Chemistry Unit 1 Test
1/76
Earn XP
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
77 Terms
Adding/subtracting significant digits
Round to the least precise place value found in originals.
Beaker
A simple container used to hold and mix chemicals.

Bunsen burner
Produces a flame for heating substances during experiments.

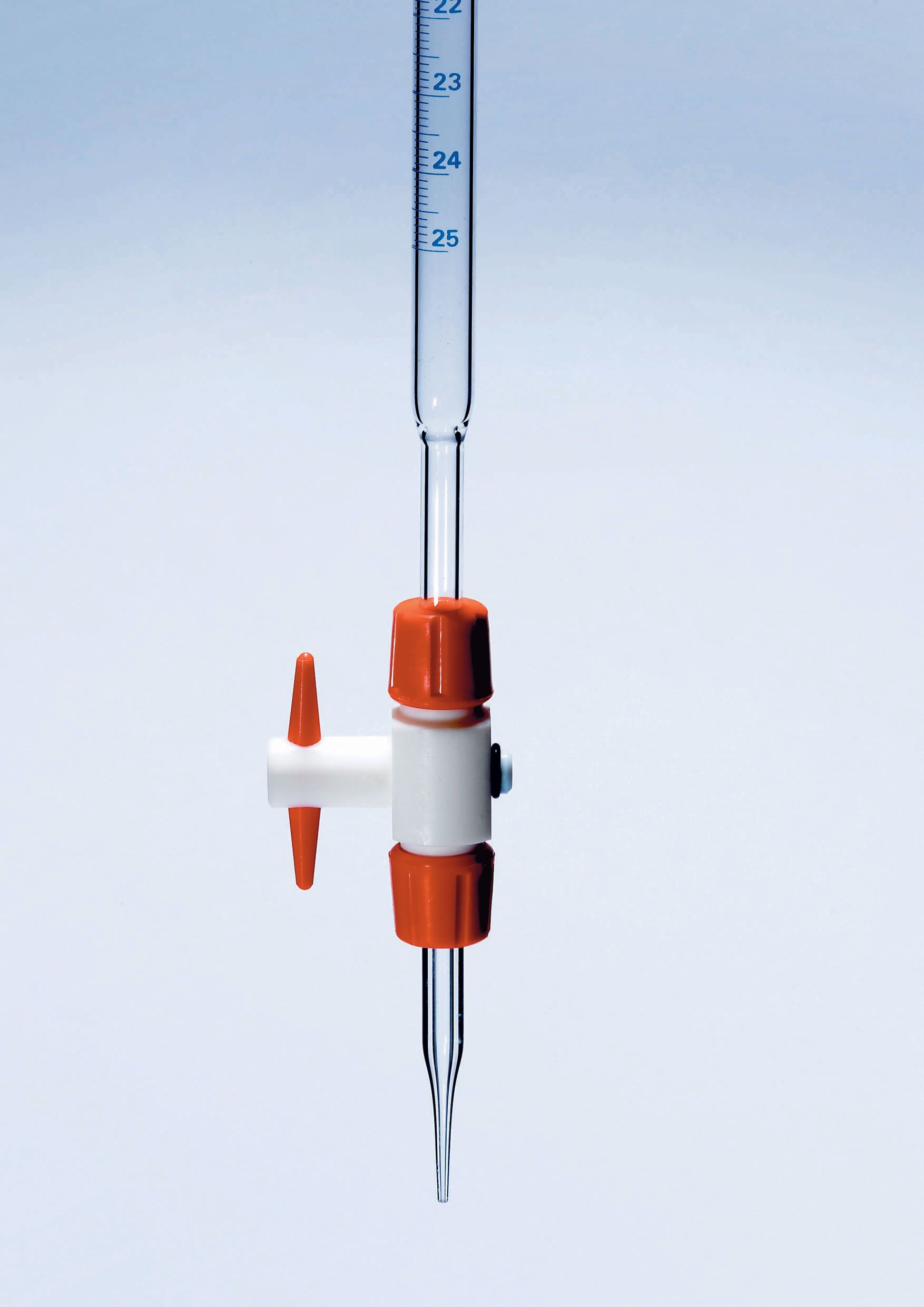
Burette
Measures and dispenses precise amounts of liquid for titrations.
Chemical Properties
Characteristics that describe a substance's ability to undergo chemical changes.
Chromatography
Separates components based on their different movement through a stationary medium.
Compounds
Only one type of particle: molecules of different elements.
Condensation
The process of a gas turning into a liquid.
Critical point
When a substance cannot be distinguished between gas or liquid.
Crucible and cover
Heats small amounts of substances at high temperatures.

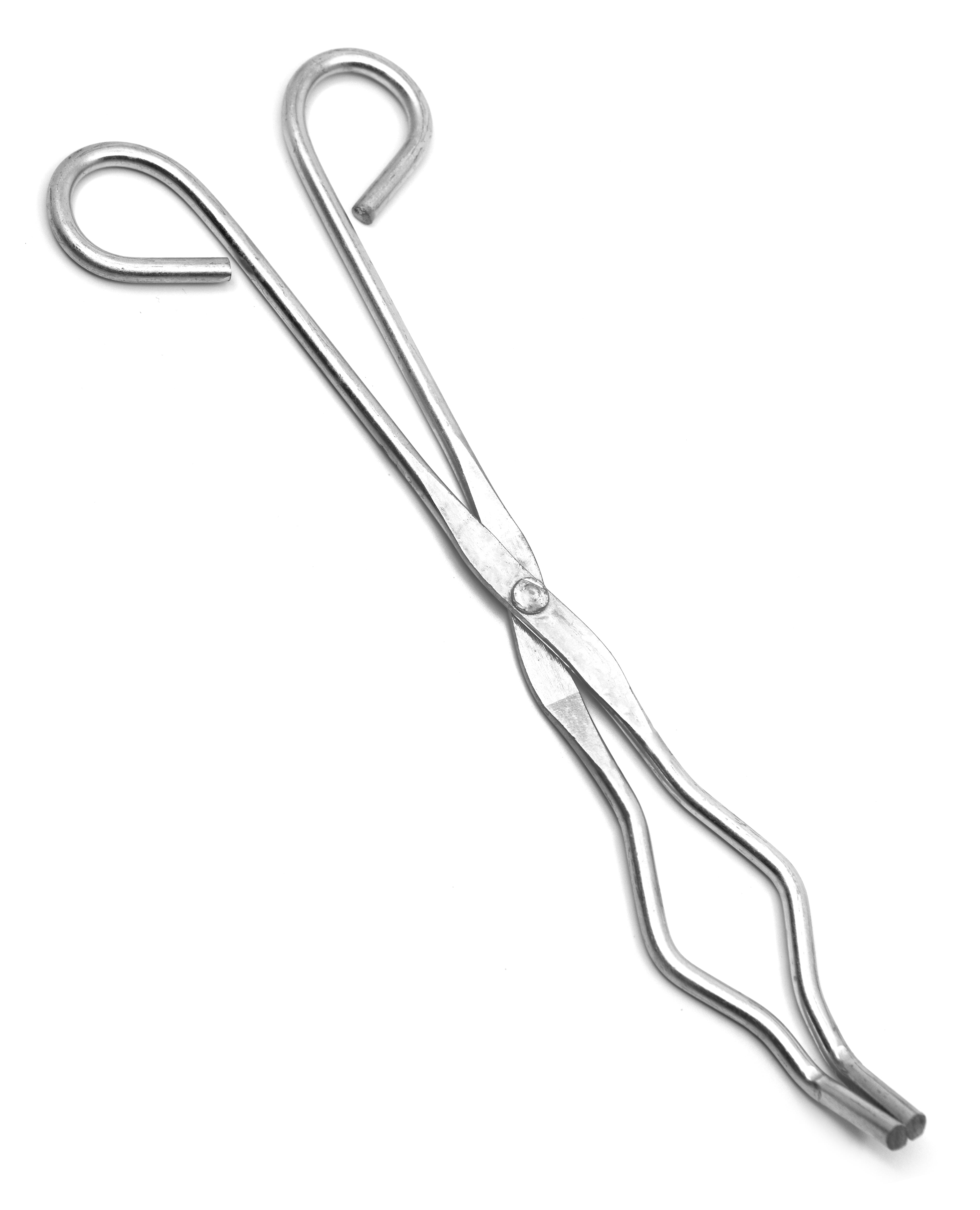
Crucible tongs
Safely handles hot crucibles or other heated items.
Crystallization
Separates a dissolved solid from a solution by cooling or evaporating the solvent to form crystals
Density
A ratio equal to mass divided by volume.
Deposition
The process where a gas changes directly into a solid.
Dimensional Analysis
Done by the fencepost method or fractions: use equivalent ratios to cancel and replace units.
Distillation
Separates components of a solution based on differences in boiling points.
Elements
Pure substances made of only one type of atom.
Erlenmeyer flask
Mixes and stores liquids, often used for titrations.

Evaporating dish
Evaporates excess solvent from a solution.

Evaporation
The process of a liquid turning into a gas.
Evaporation Method
Removes the liquid from a solution by heating, leaving the dissolved solid behind
Extensive Properties
Properties that depend on the amount of substance present (e.g., mass, volume).
Filtration
Separates solid particles from liquids or gases by passing the mixture through a filter
Freezing
The process of a liquid turning into a solid.
Funnel
Used for transferring liquids or fine-grained materials into containers.

Gas
Has neither a fixed shape nor volume, with particles spread far apart.
Goggles
Protects the eyes from chemical splashes or debris.

Golden rules of lab safety
Don’t play around; verify everything with instructor; hot and cold glass look the same.
Graduated cylinder
Measures the volume of liquids accurately.

Heterogeneous
A mixture with visibly different components or phases.
Homogeneous
A mixture with a uniform composition throughout.
Intensive Properties
Properties that do not depend on the amount of substance present (e.g., density, boiling point).
Interfaces of Matter
Boundaries where different phases meet in a heterogeneous mixture.
Lab apron
Protects clothing from chemical spills or splashes.

Liquid
Has a fixed volume but takes the shape of its container; particles are loosely packed.
Magnetic Separation
Uses a magnet to separate magnetic materials from non-magnetic ones. Think of sand and iron!
Melting
The process of a solid turning into a liquid.
Metric Length
Measured in terms of meters.
Metric Mass
Measured in terms of grams.
Metric Temperature
Measured in terms of kelvin.
Metric Time
Measured in terms of seconds.
Metric Volume
Measured in terms of liters.
Mixtures
Physical combinations of two or more particles.
Mortar and pestle
Grinds solids into powders for easier dissolution or reactions.

Multiplying/dividing significant digits
Round to the least number of significant digits found in originals.
Normal/standard pressure
1 atm
Phase change diagram
Models removal and addition of heat.
Phases of Matter
Components of a mixture
Physical Properties
Characteristics that can be observed or measured without changing the substance's composition.
Pipestem triangle
Supports a crucible during heating with a Bunsen burner.

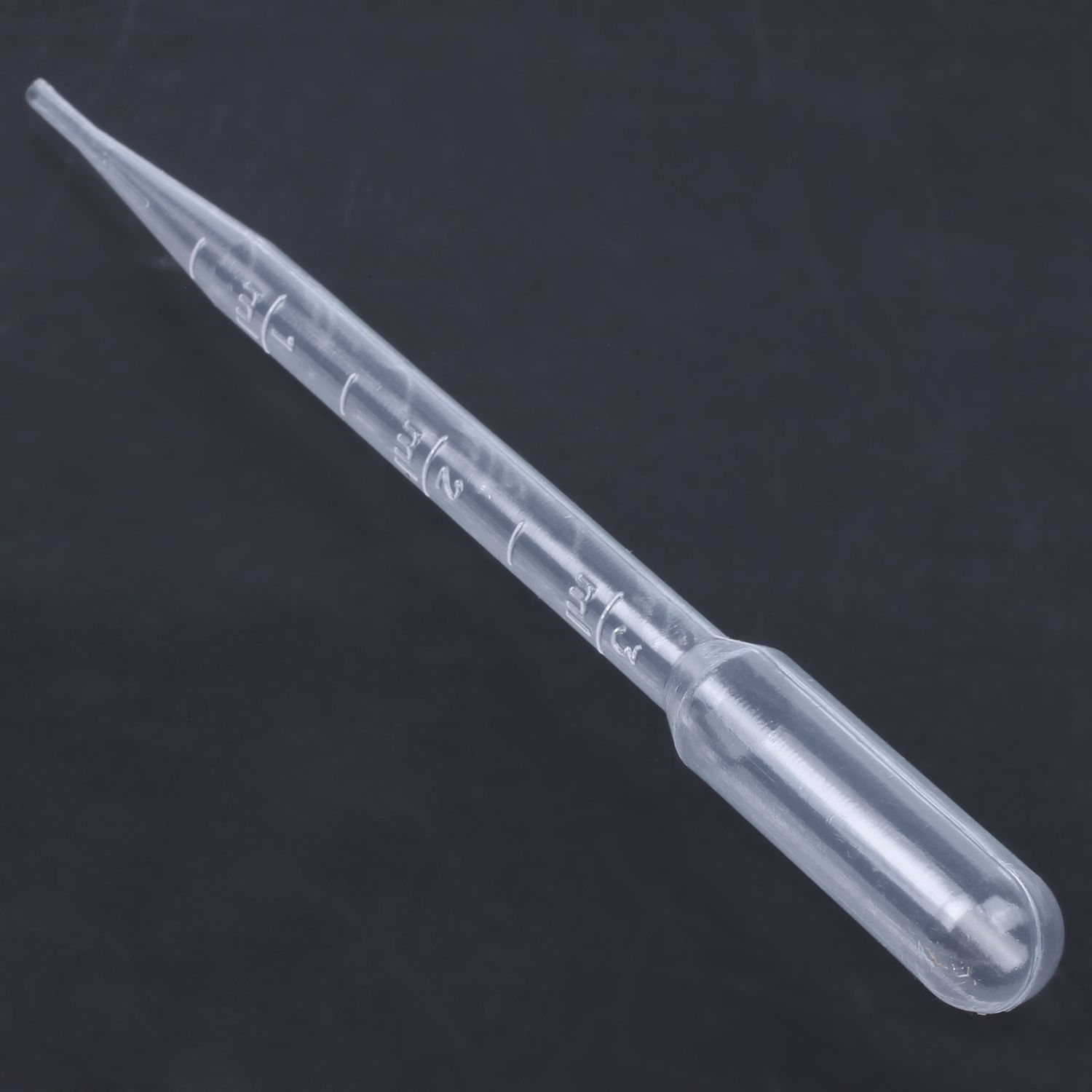
Pipette
Transfers small volumes of liquid precisely.
Prefix: Micro (μ)
10^-6
Prefix: Nano
10^-9
Pure Substance
A material with a constant composition- the same everywhere.
Reading Instruments
Read one significant digit further.
Ring clamp
Used to support glassware, such as funnels or flasks, by attaching to a stand during experiments.

Rubber stopper
Seals flasks or test tubes to prevent spills or contamination.

Scoopula
Scoops and transfers small amounts of solid chemicals.

Showing a zero is significant
Decimal point; adding a line; scientific notation.
Significant digits
Nonzero digits; last digit a 0 preceded by a decimal; all zeros between significants.
Solid
Has a fixed volume with relatively rigid structure; particles compact.
Stand
Holds rings, clamps, and other equipment in place.

Stirring rod
Used to mix chemicals or solutions in a beaker or flask.

Sublimation
The process where a solid changes directly into a gas.
Test tube
Holds small amounts of substances for reactions or heating.

Test tube brush
Cleans the inside of test tubes effectively.

Test tube clamp
Holds test tubes securely on a stand.

Test tube holder
Safely holds a few test tubes during heating or reactions.

Test tube rack
Organizes and holds many test tubes upright.

Thermometer
Measures the temperature of liquids or gases.

Tongs
Used to hold or move hot glassware or other lab equipment. You wouldn’t wanna use your hands, would you?

Triple point
The temperature at which a substance can exist in all three forms.
Volumetric flask
Used to prepare solutions of precise volumes.

Watch glass
Holds small amounts of substances or covers beakers during reactions.

Water bottle
Rinses glassware and adds small amounts of water to reactions.

Well plate
Holds small samples for reactions or mixing on a micro scale.

Wire gauze
Supports glassware and distributes heat evenly during heating.
