Biochemistry Exam 4
1/96
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
97 Terms
Benefits of enzymes
Higher reaction rates
Mild conditions
Greater specificity
Enzymes rarely produce side products
true
Enzymes are composed of
proteins
Metal atom bonded to a protein
cofactor
Holoenzyme
protein + cofactor
Apoenzyme
protein without REQUIRED cofactor
Active site
Where substrate binds
The active site creates a unique
microenviornment
Reaction rate
collision frequency X energy factor X probability factor(oreintation)
how do enzymes enhance the probability factor
by oreinting the substrate in an optimal postion
How do enzymes lower the energy factor
Stabilizes the transition state of substrate
replaced interaction with water in solvetaed state
directs the path leading to only one product formation
How do enzymes increase the collsion factor
Reactants bound at an enzyme are at infintite conentration
Change of G = ____ at equilibrium
0
Negative change of G reflects
a spontaneous reaction
Change of G relates to
Rate of Reaction
An enzyme cannot
alter the postion of the equilibrium
A enzyme only allows the reaction to
Reach equilibrium faster
An anzyme increases the rates of the
forward and reverse reactions equally
Binding energy
facilitates the binding of E+S into the ES complex
2 parts of active site
specificity
Catalytic
What will happen if an enzyme has too much specificity
the ES complex will become too stable and not go into the transition state
To keep the enzyme from getting stuck at the ES phase, enzymes must be complementary to
Transtion state NOT the substrate
2 ways active site can bind
Lock and Key —>affinity for substrate without conformation change
Induced fit —> Site increases affinity for the substate with a conformational change upon binding
Maximum velocity is reached when
all the active binding sites are filled with substrate
Reaction rate is limited by two things
How many active sites are avalible
How fast the enzyme works
In a higher the concentration the rate __________
increases (until all the active sites are taken up and the speed flatlines)
V
number of moles of product formed per second
Michaelies-Menten is a plot of
V vs [S]
Michealies-Menten shows a _____ relationship at low [S] concentrations
linear
At high [S] in they enhance the probaility factor we close in on _____ ebing the only thing in solution
ES complex
Velocity of a reaction =
rate constant of step X [reactants]
Steady state
the concentration of intermediates remains constant over time because their rate of production equals their rate of consumption.
[E] =
[total enzyme] - [ES] (complexing enzyme)
Michealis-menten equation if [S] is large
V0 = Vmax (([S]) / ([S}+Km))
Km
(the Michaelis constant) is defined as the substrate concentration required to reach half the maximum velocity ()
Lineweaver-Burk Plot

y intercept of Lineweaver-burk gives
1/Vmax
x intercept of lineweaver Burk plot
-1/Km
Slope of linearweaver Burk Plot
Km/Vmax
y of lineweaver burk
1/V0
x of lineweaver burk
1/[S]
Eadie-Hofstee plots
V0 vs V0/[S]
Eadie-Hofstee Y intercept
Vmax
Eadie-Hofstee slope
-Km
Km = [S] when
hald of the active sites on the enzyme are filled
Small Km
high affinity of enzyme for subtrate (k1 is high)(fomration of ES complex is high)
large Km
low affinity of enzyme for subtrate (K2 is high)( decompostion of ES complex is high)
turnover number
number of substrate molecules converted into products by an enzyme per unit of time when FULLY saturarted with substrate
Kcat (turnover number) is equal to
K2
Vmax
represents the maximum rate (velocity) an enzyme-catalyzed reaction reaches when the enzyme is fully saturated with substrate
[Et]
number of active sites
Vmax = ________
k2 [Et]
Limit to Vmax
a reaction cannot proceed faster then the substrate can find the enzyme (rate of diffusion)
Two types of enzyme inhibition
Irreversible inhibition —> stop enzyme and it dies forever
reversible inhibition (4 types) —> remove the drug and remove the inhibition
Irreversible Inhibitor (I) will bind _______ to enzyme at active sight
TIGHTLY —> covalently links
Most Irreversible Inhibitors (I) are often
alkylating agents that react with active site of AA residues
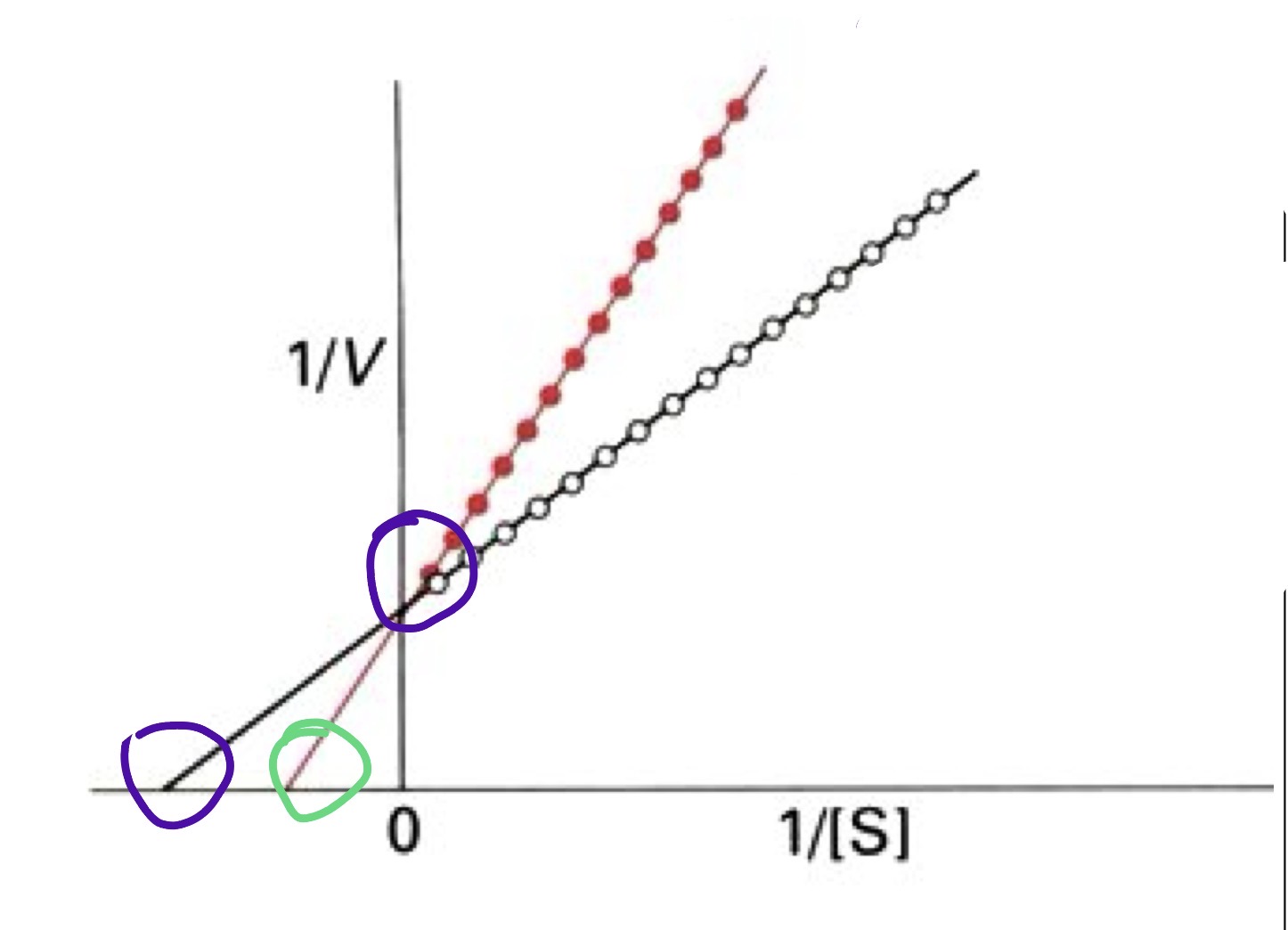
Competivtive Inhibition
I binds to the same active site as the substrate.
Competivtive Inhibition can be overcome by
high [S]
How does Vmax and Km change with competitive inhibtion
Km increases → the enzyme appears to have lower affinity for the substrate. It takes a higher substrate concentration to reach half of Vmax.
Vmax unchanged → if you add enough substrate, the substrate can outcompete the inhibitor and the enzyme can still reach the same maximum rate.
α represents
degree of Inhibition
A larger α means
more inhibition
α =
slope + inhibitor / slope uninhibited enzyme
Ki
Inhibitor dissocitation constant —> How well the inhibior biunds to the enzyme
Low Ki
inhibitor binds tightly to enzyme —> less concentration is needed to achieve 50% inhibition
high Ki
inhibitor binds loosley to enzyme
Ki =
[I] / (α-1)
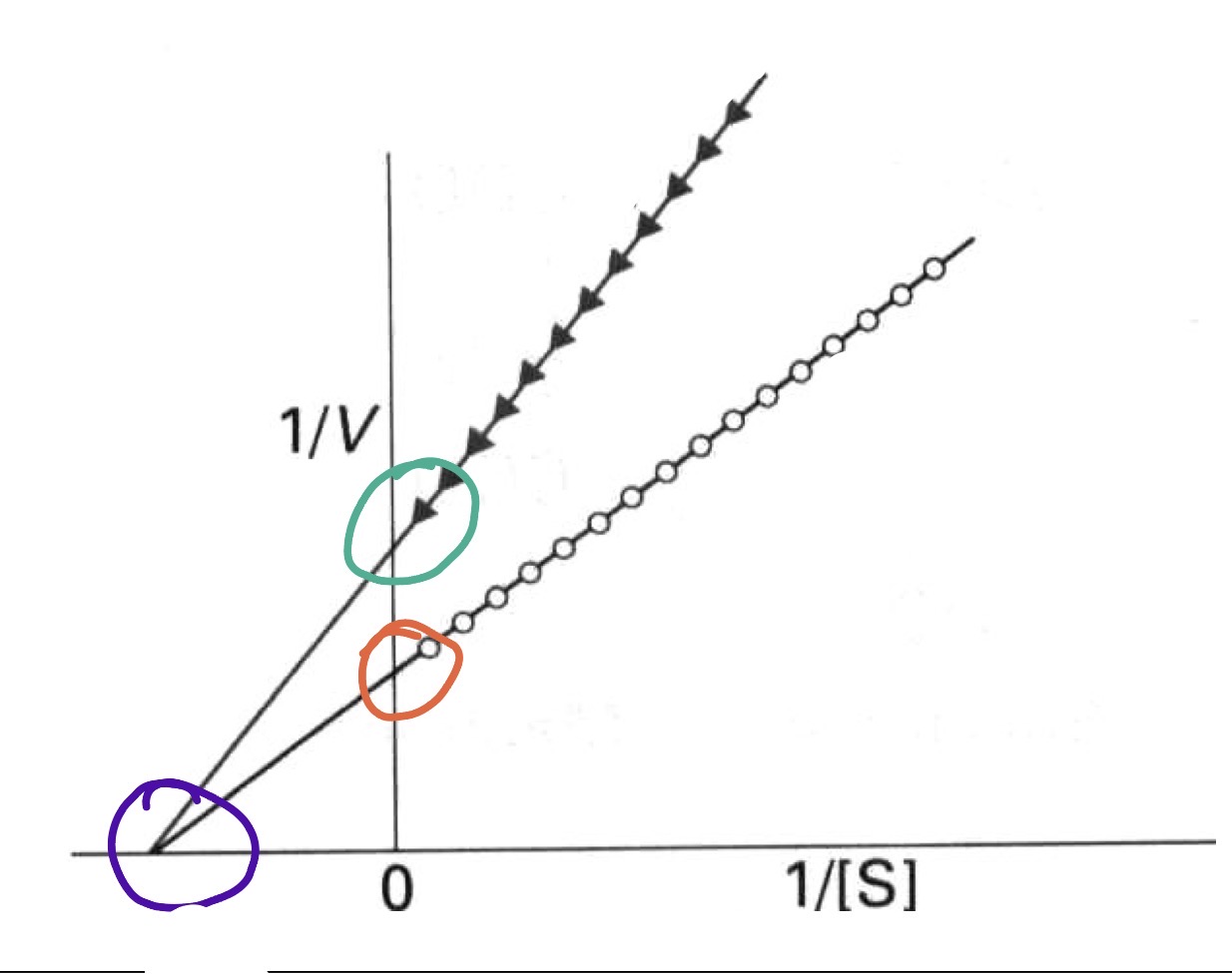
Noncompetitive Inhibition
S and I bind at the same time
I will inhibit by decreasing the turnover number
Cannot be overcome by an increase in [S]
Noncompetitive Inhibition effects on Vmax and Km
Vmax decreases —> the inhibitor decreases full activity
Km is not altered —> The inhibitor does not change substrate binding affinity, it takes the same [S] to reach 1/2 new vmax
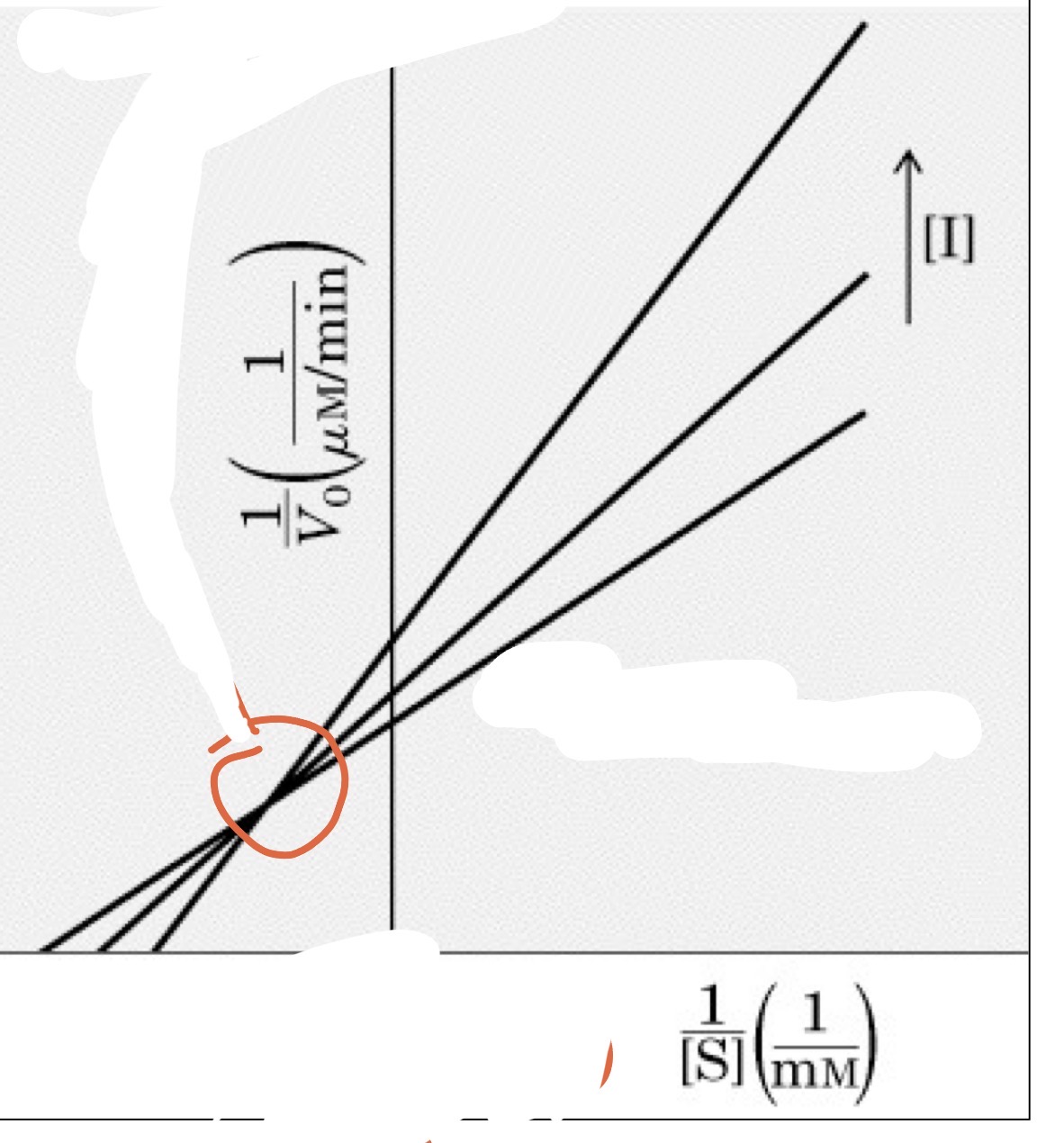
mixed inhibition
Both Vmax (decrease) and Km change (K could increase or decrease)
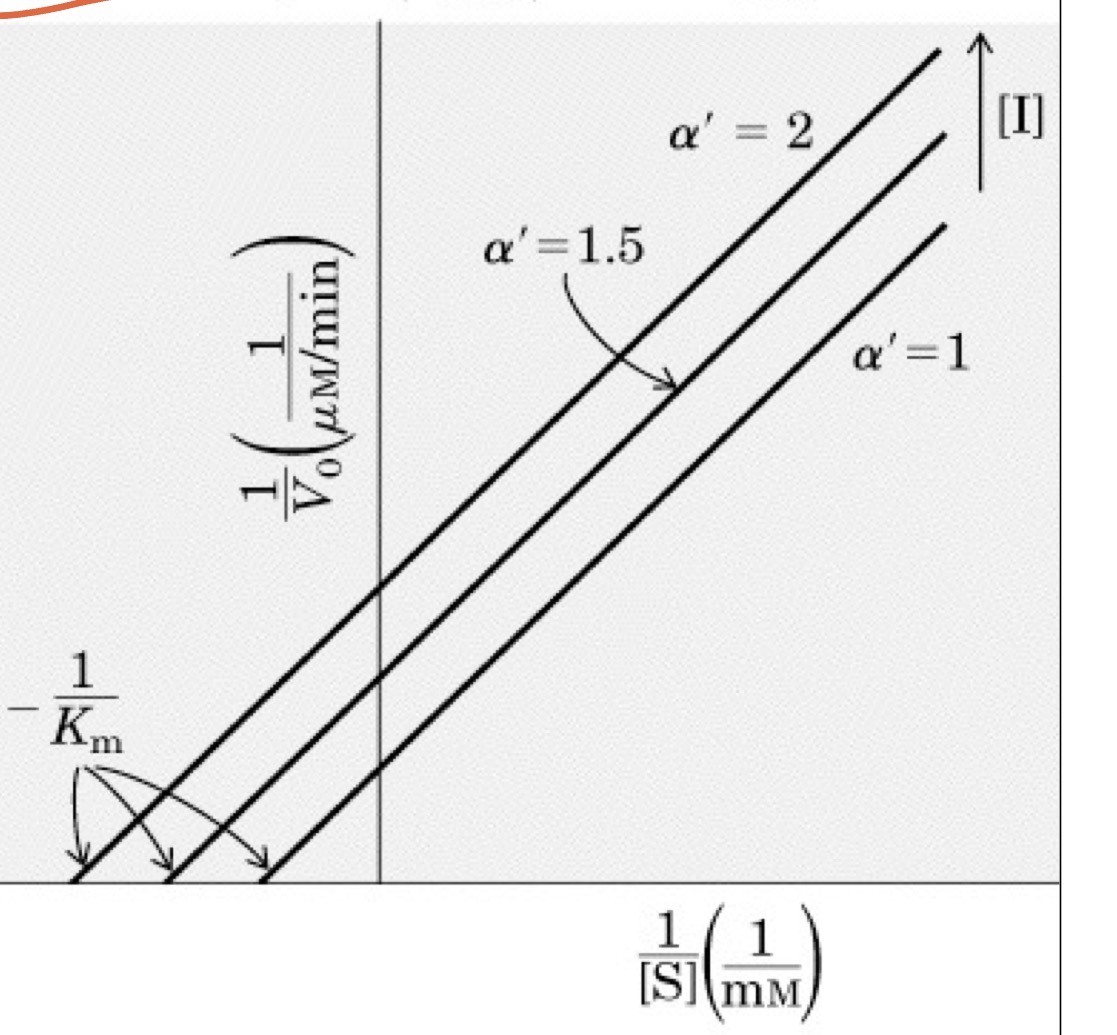
Uncompetitive Inhibition
Inhibitor can only bind to ES compelx
Both Km and Vmax are decreased
Competitive Inhibition
Noncompetitive Inhibtion
Mixed Inhibition
Uncompetitive Inhibtion
Serine Protease
Class of enzymes with a reactive serine OH at their active site
Serine Protease enzymes are
digestive enzymes that catalyze the hydrolyssi of peptide bonds
Chymotrpsin, trypsin and eleastase are all
Serine Protease digestive enzymes
Protease
Breakage of peptide bonds
What class of protease is chymotrypsin?
Serine
Chymotrypsn in vivo specifically hydrolyzes
peptide bonds on the carboxyl side of aromatic bulky side chains
esters
Bulk aromatic amino acids
Phenylalanine (Phe), Tyrosine (Tyr), and Tryptophan (Trp)
How many amino acids are in the active form of chymotrypsin
241
Overall rate determining step
Slow step (usually after the burst phase)
Burst phase
Inital reaction phase were a lot of product is formed quickly
Chymotrypsin specficity
bulky nonpolar
Chymotrypsin binding pocket
Large and nonpolar
Trypsin specificity
Arg and Lys (+) residues
Trypsin binding pocket
Negatively charged residues
Elastase specificty
Small nonpolar
Elastase binding pocket
Thr and val residues close off binding pocket so that only small residues can be accommodated
Catalytic triad
Serine, Histidine, Aspartic Acid
Enolase
catalzyses a step of glycosis
Enolase Co-factor
Mg
Enolase facilates a
dehydration
Ribonuclease A is secreted by the
pancreas
Ribonuclease A catalzyes the
hydrolysis of phosphodiester bonds in RNA