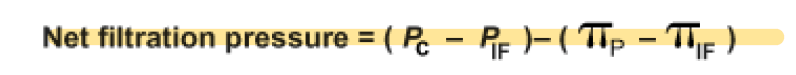Biophysics lecture 6 revision
1/18
Earn XP
Description and Tags
Use these flashcards to study for lecture 6.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
19 Terms
Why do small molcules diffuse across the lipid bilayer without the expenditure of energy?
The small molecules that directly diffuse across the bilayer do so because of the inherent kinetic energy driving their motion, which is observed at temperatures above absolute zero (0K → -273.16 °C). The higher the temperature, the higher the kinetic energy and, therefore, the more motion the molecules experience.
Relationship between concentration gradients and diffusion rate
The steeper the concentration gradient, the faster the diffusion rate is, making them directly proportional to each other. In addition, the likelihood that a molecule will move from a higher concentration to a lower concentration of that same molecule increases.
Fick’s Law
Describes the phenomenon that states that molecules tend to flow from areas of high concentration to areas of low concentration.
\frac{dM}{\differentialD t}=-DS\left(\frac{dc}{\differentialD x}\right)
Where \frac{dM}{\differentialD t} represents the no. of atoms crossing area A per unit time
−D represents the diffusion coefficient/diffusivity (negative means mass transport down concentration gradient)
S represents the cross-sectional area
\frac{dc}{\differentialD x} represents the concentration gradient
Diffusion coefficient equation

Thickness of the membrane
The thicker the membrane, the longer the distance the solute has to travel to diffuse, thus leading to a lower rate of diffusion.
Gay Lussac’s Law
The pressure of a given amount of gas at constant volume (in a container) is directly proportional to the Kelvin temperature.
As a gas in a constant volume is heated, the kinetic energy of the gas molecules rises, making them faster and increasing the number of collisions on the walls of the container, thereby increasing pressure.
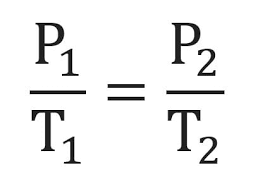
Boltzmann constant

Factors that can influence rate of diffusion
1) Temperature → The higher the temperature, the faster the rate of diffusion is and the faster the equilibrium is reached.
2) The state of the solvent → Diffusion occurs much faster in gases than in liquids and solids.
3) The size of the molecules → Smaller molecules diffuse faster than larger molecules.
4) The diffusion gradient → The steeper the difference between the ECM and cytosol is, the faster the rate of diffusion. If the concentration is equal in both locations, diffusion is terminated.
Osmosis
The movement of water from areas of high concentration to low concentration of water through a semipermeable membrane.
Note that water has a higher likelihood of flowing from areas of low solute concentration to high solute concentration, because in areas of high solute concentration, the large solutes that do not easily diffuse across the semipermeable membrane may mechanically block water molecules from passing through to the side with lower solute concentration, or electrically influence the water molecules (such as in the case of sodium and chlorine ions).
No metabolic energy is needed in osmosis.
Jet flow

Hydrostatic and osmotic pressure (or oncotic pressure) in capillaries
In the arterial end of the capillary bed, hydrostatic pressure is high due to the high-pressure flow of arterial blood. This high hydrostatic pressure is what drives oxygen and nutrients outside of the pores of the capillary beds. As the blood moves towards the venous end of the capillary bed, osmotic pressure (specifically oncotic pressure) is what helps reabsorb waste products (like carbon dioxide and urea) and water that mainly stems from cellular respiration. This is due to the higher concentration of proteins, like albumin, inside the walls of the capillaries that cannot diffuse outside of the capillary. So, following the rules of osmosis, the water flows from areas of low solute concentration (the interstitial fluid in this case) to areas of high solute concentration (the lumen of the capillaries) until equal pressure is established.
A disruption in this process can lead to edema (swelling of parts of the body due to excess fluid buildup in the extracellular space that tissue inhabits. This can be caused by high hydrostatic pressure resulting from hypertension (causing more fluid to be pushed out) or low albumin levels in the capillary bed, leading to lower oncotic pressure (so less fluid is reabsorbed).
Osmolarity
Describes the total number of solute particles in relation to the volume of the solution. Unit is osmoles per liter (osm/L).
Where phi represents the osmotic coefficient, which depends on the degree of dissociation of the dissolved substance. This value is usually between 0 and 1, with 1 indicating complete dissociation of a compound. However, the value may be higher than 1, as is the case with saccharose.
-[n] represents the quantity of ionic particles formed after dissociation.
C represents the molar concentration of the dissolved substance.
Index i identifies dissolved substances.
“Osmoles” refers to the number of particles that contribute to the osmotic pressure of a solution.
![<p>Describes the total number of solute particles in relation to the volume of the solution. Unit is osmoles per liter (osm/L).</p><p>Where phi represents the osmotic coefficient, which depends on the degree of dissociation of the dissolved substance. This value is usually between 0 and 1, with 1 indicating complete dissociation of a compound. However, the value may be higher than 1, as is the case with saccharose.</p><p>-[n] represents the quantity of ionic particles formed after dissociation.</p><p>C represents the molar concentration of the dissolved substance.</p><p>Index i identifies dissolved substances. </p><p><span style="color: rgb(154, 154, 154);"><em>“Osmoles” refers to the</em><strong><em> number</em></strong><em> of </em></span><span style="color: rgb(154, 154, 154);"><em>particles that contribute to the osmotic pressure of a solution.</em></span></p>](https://assets.knowt.com/user-attachments/f69250cd-ac86-47a3-8f92-7eebd85090da.png)
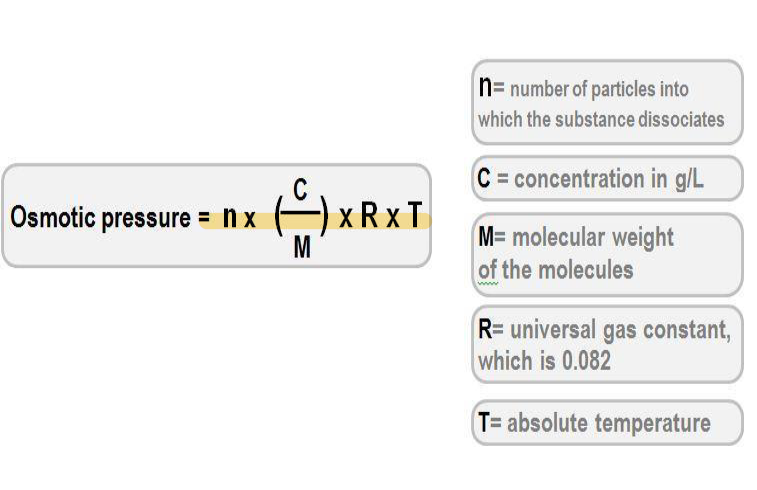
Osmotic pressure equation
Second osmolarity equation

Osmotic pressure equation

Determination of i in dissociation equations
i is found by calculating the total number of ions that result after the dissociation of one compound. For example, NaCl dissociates into 1 sodium and 1 chlorine ion. So, i in this case would equal 2.
van’t Hoff equation (osmotic pressure)

poiseuille's equation
Is used to calculate the filtration speed of blood as it passes through the capillary bed from the arterial end to the venous end.

Specific numbers for hydrostatic and osmotic (oncotic) pressure in arterial and venous ends of the capillary bed
Equation for net filtration pressure:
In the arterial end:
Pc (capillary hydrostatic pressure) = 35 mmHg
PIF (interstitial fluid hydrostatic pressure) = 0 mmHg
πP (osmotic pressure due to plasma protein concentration inside capillaries) = 26 mmHg
πIF (osmotic pressure due to plasma protein concentration in interstitial fluid) = 3 mmHg
Net filtration pressure here is 10 mmHg, promoting filtration.
In the venous end:
Pc (capillary hydrostatic pressure) = 15 mmHg
PIF (interstitial fluid hydrostatic pressure) = 0 mmHg
πP (osmotic pressure due to plasma protein concentration inside capillaries) = 26 mmHg
πIF (osmotic pressure due to plasma protein concentration in interstitial fluid) = 3 mmHg
Net filtration pressure here is -10 mmHg, favoring reabsorption.