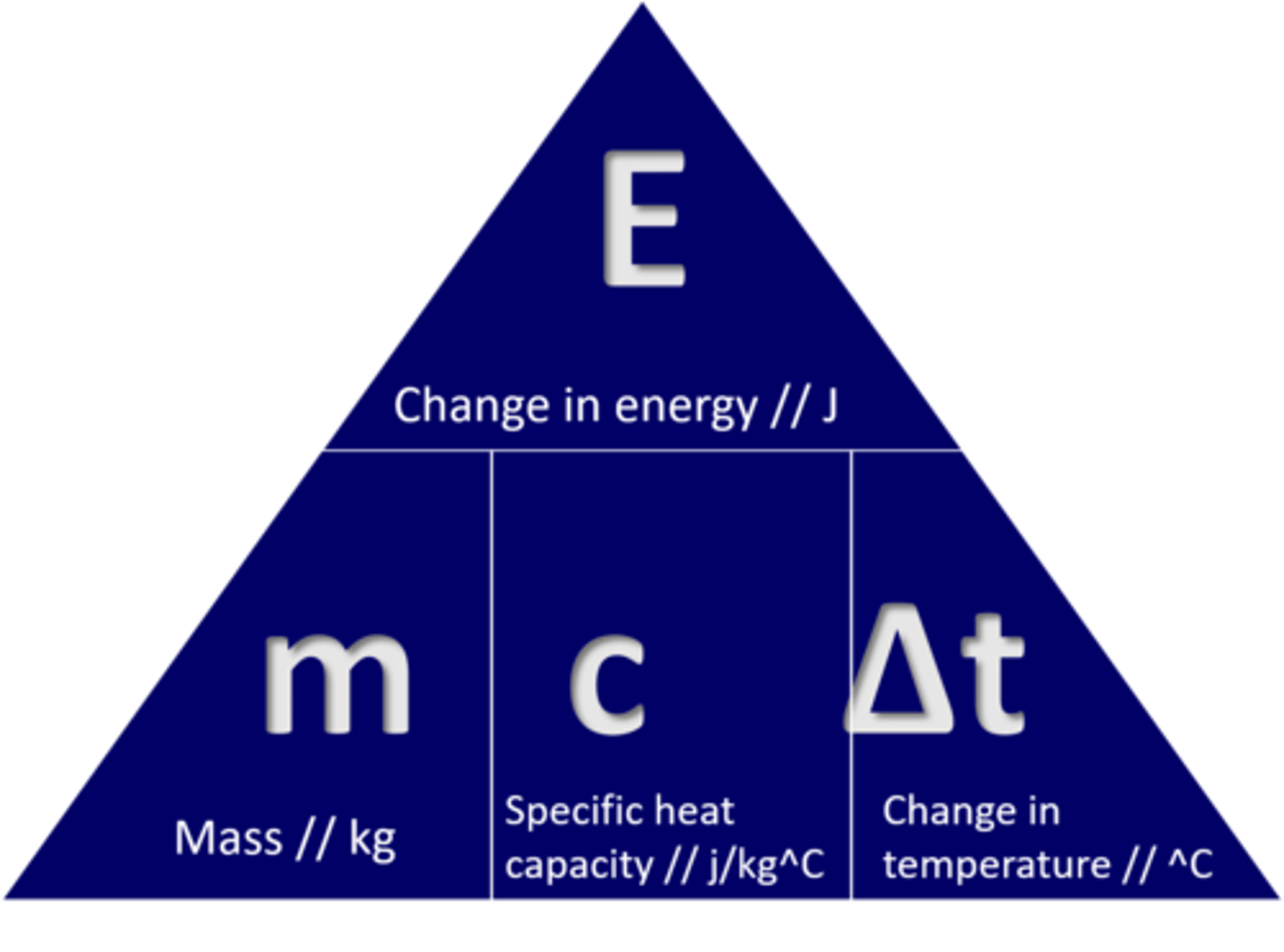
What is specific heat capacity?
The amount of energy required to increase the temperature of 1kg of a substance by 1oC
Given that the specific heat capacity of water is 4.2 J / (g Celcius). The heat capacity off 80 g of water in j/c is ...?
HEAT CAPACITY OF AN OBJECT IS C= M X C
M= 80 G
C= 4.2 j
M x C --> 80 x 4.2= 336 j/c*
heat equation
q=mc∆T
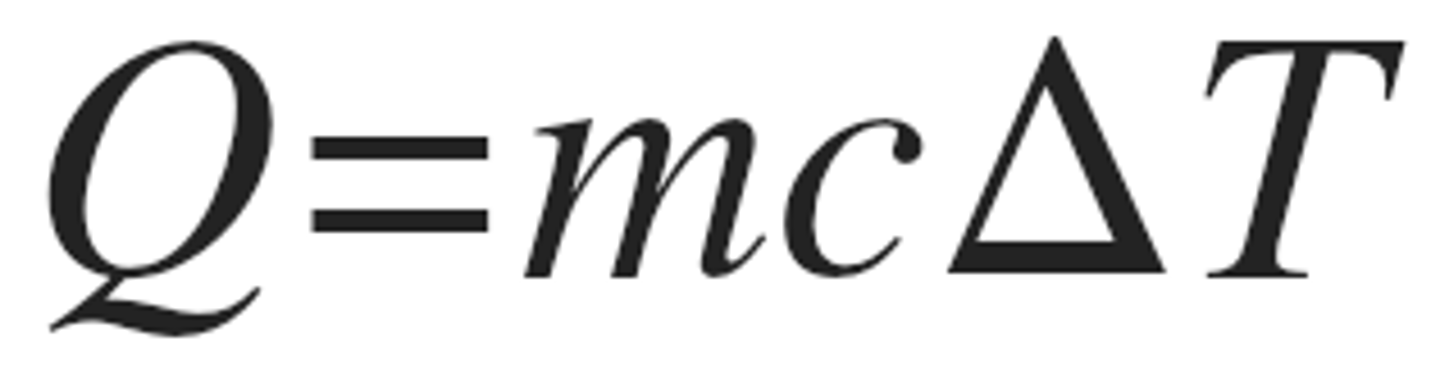
If 2016 J of heat are gained by 70 g of oil, what is the tempature change in oil.
Oil specific heat capacity is 2.4 j (gxC*)
GIVEN
q= 2016 j
m= 70 g
c= 2.4 J/(gxC*)
∆T=??????
Original equation --> q=mc∆T
REARRANGE
∆T= q/mc --> 2016/70x2.4= 12
a displacement reaction will occur when ....
a more reactive metal displaces a less reactive metal from its compound
Reactivity series
A list of metals which shows them in order of their reactivity, with the most reactive at the top.

what is the product of a reaction between magnesium metal and hydrochloric acid
magnesium chloride and hydrogen gas
What is a flame test?
Testing chemicals by burning a compound to look at its flame color.
What do certain compounds and elements produce when burned?
Distinctive flame colors.
What factor influences the color produced in a flame test?
Electron configuration.
Potassium Flame Color
lilac

Sodium Flame Color
yellow-orange

Lithium Flame Color
crimson

Calcium Flame Color
Brick red

Barium Flame Color
yellow-green

a flame test can be use to identify _________ cations
metal

Copper Flame Color
blue-green

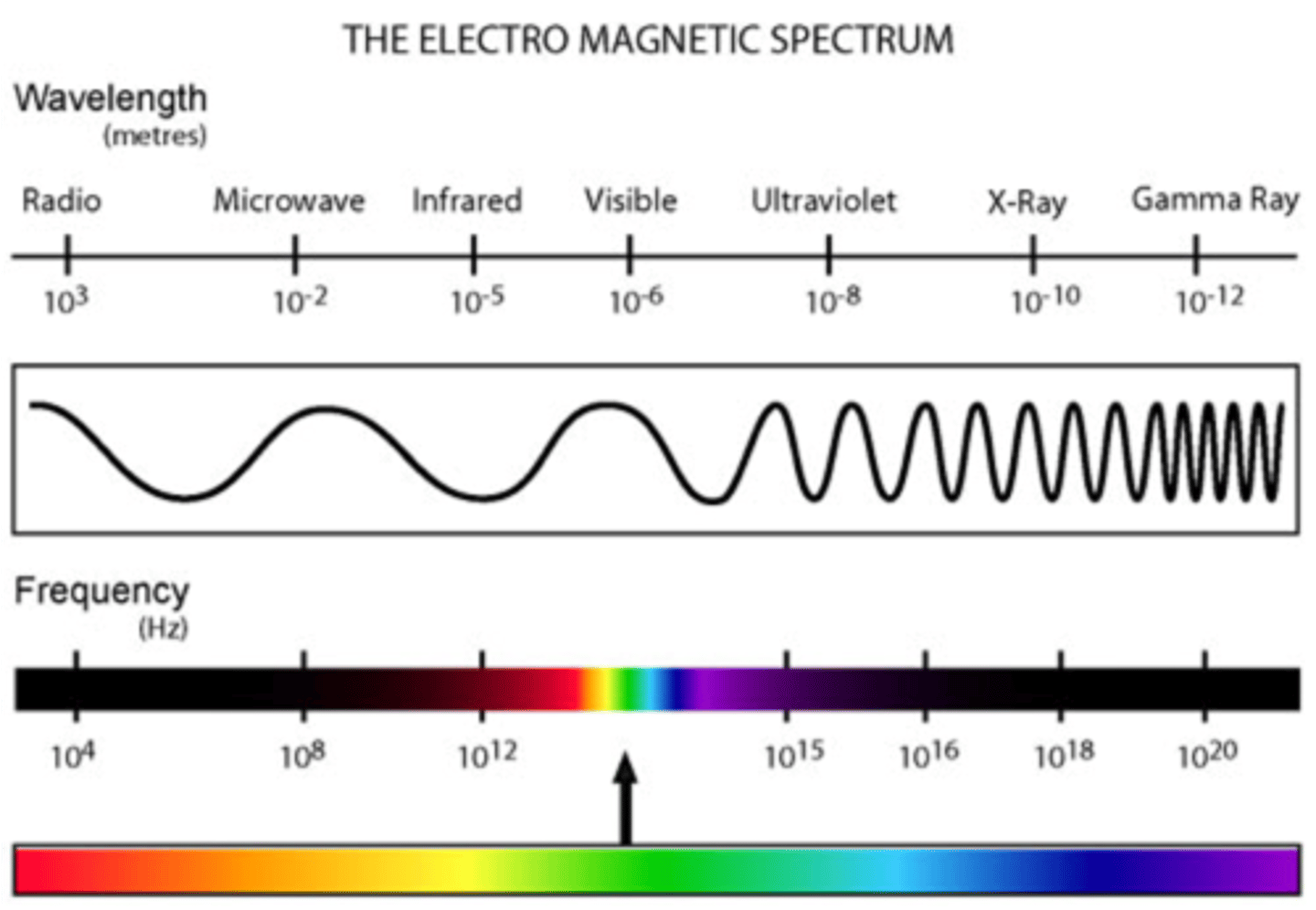
which portion of the visible spectrum has longer wavelengths
RED
NaCl and Water is a strong ______
electrolyte
TRUE OR FALSE
A strong electrolyte conducts electricity because it ions can move in response to electrical forces
TRUE
The pH of chicken soup is 5.80. Which of the following statements apply to chicken soup?
*look at picture*
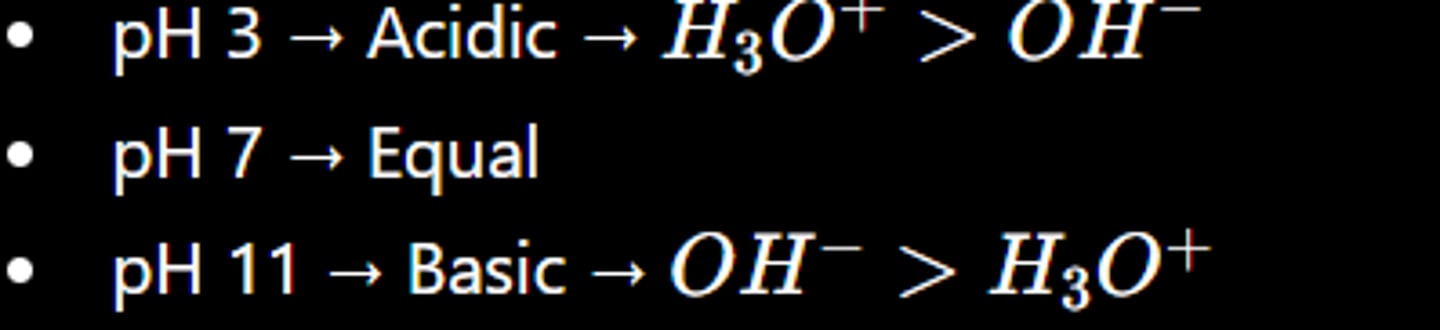
Sources of error in pH and conductivity experiment.
-pH probe not calibrated properly
-Probe not rinsed between samples
- Probe not dried, causing dilution
-Cross-contamination between solutions
-Dirty conductivity electrodes
-Not enough time for reading to stabilize
-Temperature changes affecting readings
-Wrong volume/concentration of solution
-Misreading meter values
-Recording data incorrectly

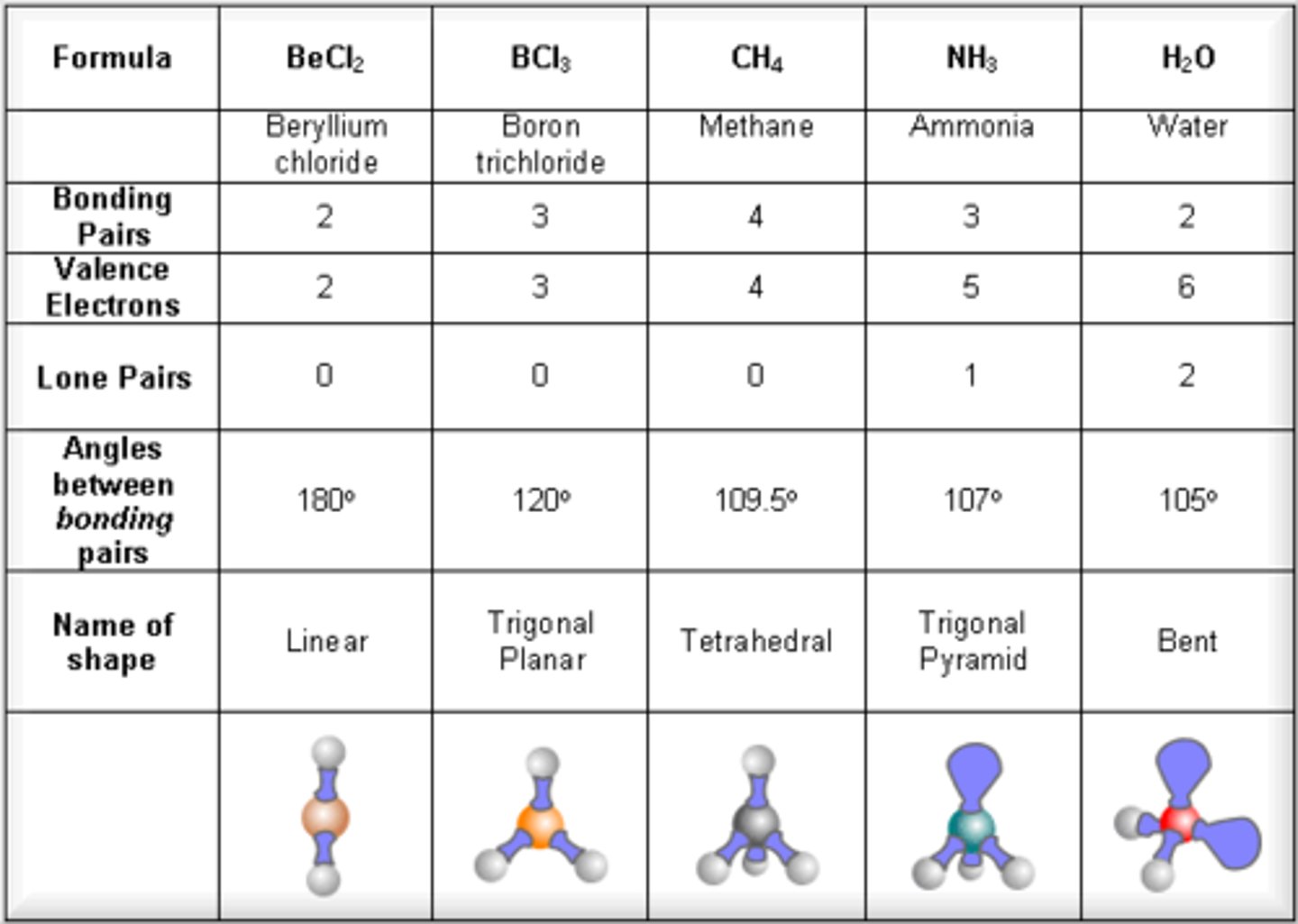
What is electronic geometry?
The spatial arrangement of all pairs of electrons around a central atom.
What types of electrons are considered in electronic geometry?
Both bonding electrons and lone pairs of electrons.
common electronic geometries
-Linear
-Trigonal planar
-Tetrahedral
-Trigonal bipyramidal
-Octahedral