Type 2 DM
1/66
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
67 Terms
How is Type 2 Diabetes Mellitus characterized in terms of insulin dependence?
Patients are not absolutely dependent on insulin for life, though many may eventually require it.
What is the primary cause of hyperglycemia in Type 2 DM?
A relative (not absolute) deficiency of endogenous insulin, combined with insulin resistance.
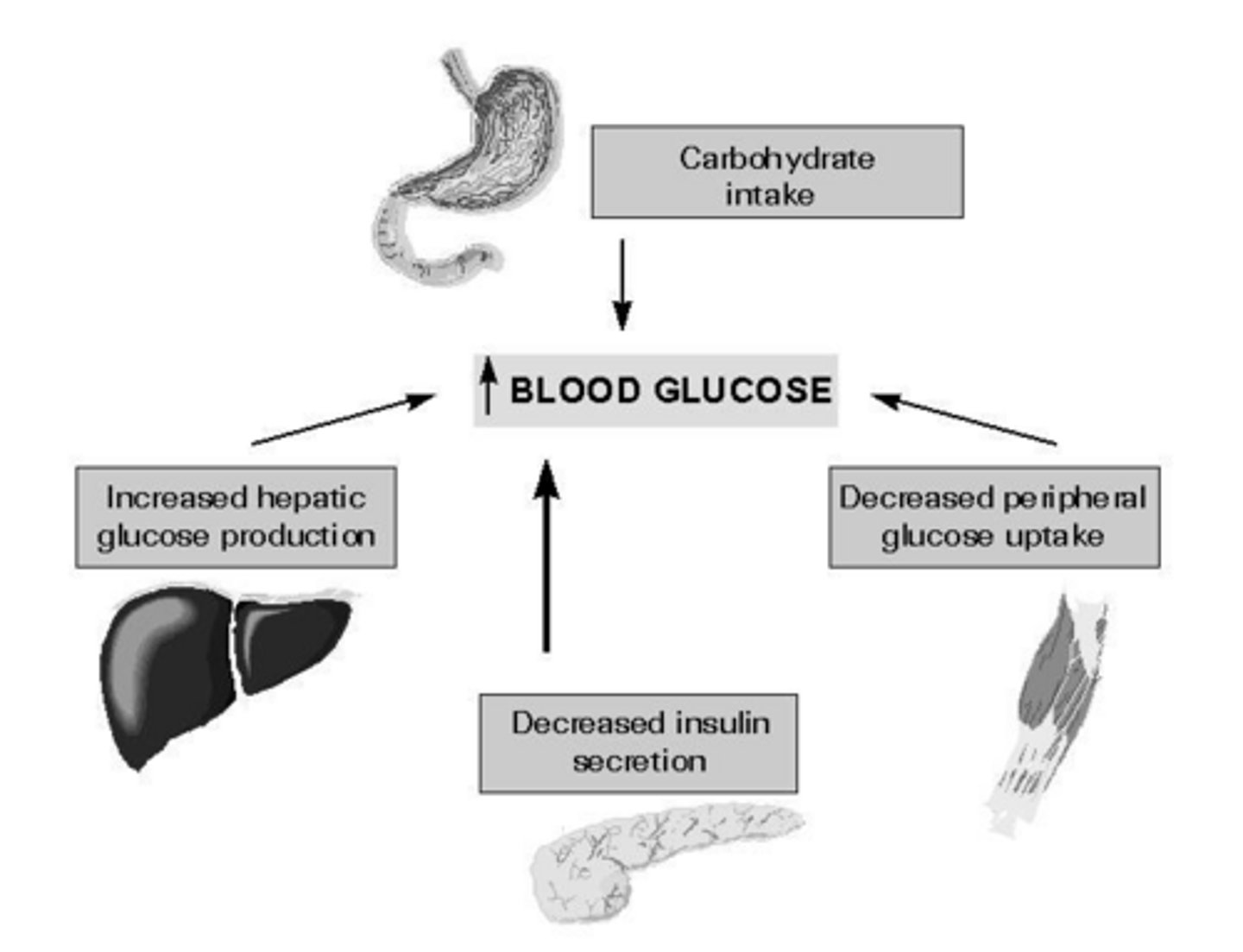
What does insulin resistance lead to?
Decreased glucose transport in muscle, elevated hepatic glucose production, and increased breakdown of fat.
What are the three primary sites of insulin resistance in Type 2 DM?
Muscle, fat, and the liver.
What role do free fatty acids play in the pathophysiology of Type 2 DM?
Elevated levels of free fatty acids contribute to insulin resistance.
What is the relationship between hyperglycemia and microvascular complications?
Hyperglycemia is the direct cause of microvascular complications.
When does macrovascular risk typically begin in the progression of Type 2 DM?
It begins with the onset of insulin resistance, often prior to the development of hyperglycemia.
What are symptoms that might suggest hyperglycemia?
Blurred vision, lower extremity paresthesias, or yeast infections.
What are the four diagnostic criteria for Type 2 Diabetes according to the ADA?
A1C ≥6.5%, FPG ≥126 mg/dl, 2-h plasma glucose ≥200 mg/dl during an OGTT, or a random plasma glucose ≥200 mg/dl in a patient with classic symptoms.
What are common symptoms that might suggest hyperglycemia in an undiagnosed patient?
Blurred vision, lower extremity paresthesias, or yeast infections.
How is microalbuminuria defined in terms of daily urine albumin excretion?
30-300 mg/day.
What is the recommended frequency for screening urine microalbumin in patients with diabetes?
Yearly.
Are antibodies to insulin, islet cells, or GAD typically present in Type 2 DM?
No, these are absent in Type 2 DM.
What are the primary goals of treatment for Type 2 DM?
Elimination of symptoms, reduction of microvascular risk (via glycemia/BP control), reduction of macrovascular risk (via lipid/BP control), and metabolic risk reduction.
How often should A1C be tested in patients who are meeting treatment goals and are stable?
At least twice annually.
How often should A1C be tested in patients whose therapy has changed or who are not meeting goals?
Quarterly.
What is the standard recommended A1C goal for many nonpregnant adults?
<7% (53 mmol/mol).
Under what circumstances might a clinician consider a less stringent A1C goal (e.g., <8%)?
In patients with a history of severe hypoglycemia, limited life expectancy, or other conditions that make <7% difficult to attain.
What did the UKPDS find with respect to microvascular complications of DM2?
Microvascular complications are reduced by 25% when median HbA1c is 7% compared with 7.9%
What is the relationship between HbA1c levels and microvascular complications according to the UKPDS?
There is a continuous relationship, with a 35% reduction in risk for every 1% decrement in HbA1c.
What is the impact of glycemic control on macrovascular disease risk in type 2 diabetes?
Glycemic control has minimal effect on macrovascular disease risk; such risk is more closely related to factors like dyslipidemia and hypertension.
What is the primary cause of death in approximately two-thirds of people with diabetes?
Heart disease or stroke.
Which patient populations should be considered for diabetes screening?
Obese patients, those with a first-degree relative with Type 2 DM, high-risk ethnic groups, women with a history of gestational diabetes or delivering a >9 lb infant, and patients with hypertension or high triglycerides.
Why are insulin levels often high early in the course of Type 2 DM?
They are high as a compensatory response to insulin resistance, though they are still inappropriately low for the level of glycemia.
What is the clinical significance of an abnormal urine microalbumin test (>30 mg/g)?
It should be followed by a quantitative timed urine specimen (overnight, 10-hour, or 24-hour) to confirm.
Which patients are strongly recommended to use Continuous Glucose Monitoring (CGM)?
Patients on insulin (especially basal-bolus regimens) and those at high risk for hypoglycemia.
Why is CGM recommended for patients with poor glycemic control despite therapy?
It helps identify glucose patterns such as post-meal spikes and overnight highs, and manages large glucose variability.
In what scenarios is CGM recommended for patients using basal insulin alone?
When the patient's A1C is not at goal and they have a risk for hypoglycemia.
What are the primary benefits of using CGM for non-insulin treated type 2 diabetes?
It provides lifestyle feedback, facilitates behavior modification, and allows for short-term optimization of therapy.
What are the main indications for using Automated Insulin Delivery (AID) systems in type 2 diabetes?
Insulin-dependent patients with poor control despite MDI, frequent hypoglycemia, or those already using insulin pumps.
What are the primary dietary goals for patients with type 2 diabetes?
Caloric restriction is of first importance, along with modest restriction of saturated fats and simple sugars.
What type of diet has been proven beneficial for type 2 diabetes management regarding macronutrient composition?
Low-carbohydrate diets with increased protein intake.
What are the benefits of aerobic exercise for patients with type 2 diabetes?
It improves insulin sensitivity and can markedly improve glycemia.

Which patients should undergo a cardiovascular evaluation before starting a significant exercise regimen?
Older patients, those with long-standing disease, patients with multiple risk factors, or those with evidence of atherosclerotic disease.
What is the relationship between exercise and arterial stiffness in type 2 diabetes?
Long-term endurance and strength training improve metabolic control and reduce arterial stiffness, leading to cardiovascular risk reduction.
When might a patient with type 2 diabetes require transient insulin therapy at presentation?
If they are symptomatic (e.g., polyuria, polydipsia) to reduce glucose toxicity.
What is the typical initial treatment approach for patients with an HbA1c less than 7.5%?
Usually treated initially with single agents.
What is the recommended initial therapy for patients with an HbA1c between 8% and 10%?
They may benefit from initial therapy with two agents or insulin.
What class of medication is Glyburide, and what is its primary mechanism?
It is a sulfonylurea, which acts as an insulin secretagogue.
Which sulfonylurea is noted for potentially causing more physiologic insulin release with less risk of hypoglycemia and weight gain?
Glipizide.
What unique safety profile does Glimepiride offer compared to other sulfonylureas?
It has a different interaction with cardiac potassium channels, suggesting greater safety in patients with ischemic heart disease.
What are the primary clinical benefits of Metformin in obese patients with type 2 diabetes?
It frequently results in weight loss and mild improvement in the lipid profile.
What are the contraindications for Metformin use?
Hepatic insufficiency or decompensated congestive heart failure requiring pharmacological therapy (due to risk of lactic acidosis).
Why is it recommended to titrate Metformin slowly and take it with meals?
To minimize gastrointestinal adverse effects.
What are the primary advantages of metformin therapy?
Efficacy, absence of weight gain or hypoglycemia, low side effect profile, high patient acceptance, and low cost.
What are the absolute contraindications for metformin use?
Severe decompensated congestive heart failure, renal impairment (GFR < 30), and advanced hepatic cirrhosis.
What are the primary mechanisms of action of GLP-1 receptor agonists (incretin-mimetics)?
Stimulates glucose-dependent insulin release, reduces glucagon, slows gastric emptying, and promotes satiety.
Which GLP-1 receptor agonist is associated with both weight loss and cardiovascular benefits?
Semaglutide (Ozempic, Wegovy).
What is a specific contraindication for the use of semaglutide?
Patients with a history of MEN2 or medullary thyroid cancer.
What is the unique mechanism of action of Tirzepatide?
It is a dual incretin agonist, targeting both GIP and GLP-1 receptors.
What are the GLP-1 effects of Tirzepatide?
Increased insulin, decreased glucagon and slows gastric emptying increasing satiety.
What are the GIP effects of Tirzepatide?
Enhances insulin secretion, weight loss, and insulin sensitivity.
What are the primary cardiovascular benefits of Liraglutide?
Proven reduction in the risk of myocardial infarction, stroke, and cardiovascular death.
What is a critical clinical precaution when using DPP-4 inhibitors?
They should not be combined with GLP-1 receptor agonists.
How do SGLT-2 inhibitors lower blood glucose levels?
They increase urinary glucose excretion by lowering the renal glucose threshold, acting independently of insulin.
What are the major cardiovascular and renal benefits of SGLT-2 inhibitors?
Reduction in heart failure hospitalizations, reduced cardiovascular mortality, slowing of CKD progression, and reduction of albuminuria.
What are common side effects or cautions associated with SGLT-2 inhibitors?
Recurrent genital infections, risk of dehydration, and caution in advanced renal failure.
What is the primary role of Pramlintide (an amylin analogue)?
It delays gastric emptying, decreases postprandial glucagon release, and modulates appetite to promote satiety.
Why is insulin therapy often necessary for patients with type 2 diabetes?
Many patients eventually become markedly insulinopenic, and insulin is the only therapy that corrects this specific defect.
What is the target blood pressure goal for most patients with diabetes?
Less than 130/80 mm Hg.
Which classes of antihypertensive medications are preferred for diabetic patients and why?
ACE inhibitors and ARBs, due to their proven renal protection effects.
What is the most common complication observed in patients with type 2 diabetes?
Peripheral neuropathy.
What is the typical clinical presentation of diabetic peripheral neuropathy?
Paresthesias, numbness, or pain, typically following a 'stocking and glove' pattern, affecting the feet more often than the hands.
What is the leading cause of end-stage renal disease (ESRD) in the United States?
Diabetes mellitus, particularly type 2.
What are the primary strategies for managing claudication in patients with peripheral vascular disease?
Smoking cessation, correction of lipid abnormalities, and antiplatelet therapy.
How often should a lipid profile be screened in adults with diabetes not taking statins?
At diagnosis, at the initial medical evaluation, and every 5 years thereafter (or more frequently if indicated).
Why is a multidisciplinary team approach essential for diabetes education?
Diabetes management is a lifetime exercise that requires more than brief instructions; a team including nutritionists and educators provides more comprehensive and sustainable care.