Bio R and A final
1/158
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
159 Terms
accuracy
true value
precision
how close your measurements are to eachother
mass
a measure of the amount of matter an object has
weight
a measure of how strongly an object is pulled by the planets gravity
Weight is an accuracte indicator of
mass on Earth

Top loading
weights .01g

Analytical balance
weights to .0001g
why does an analytical balance have a wind screen
bc pan is so sensivtive
Microbalance
weighs to .000001g (=10^-6 g= 1 microg) or even less
round to
second to last digit
be sure surface of balance is
level (bubble must be cenetered)
1mL=
1cc
1mL of H20 weighs
1g
1L of H20 weighs
weighs 1kg
serological pipettes
many graduations, can measure many values
adjustable pipettors
smaller measurements
beakers and erlenmyer flasks
good for mixing solutions
neither accurate or precise for measuring volumes
Graduated cylinders
good precision and accuracy
not ideal shape for mizing
volumetric flasks
excellent accuracy and precision
shape is ideal for swirling
can only be used to measure one volume
water can cling to
glass, dispensing slightly less than originally poured in
TC
to contain
TD
to deliver
when an instrument marked TC is filled, it ______
when emptied, it will_____
contains that volume
deliver less than that volume, due to clinging
TD instruments actually contain ____ but they deliver ___
slightly more than the marked volume
the specified amount
cant use TD for liquids
with different viscosity than water bc a greater amount might cling
can use TC for
liquids other than water
Use TD if you are
using water and care most about the volume that comes out
Use Tc if you care
most about how much is inside
pipettes labeled
TD
Importance of water in biological research
important component of many solutions
water quality, composition, and pH can affect experimental outcomes
Tap water
variable chemistry and purity depending on region and local treatment of municipal water
Lab grade water
from reverse osmosis or distillation
for rinsing glassware, prep of some growth media
reagent grade water
from filtration, deionization, and carbon absorption
used when highest water purity required
solution
a mixture in which molecules are dispersed into a liquid
solvent
does the dissolving
majority of a solution
water or alc
solute
dissolved
minority of a solution
often a solid before mixing
Units of concentration
amounts of solute per volume of solution
1 part per million
1 mg/L
parts per hundred
%
weight per volume
mg/L or microg/L
molarity unit
mol/L
concentration as %
% by volume/volume
mL per hundred m: of solution
Molarity
moles of solute per L of solution
the mass of 1 mole of a compound is the
molecular weight/formula weight of that compound
to prepare a solution of a given molarity, need
volume and concetration desired
MW/FW of solute
A 1M solution is the
MW or FW of a substance in 1 L of solution
Normality
molarity of equivalents produced by a compound in solution
for many chemicals the molarity and normality are
the same
differences occur when molecules have >1 exchangeable proton
To make very dilute solutions
make a concentrated stock solution and then dilute from stock
Dilution from 1 concentration to another formula
C1V1=C2V2
c1=initial concentration
v1= initial vol
c2= desired concentration
v2- desired final volume
add water to
acid not acid to water
prevents splashing and over-heating
full strength is not
necessarily 100%
types of two group comparisons
independent samples
related samples
independent samples
each observation in sample 1 has no special relationship ito an observation in sample 2
related samples/matched pairs
each individual is measured twice
carefully matched pairs of individuals are measured
Independent samples parametric
independent samples t test
Independent samples nonparametric
Mann-Whiteney Test (Wilcoxon rank sum test)
related pairs parametric
paired t test
related pairs parametric nonparametric
wilcoxon signed ranks test
assumptions of independet samples t test
the two samples are unbiased samples from the population of interest
the measured variable is continuous and normally distributed
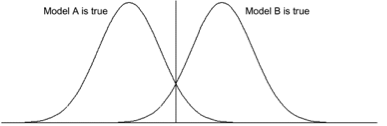
two normal groups with equal variances
two normal groups with unequal variance
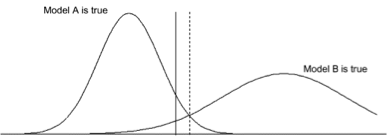
we will use _____ to test for equality of variances
Bartlett’s test
null and alternative for Bartlett’s test
Ho: variances of the 2 groups are equal
Ha: variance are uneqal
if p<a, we conclude that the two varinces are unequal
Welch’s test uses fewer
df, so its lest powerfull that bartletts
the null hypothesis states that
there are no differences between the two pop means
null and alternative for two tailed test
Ho: mua=mub
Ha: mua does not equal mub
equation for test statistic
mua-mub is usually 0
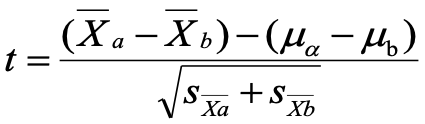
Mann whitney test
tests whether medians of 2 pops differ
null and alternative for mann whitney test
Ho: median a=median b
Ha: median a does not equal median 2
assumptions of mann whitney test
2 population distribution have the same shape
have unbiased samples drawn from two pops
in mann whitney, data from the 2 samples are
first combined and then ranked
lowest value is ranked 1
tied scores are assigned avg of 2 ranks
the outcome of mann whitney depends on
the sum of ranks in eahc group
more variable delta, higher
SE
Paired t test assumptions
each individual is measured twice or that matched pairs of individuals are measured
Paired t test 2 tailed null and alternative
Ho: mud=O
Ha: mud does not equal O
mud is pop mean difference
Paired t test 1 tailed null and alternative
Ho: mudO
Ha: mud d< O
tesitng Ho in a pair t test
test stat (t cal, SE, n)
compare tcalc to t critical (from t table) using df
tcalc
mean/SE
SE
SD/sq rt(N)
Wilcoxon signed ranks test
nonparametric test for paired measurements
used when the differences can be ranked with respect to eachother
why use wilcoxon ranks test
data are continuous or discrete, but not normally distributed
OR we have ordinal scale ranks
Ho for 2 tailed test wilcoxon ranks test
sum of positive ranks in a pop of differences=the sum of negative ranks in a pop of differences
ANOVA
analysis of variance
tests whether the means of three or more groups differ significantly from eachother
the problem with multiple pairwise comparisons
the probability of committing a type 1 error increase for each comparison tested in the following way: 1-(1-x)^x (where x is the number of pairwise comparisons)
ANOVA allows for
multiple pairwise comparisons without an inflated change of type 1 error
variance equation
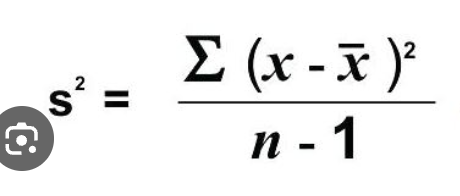
Single Factor ANOVA
completely randomized design with fixed effects
Single Factor ANOVA assumptions
same as for an independet t test
ANOVA partions the overal variance into
2 parts:
variance due to differences between groups (Effect of the treatment imposed)
variance due to uncontrolled differences among subjects within groups
Single factor ANOva hypothesis
Ho: mean1=mean2=mean3=…
Ha: at least 1 pair of means are not equal
No one tailed
ANOVA
unequal variance
Welches test
other sources of variation of mice
body mass, length, age, genetic variation
The F distribution/fcalc
used in ANOVA
F= treatment (between groups) variance/error (within groups) variance
a large Fcalc suggest that
treatment effect compared to random error
a small Fcalc suggests that treatment effects
are no greater that the random variation in a pop
if treatment has no effect, expect Fcalc to be
close to 1
if treatment has large effect expect Fcalc
to be »1
How to test Ho for ANOVA
compare Fcalc to Fcritical (value from F table)
If Fcalc>Fcritical, then reject Ho