Cycles of Nutrients
1/64
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
65 Terms
What are the two main functions of carbon?
It is a structural component of organic molecules, and it is an important element in chemical energy storage.
What reaction includes carbon?
Photosynthesis
How does carbon affect the atmosphere?
As carbon exists as carbon dioxide in the atmosphere, this gas is a greenhouse gas which traps heat on earth
What is anthropogenic activity?
Human activities that alter natural environments, especially by increasing greenhouse gases like CO₂ and CH₄, leading to changes such as global warming, pollution, and habitat loss.
How are fossil fuels made?
When dead organisms are buried in the ground over millions of years they become fossil fuels.
What reaction is responsible for returning carbon into the air?
aerobic resiration
Aerobic equation
C6H12O (aq) + O6 (g) = 6CO2 (g) + 6H2O (l)
What is combustion?
a process that releases carbon dioxide into the atmosphere when fossil fuels such as coal, wood and hydrocarbons are burnt.
When does carbon fixation occur?
Carbon fixation occurs during the process of photosynthesis when plants remove carbon dioxide from the atmosphere to make carbohydrates. Sunlight provides the energy for this reaction
Photosynthesis Equation
6CO₂ (g) + 6H₂O (l) = C₆H₁₂O₆ (aq) + 6O₂ (g)
The equation when carbon dioxide is very soluble and large amounts are removed from the atmosphere when it dissolves in the oceans:
CO₂ (g) + H₂O (I) = HCO₃⁻ + H⁺ (aq)
The role of organism in the carbon cycle
Three examples
Carbon Cycle diagram
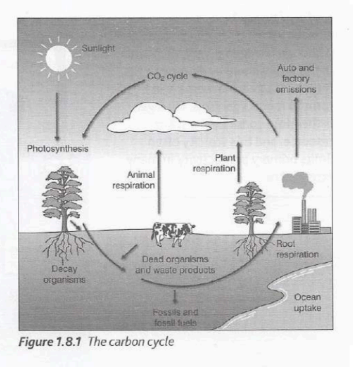
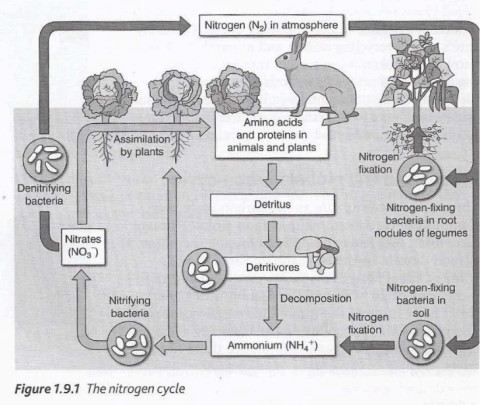
The role of nitrogen
Nitrogen is essential for living organisms because it is needed to make amino acids, peptides, polypeptides, and proteins. Green plants (producers) cannot use atmospheric nitrogen directly, so it must first be converted from dinitrogen gas (N₂) into ammonia (NH₃). Plants then use nitrogen to form proteins, which are passed to consumers through feeding.
Nitrogen exists in both inorganic forms (e.g. ammonia, nitrates) and organic forms (e.g. amino acids and nucleic acids). It is continuously recycled in ecosystems through processes such as nitrogen fixation, nitrification, ammonification, and denitrification, which are mainly carried out by microorganisms.
Nitogen Fixation
Nitrogen fixation is the process that converts atmospheric nitrogen (N₂) into ammonia (NH₃) or ammonium ions (NH₄⁺). It is a multi-step process catalysed by the enzyme nitrogenase.
It is carried out by free-living bacteria such as Azotobacter, symbiotic bacteria such as Rhizobium in root nodules of legumes, and cyanobacteria such as Nostoc.
Lightning may also fix atmospheric nitrogen
During lightning, high-energy discharge causes nitrogen (N₂) to react with oxygen (O₂), forming nitrogen oxides. These dissolve in rainwater to produce weakly acidic solutions, adding nitrates to the soil.
Lighting equation
N₂ + 2O₂ → 2NO₂ + H₂O → 2HNO₃
N₂ + O₂ → 2NO + H₂O → 2HNO₂
Nitrification
Nitrification is a series of oxidation reactions in which ammonium compounds are converted into nitrates, releasing energy used by bacteria.
Nitrite-forming bacteria (e.g. Nitrosomonas) convert ammonia (NH₃/NH₄⁺) into nitrites (NO₂⁻). Another group of bacteria (e.g. Nitrobacter) convert nitrites into nitrates (NO₃⁻).
Nitrates are the main form of nitrogen absorbed by plants. After absorption, they are converted into ammonium and then used to form amino acids, peptides, polypeptides, and proteins.

Denitrification
Denitrification is the process where denitrifying bacteria convert nitrates (NO₃⁻) into nitrogen gas (N₂) and nitrous oxide (N₂O), releasing them back into the atmosphere.
This is carried out by bacteria such as Pseudomonas and Thiobacillus. It commonly occurs in waterlogged soils where oxygen levels are low and there is a high amount of decomposing organic matter.

Humans and the Nitrogen Cycle
Humans affect the nitrogen cycle by adding or removing nitrogen from ecosystems. The use of nitrogen-based fertilisers increases nitrogen levels in soil and contributes to increased nitrogen fixation. Cultivation of leguminous crops also raises populations of nitrogen-fixing microorganisms in the soil.
Burning fossil fuels releases nitrogen oxides (NO, NO₂), nitrous oxide (N₂O), and ammonia (NH₃) into the atmosphere. In addition, agricultural runoff increases nitrate levels in water bodies, leading to eutrophication, which reduces oxygen levels and harms aquatic organisms.
Important Role of Phosphorus
Phosphorus is an essential element found in energy-rich compounds such as ATP, which is important in cellular energy transfer. It is also a key component of nucleic acids (DNA and RNA), making it vital for genetics.
In vertebrates, about 80% of phosphorus is found in bones and teeth. Phosphorus is often a limiting nutrient in aquatic ecosystems, so its availability affects productivity.
Excess phosphorus in water promotes algal blooms. When algae die, their decomposition by bacteria reduces oxygen levels, leading to water pollution and harm to aquatic life.
Phosphorus cycle and Importance
Sedimentary cycle involving movement of phosphate between rocks, soil, water, and organisms | Required for ATP (energy transfer), DNA/RNA (genetics), and bones/teeth in vertebrates
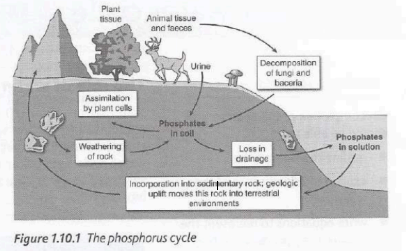
The largest reservoir of phosphorus
Physical effects of eutrophication
Productivity effects of eutrophication
Recreational impact of eutrophication