Orgo 1 - Addition Reactions
1/37
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
38 Terms
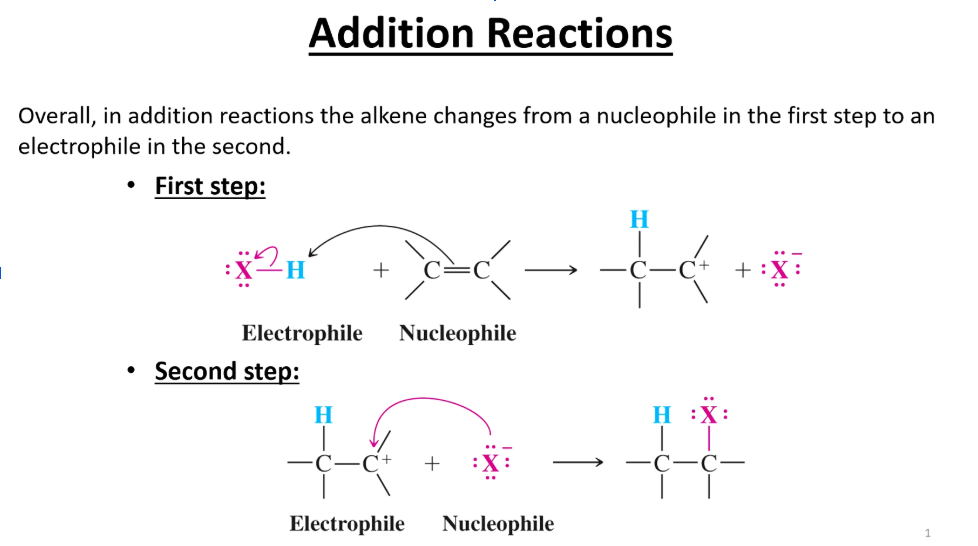
What is the general mechanism for an addition reaction?
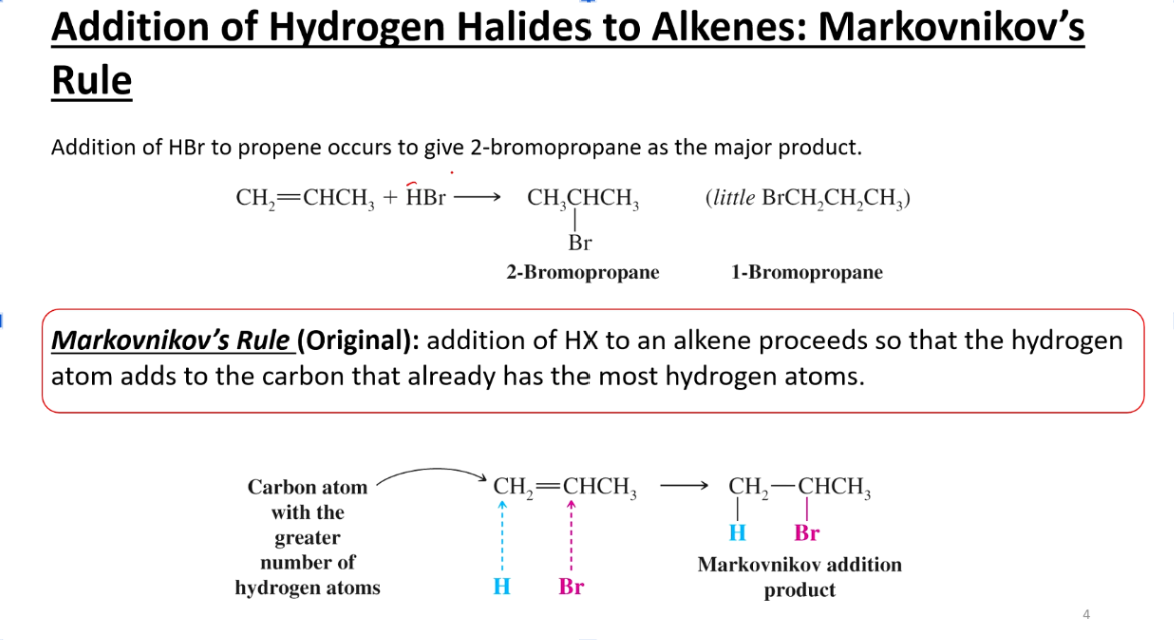
What is Markonikov’s Rule
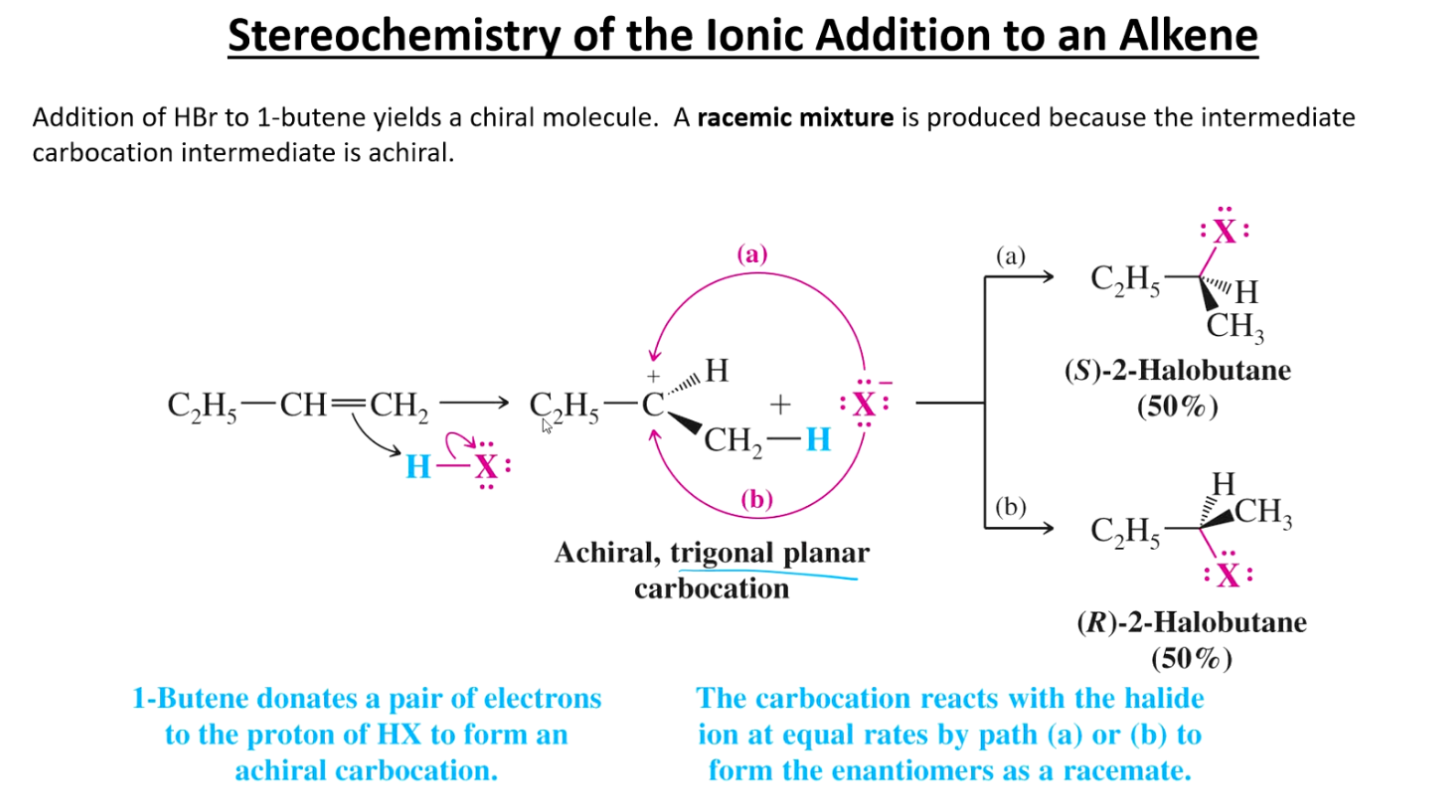
What is the chirality of a standard addition reaction on an alkene?
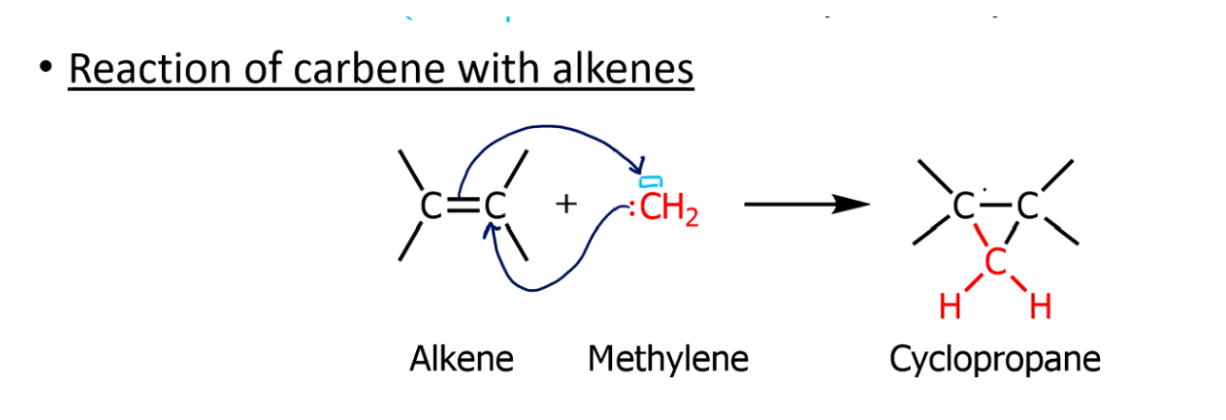
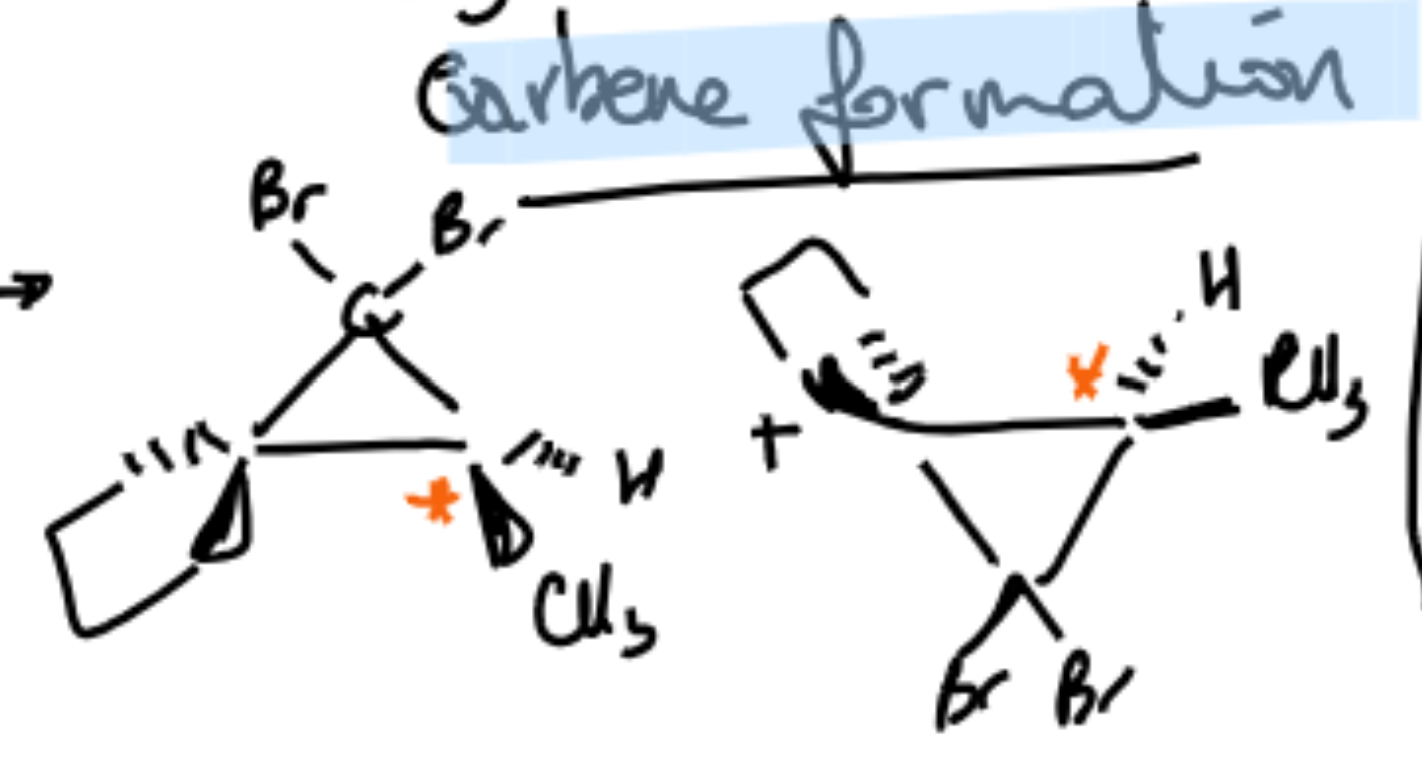
Carbene reaction mechanism, and when they happen
1. Dihalocarbenes from haloforms
Reagents like CHCl₃ + strong base (e.g., OH⁻) generate :CCl₂
This carbene adds to an alkene → dichlorocyclopropane
👉 Key signal: CHCl₃ / base or similar haloform conditions
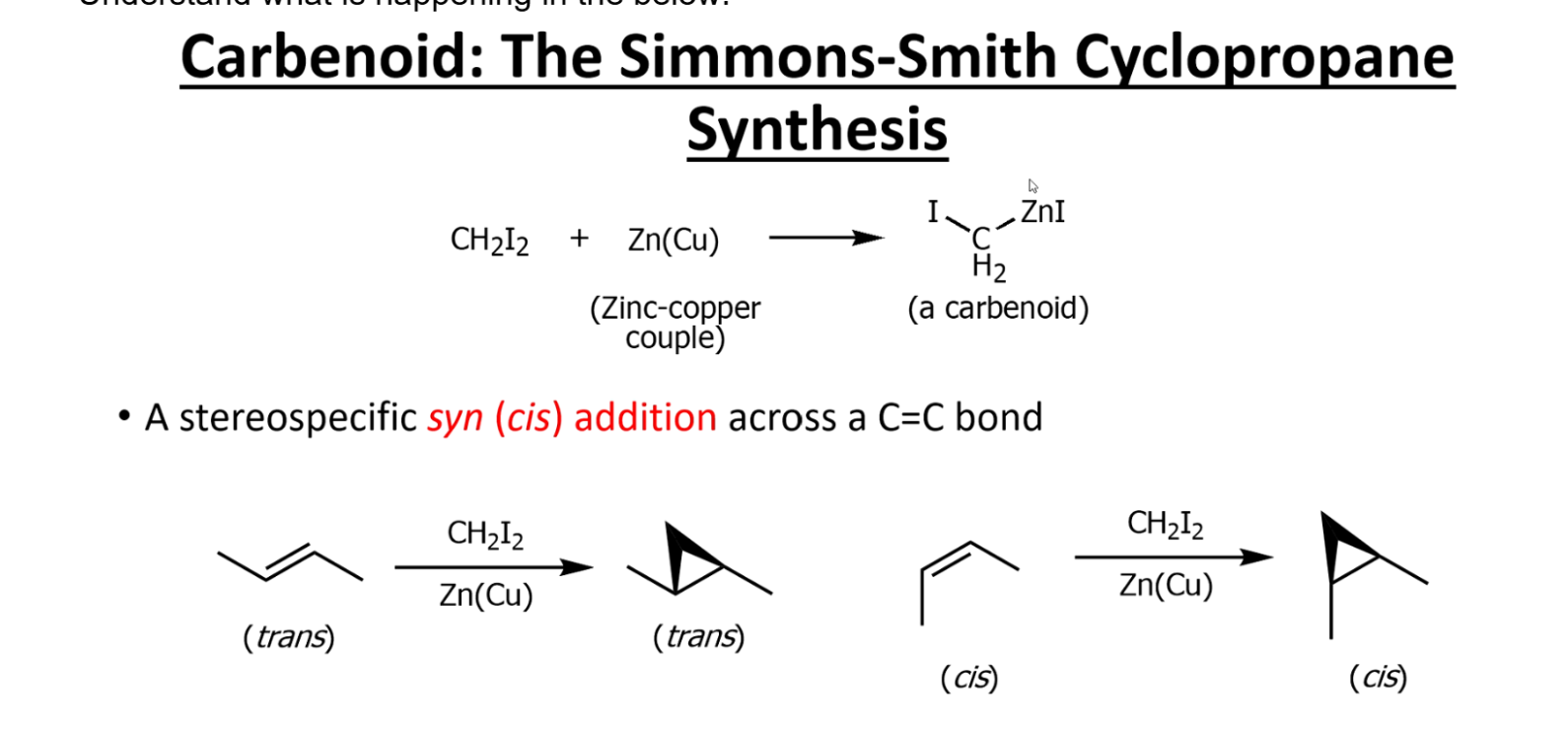
2. Simmons–Smith reaction (carbenoid, not free carbene)
Reagents: CH₂I₂ + Zn(Cu)
Forms a “carbenoid” that behaves like :CH₂
Adds to alkenes → cyclopropane
👉 Key signal: CH₂I₂ + Zn(Cu)
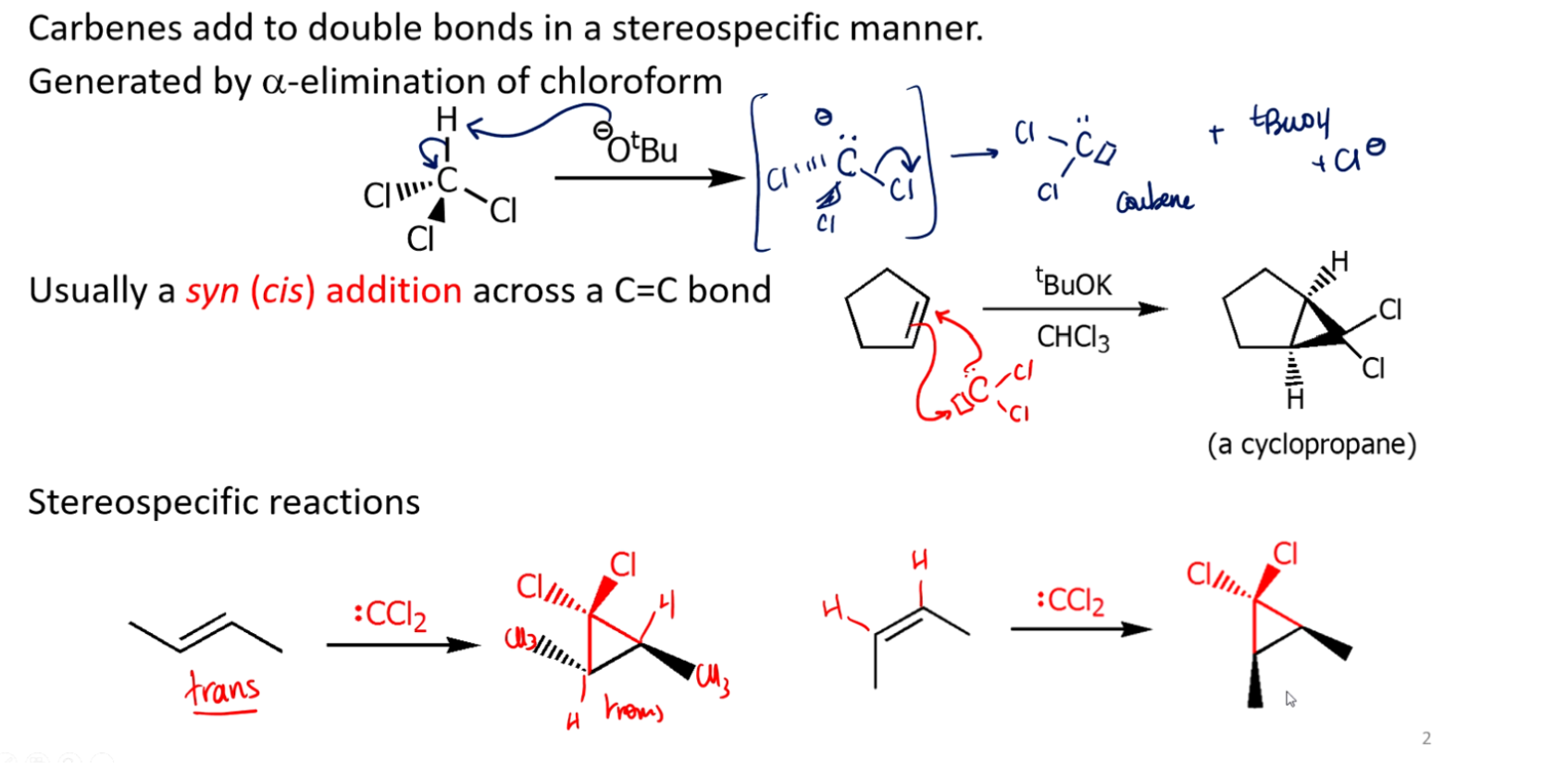
One common mechanism of carbene formation
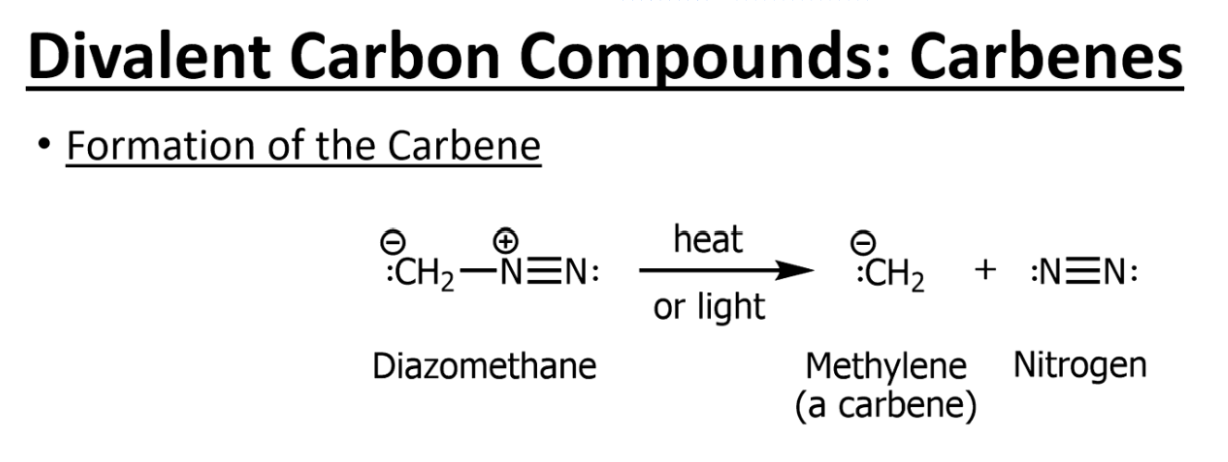
Mechanism of carbene reaction
Carbenoid (Smith-Simmons)
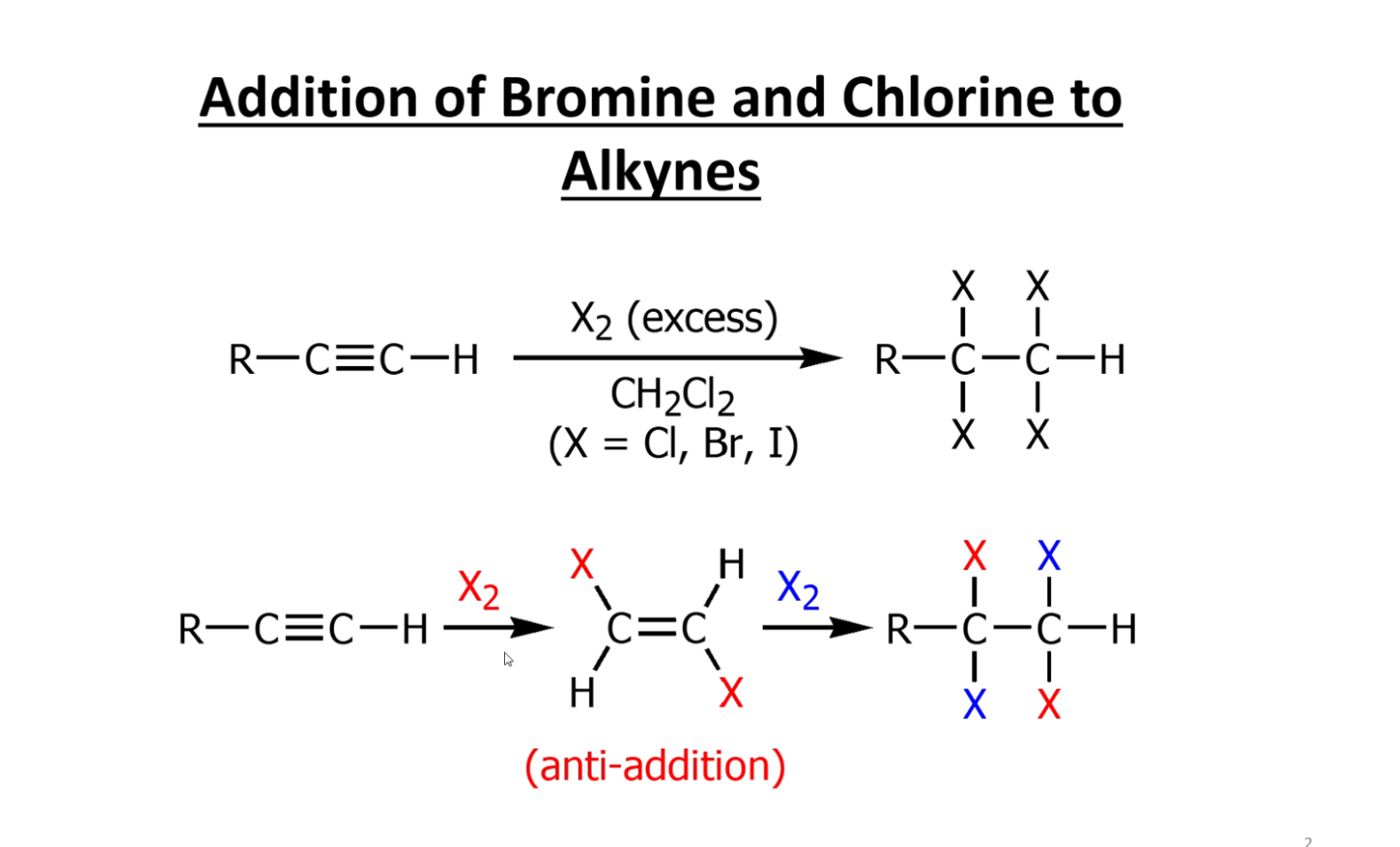
Regular halogenation in alkynes
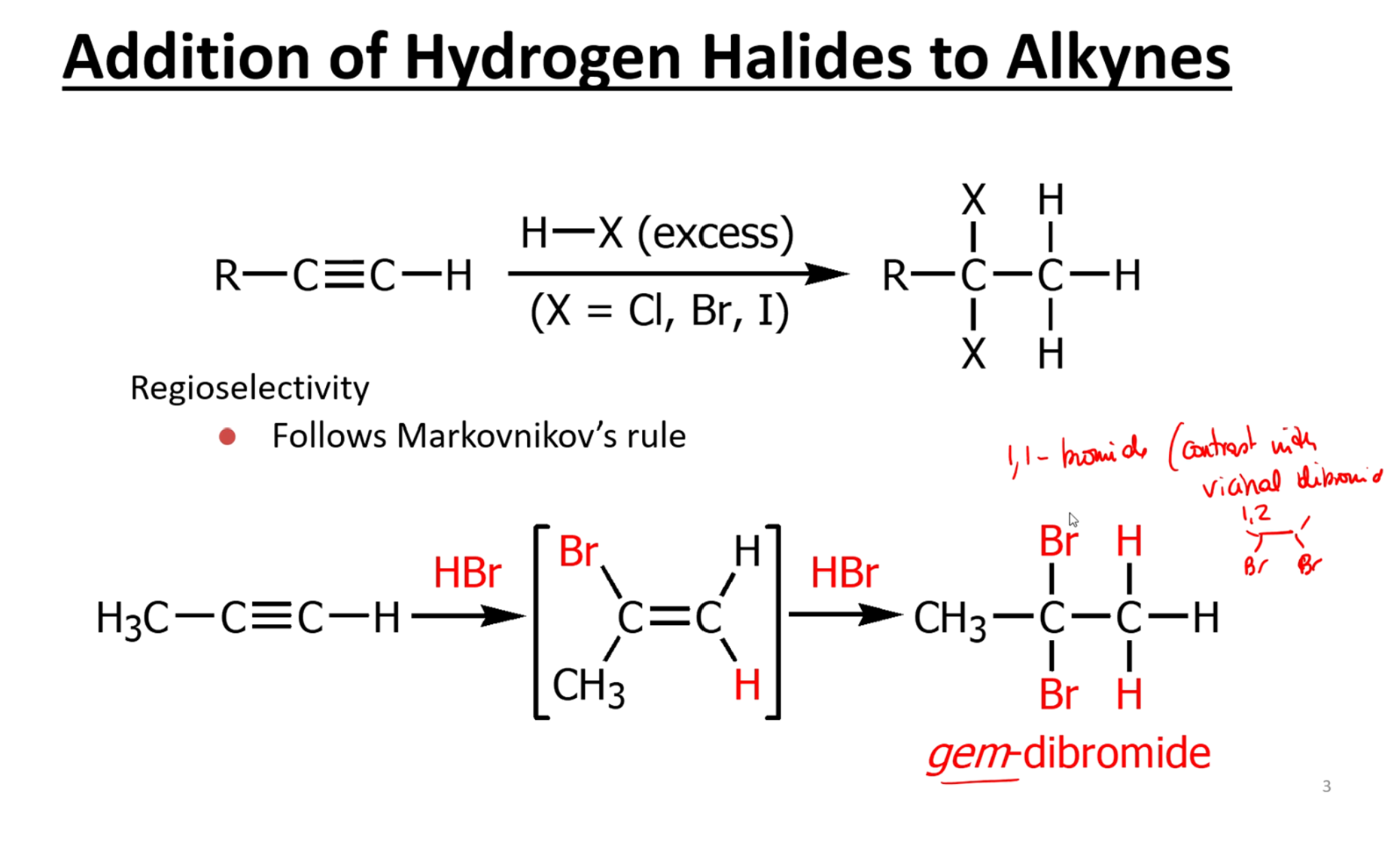
Hydrogen halide addition
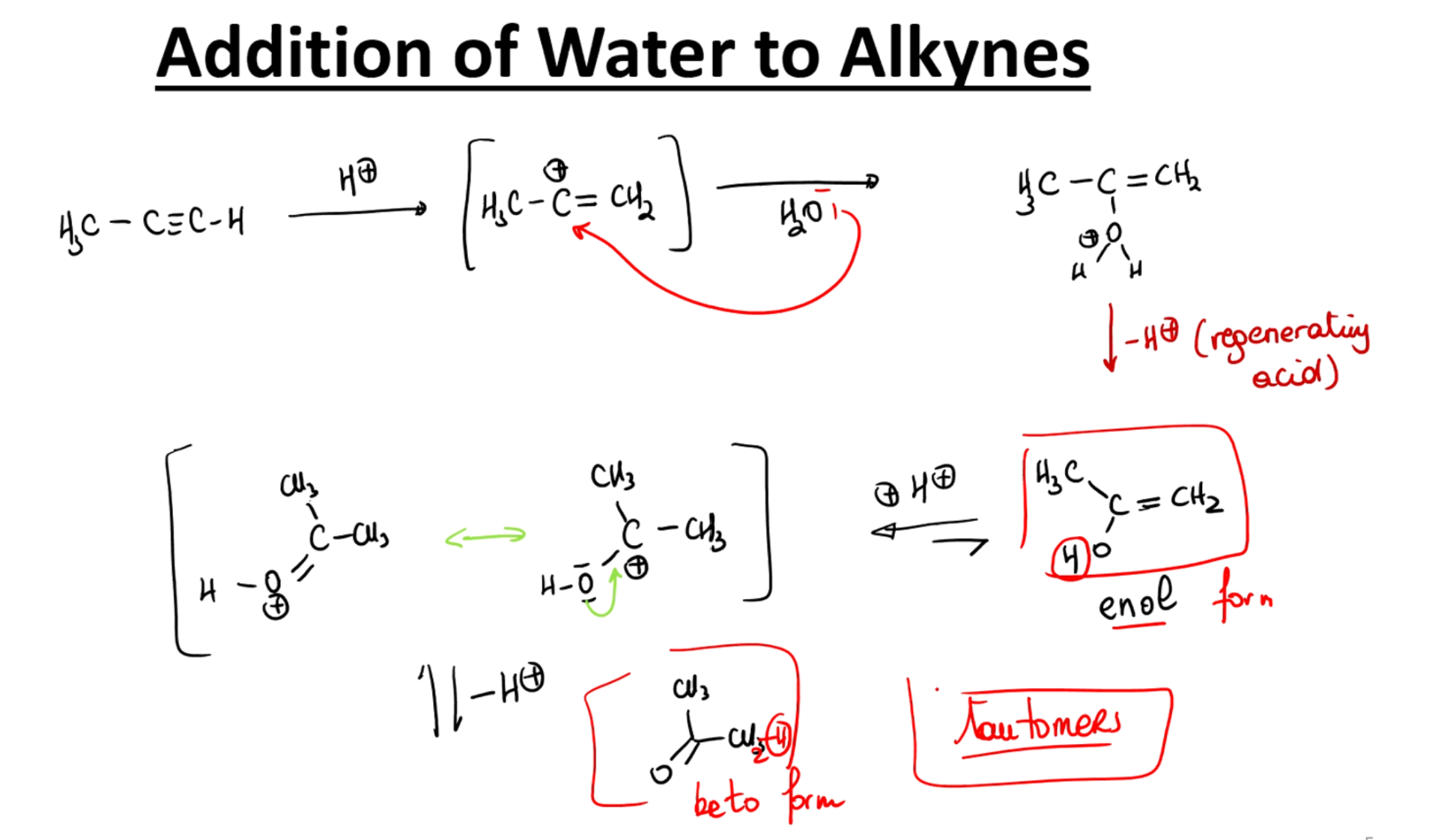
Addition of water to alkynes
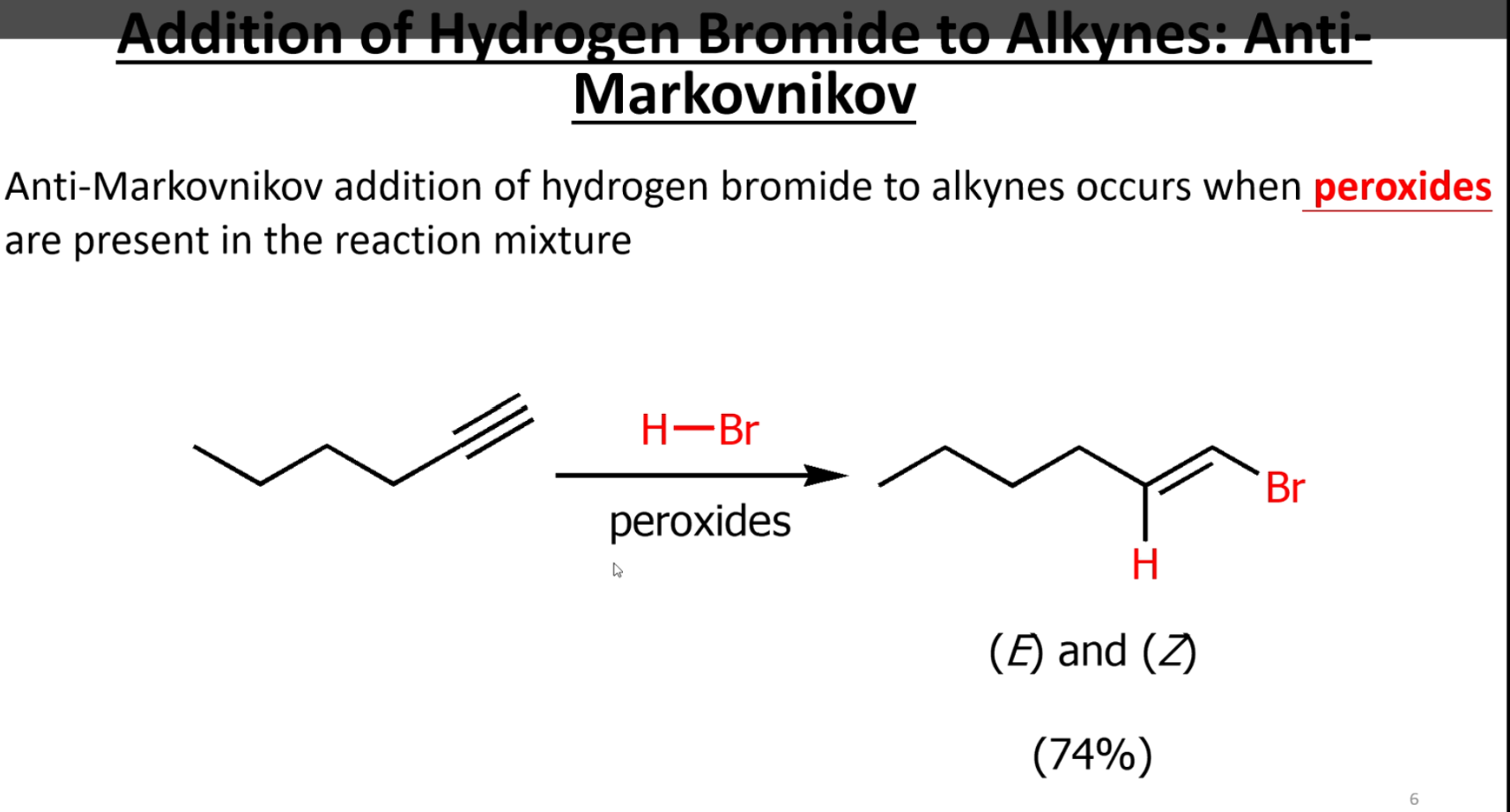
Anti Markovnikov with bromine
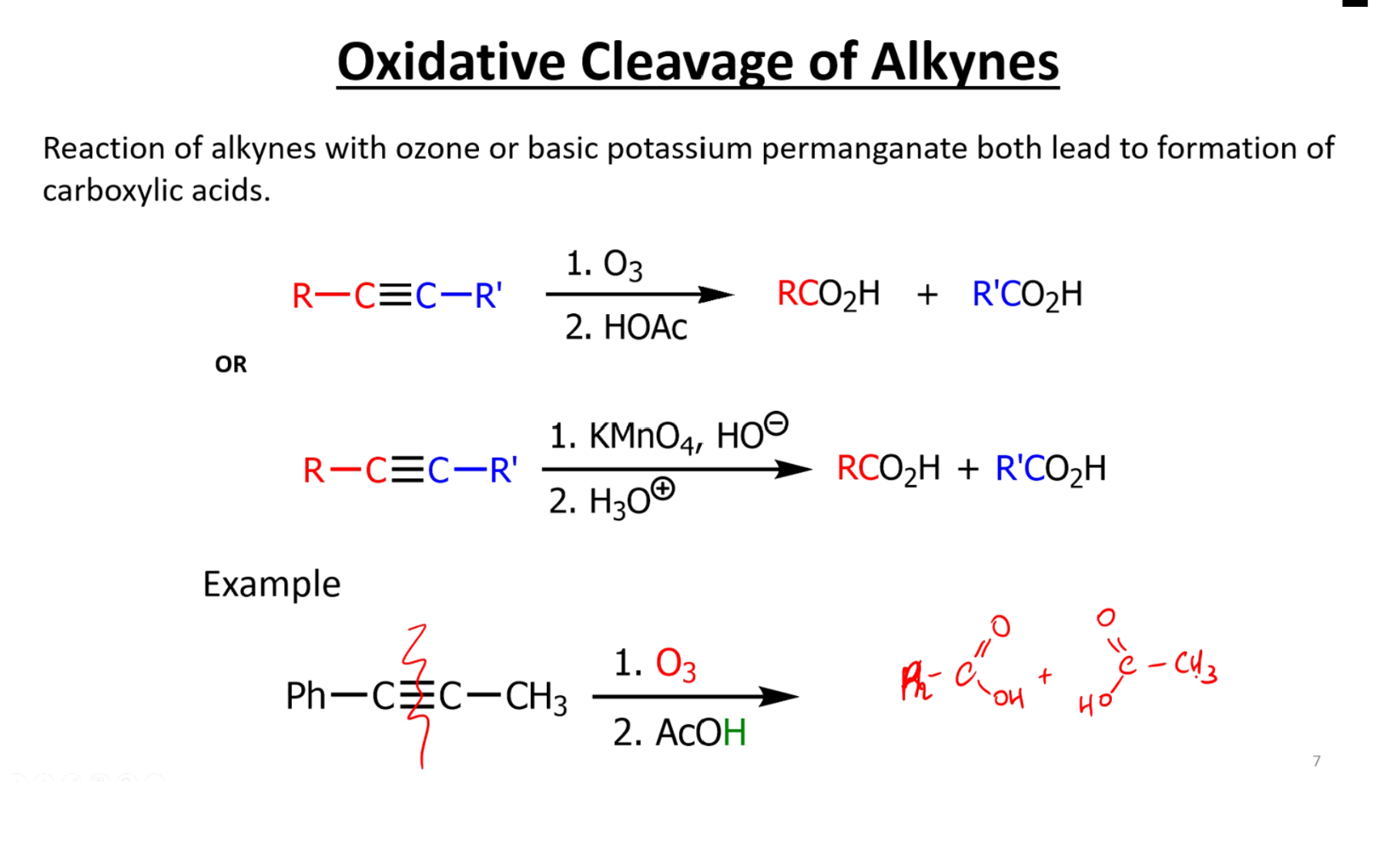
Ozone and potassium permanganate
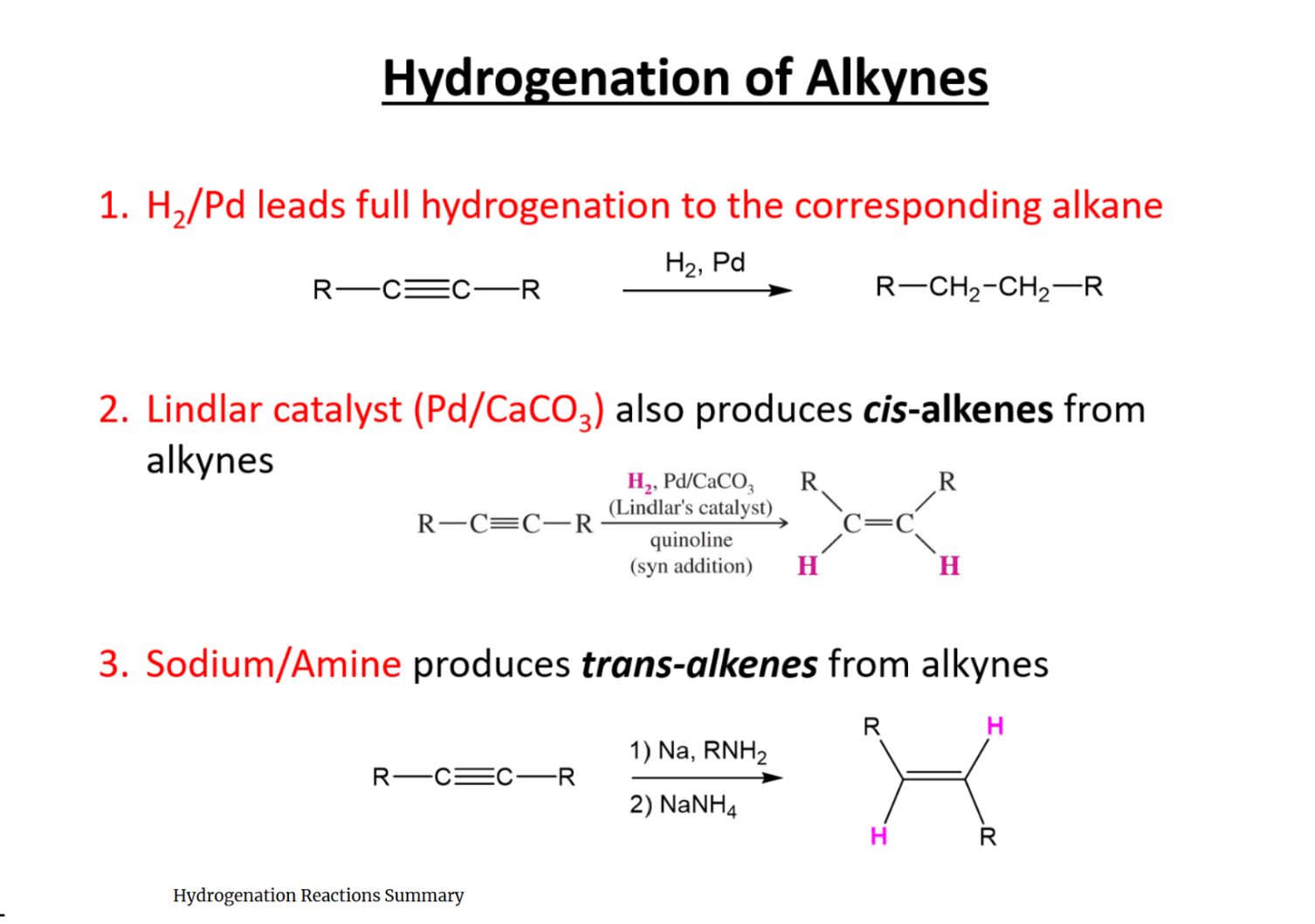
Hydrogenation

Three possibilities for hydrogenation
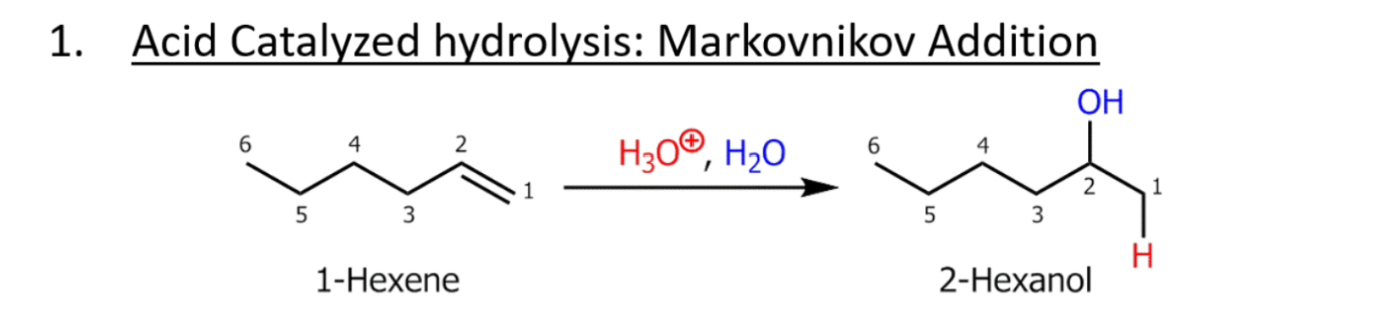
Acid catalyzed hydrolosis
Oxymercuration regiochemistry
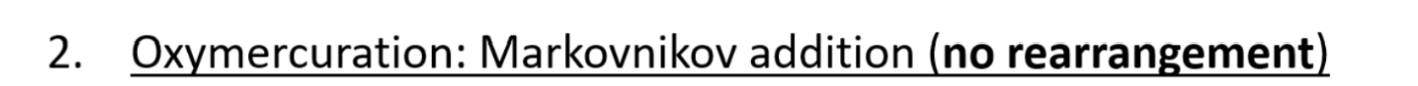
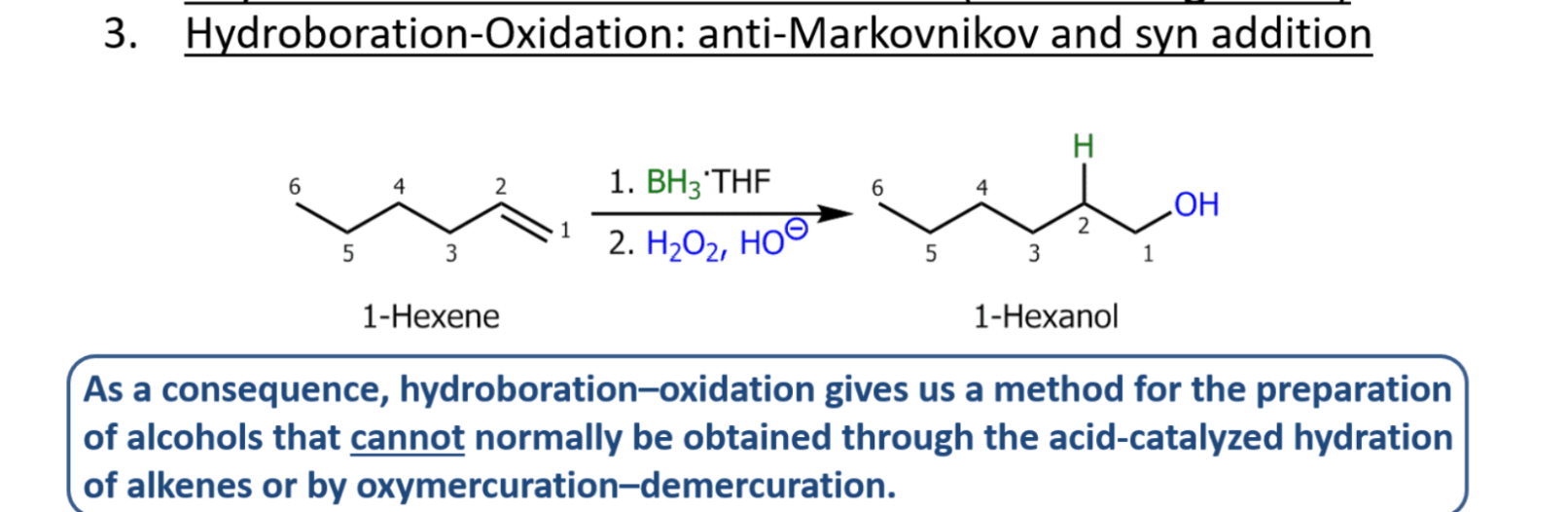
Boron hydration
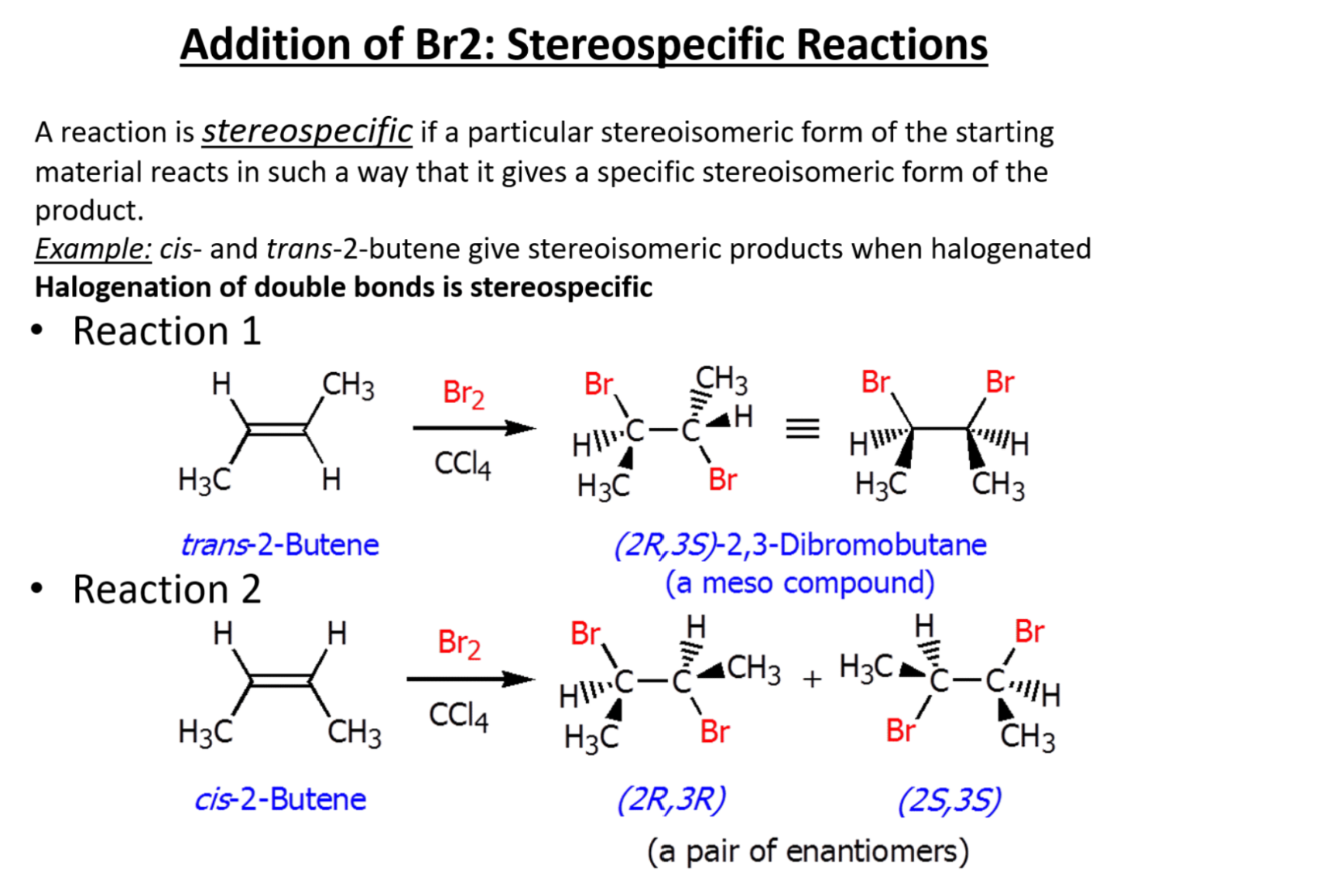
Stereospecific addition of Br2
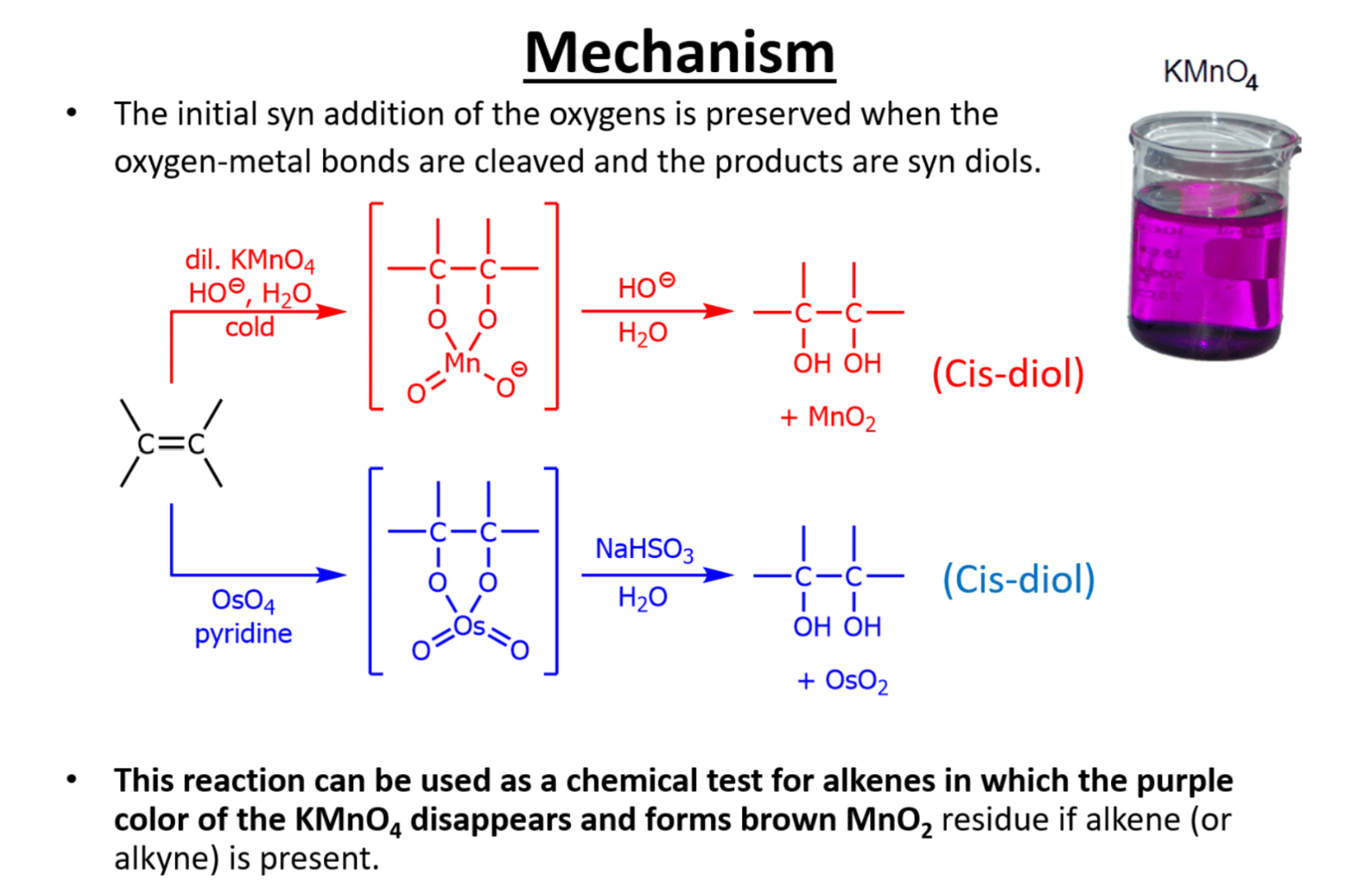
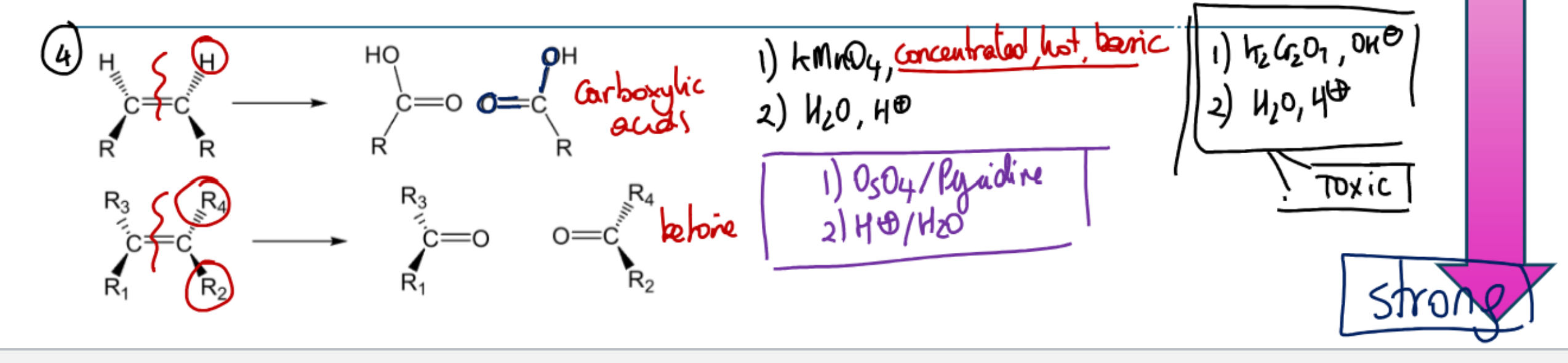
OsO4 and dilute KMnO4
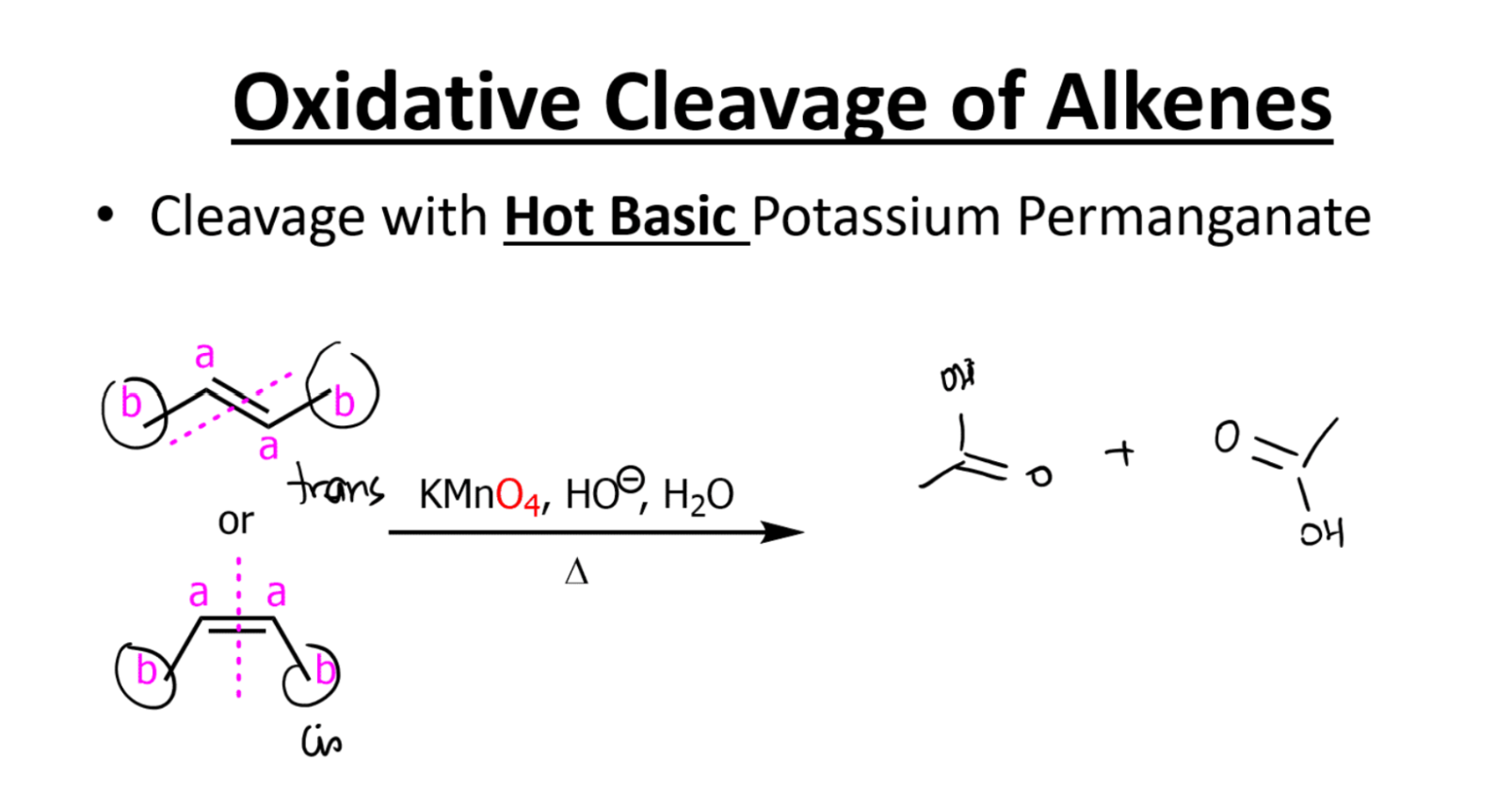
Hot and basic potassium permanganate
**Same as basic conditions, just carboxylic salt if no acid workup
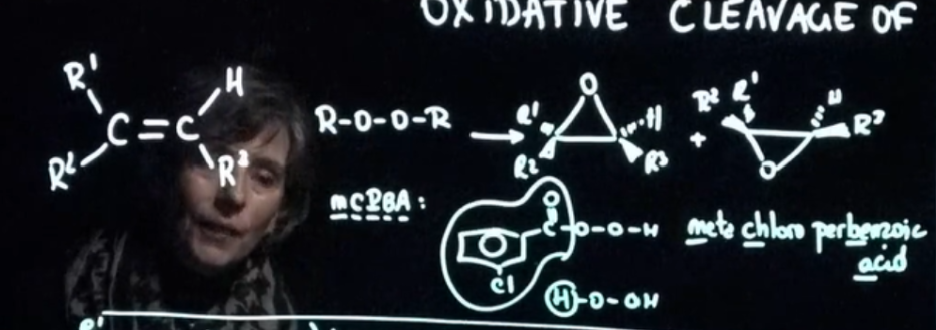
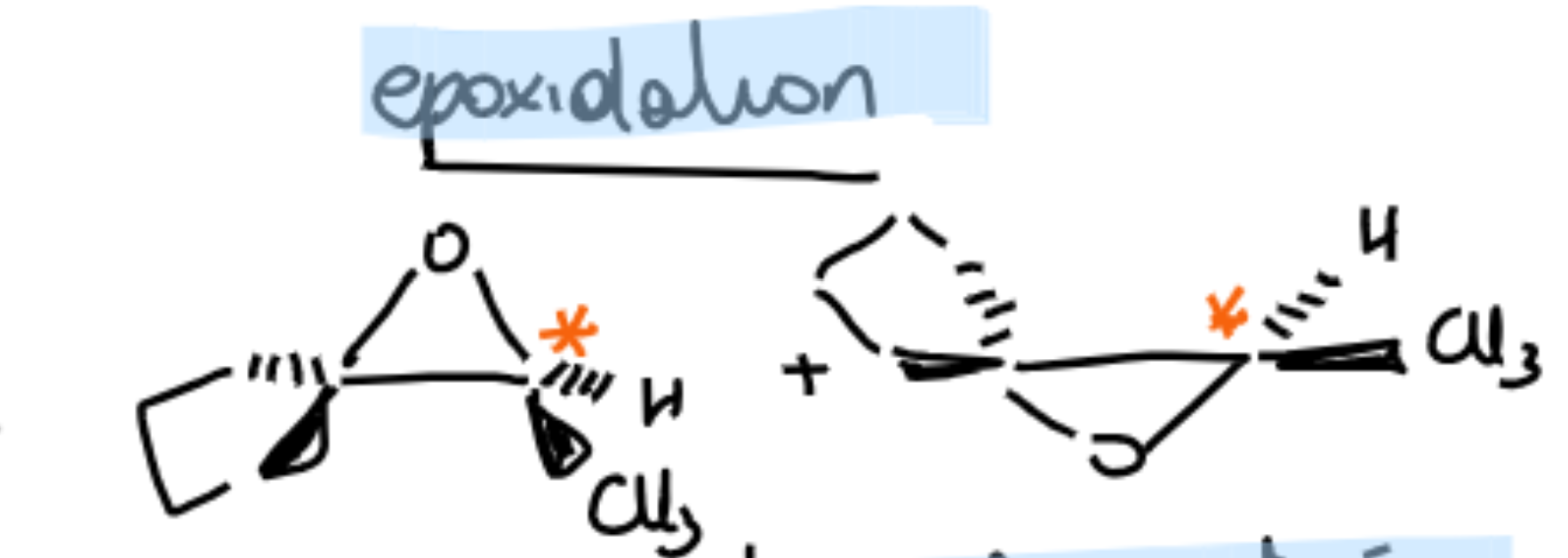
mCBA and epoxide product
Permanagante cold dilute
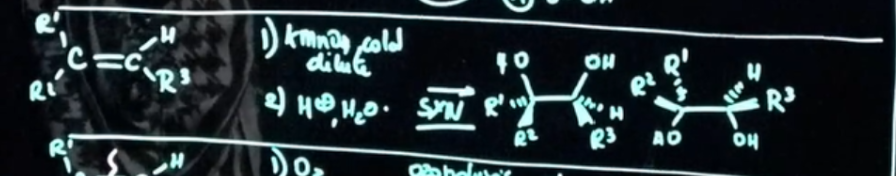
What is the order of strength for oxidative cleavage reactions

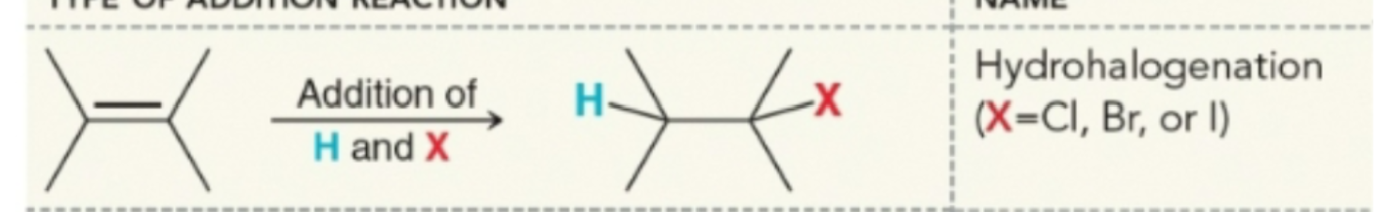
Addition of HX product
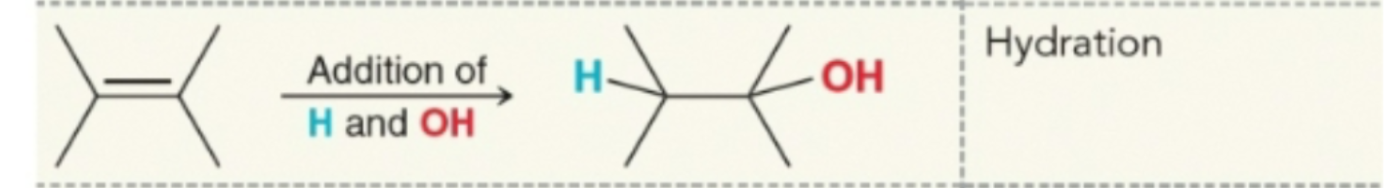
Hydration with water and like base
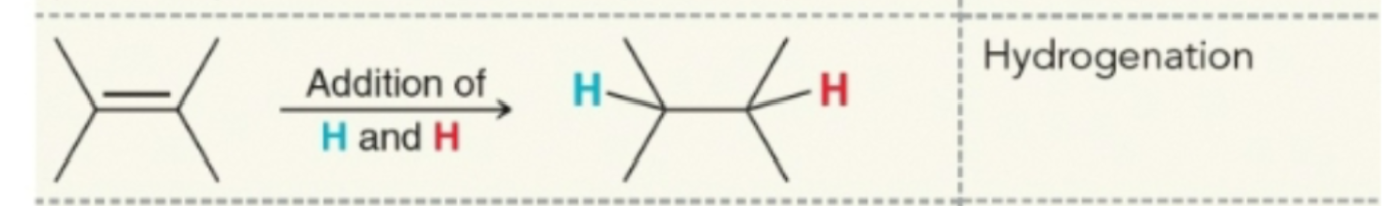
Hydrogenation products
Halogenation products
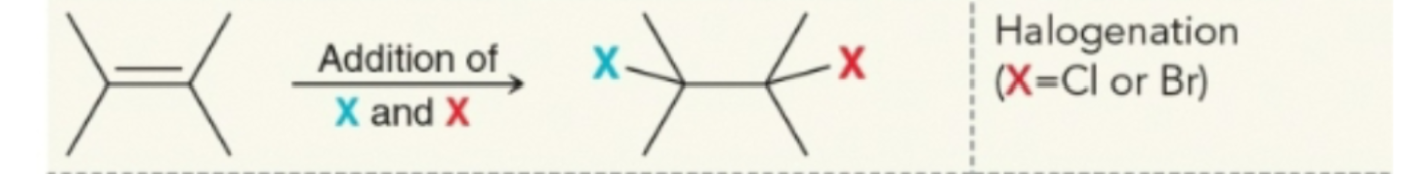
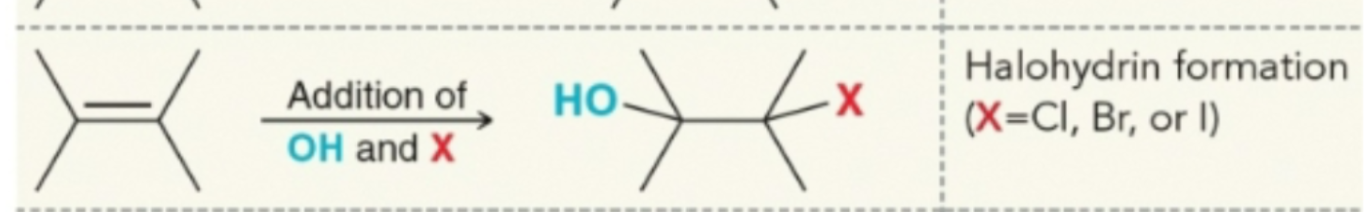
Addition of water and halogen
Water takes up the markonikov addition, halogen the anti
Degree of unsaturation formula

Different outcomes of hydration, summarized

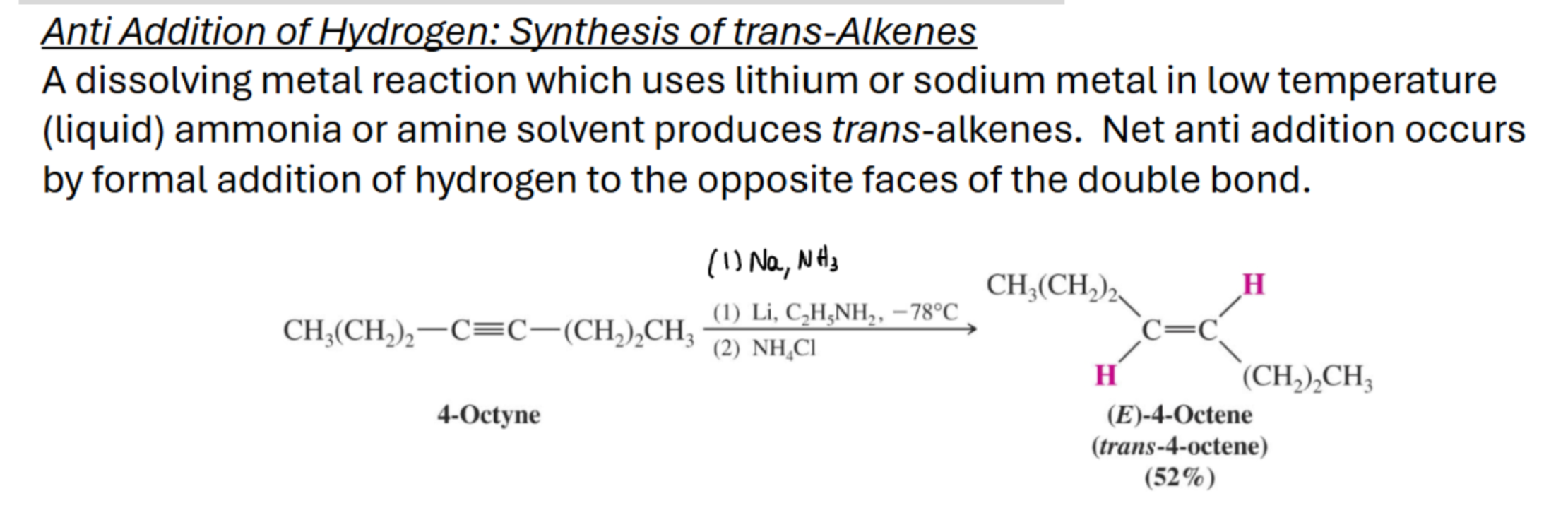
Synthesis of trans alkenes for alkynes
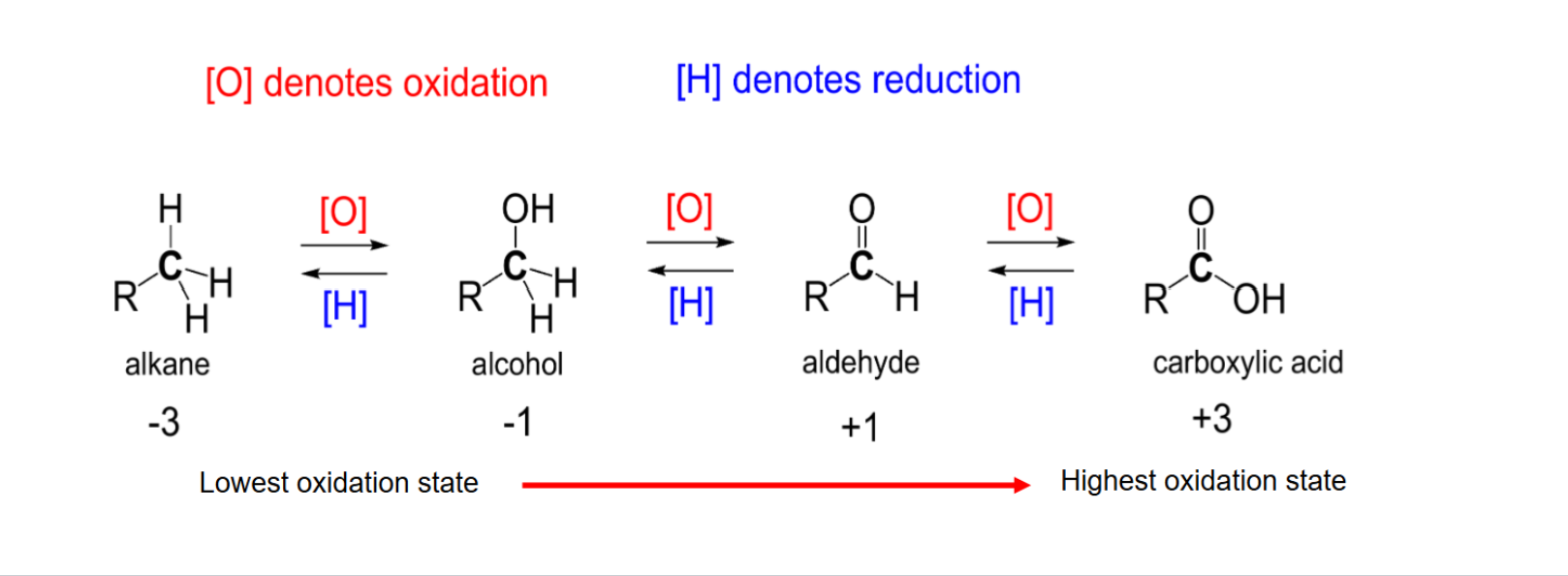
Order of oxidation states (may help you remember products for oxidative cleavage)
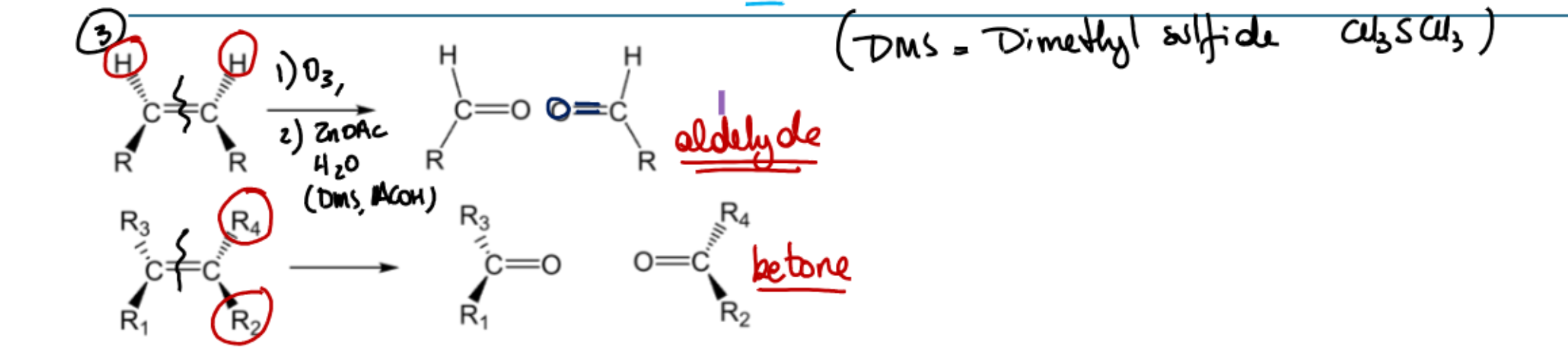
Common solvent for the ozone one? What determines if the products are aldhydes or ketones?
2 H on alkene → aldehydes, all R groups → ketones
What determines what the products will be for the strongest one? What decides carboxylic acid vs ketone?
Where to put wedges for chiralty of epoxidation?
Properly representing carbene reaction chirality on products?
What happens when you add water and a dihalogen to something, but there is only one possible site for markonikov addition?
