Identification of functional groups by test-tube reactions
1/22
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
23 Terms
What are test tube reactions used to do?
Identify the functional group present in an organic compound
To test for alkenes is to test for un…?
Unsaturation
What is the function group for alkenes?
C=C
How do you test for alkenes?
By shaking the sample with bromine water
What is the positive result for testing for alkenes?
Sample turns from orange to colourless
What is the functional group for halogenolkanes?
R—X
R = carbon chain
X = halogen
What three reagents are used to test for halegenoalkanes?
Sodium hydroxide (NaOH), nitric acid (HNO3) and silver nitrate (AgNO3)
How do you test for halogenoalkanes?
Add aqueous sodium hydroxide to the sample and heat gently.
Acidify with nitric acid then add silver nitrate
What is the expected result when testing for halogenoalkanes?
Coloured precipitates of AgX will form
What are the three AgX precipitates which can form?
Silver precipitates made of chlorine, bromine and iodine:
AgCl which forms a white precipitate
AgBr which forms a cream precipitate
AgI which forms a yellow precipitate
What is the functional group for alcohols?
R—OH
What reagent is used for testing for alcohols?
Acidified potassium dichromate (K2Cr2O7)
How do you test for alcohols?
By adding acidified potassium dichromate solution to the sample
What is the positive result for testing for primary and secondary alcohols?
The sample turns from orange to green
What is the positive result for testing for tertiary alcohols?
There is no colour change as there is no reaction
What is the functional group for aldehydes?
R—CHO
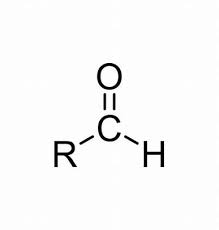
What are two ways of testing for aldehydes?
By warming the sample with Tollen's reagent or Fehling's solution
What is the positive result for testing for aldehydes with Tollen's reagent?
Silver mirror forms

What is the positive result for testing for aldehydes with Fehling's solution?
The blue solution turns into a red precipitate
What is the functional group for carboxylic acids?
R—COOH
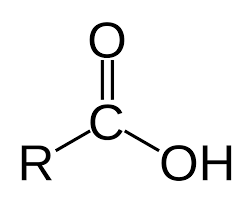
What reagent is used to test for carboxylic acids?
Sodium hydrogen carbonate (NaHCO3)
How do you test for carboxylic acids?
By adding sodium hydrogen carbonate to the sample
What is the positive result for testing for carboxylic acids?
CO2 gas is formed