2.2.13 expansion of the octet
1/11
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
12 Terms
why can elements in period 3 onwards have the possibility of having more than 8 electrons in valence shell
becuase is a d subshell that can accomadate extra electron pairs
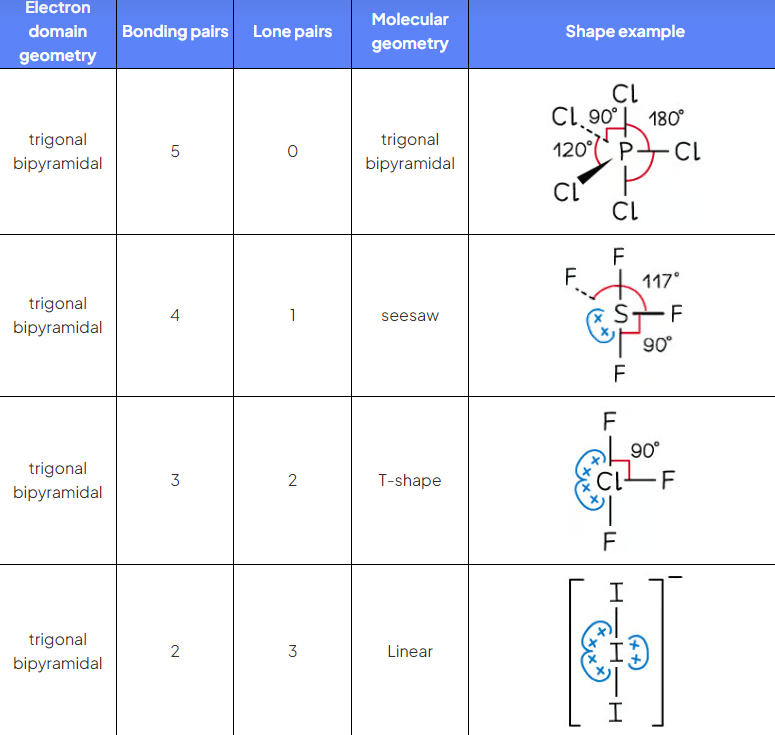
PCl5
ED?
BP?
LP?
EDG?
MG?
5 electron domains
5 BP 0 KO
EDG and MG trigonal bipyramidal 90 & 120
SF4
ED?
BP?
LP?
EDG?
MG?
5 ED
4 BP 1 LP
EDG trigonal bipyramidal 90 and 120
MG see saw, <90 and <120
ClF3
ED?
BP?
LP?
EDG?
MG?
5 ED
3BP 2LP
EDG trigonal bipyramidal 90 and 120
MG T shaped, less than 90
I3-
ED?
BP?
LP?
EDG?
MG?
5 ED
3 BP 3 LP
EDG trigonal bipryamdial 90 and 120
MG linear 180
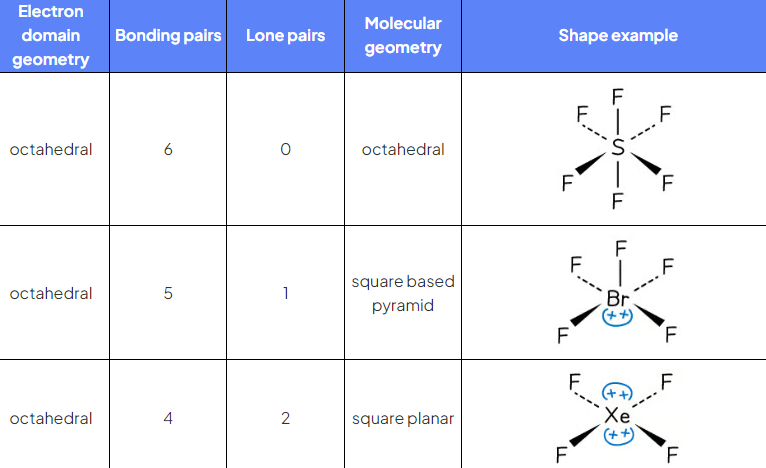
SF6
ED?
BP?
LP?
EDG?
MG?
6 ED
6BP 0LP
EDG and MG octahedral 90
BrF5
ED?
BP?
LP?
EDG?
MG?
6 ED
5 BP 1 LP
EDG octahedral 90
MG square pyramidal <90
XeF4
ED?
BP?
LP?
EDG?
MG?
6 ED
4BP 2 LP
EDG octahedral 90
MG square planar 90
what does molecular geometry refer to
shape of the molecules based on the relative orientation of the atoms w
what is the EDG
relative orientation of all BP and LP
5 ED summary table
6 ED summary table