ROM Exam 4
1/27
Earn XP
Description and Tags
Part of Unit 5 and all of Unit 6
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms
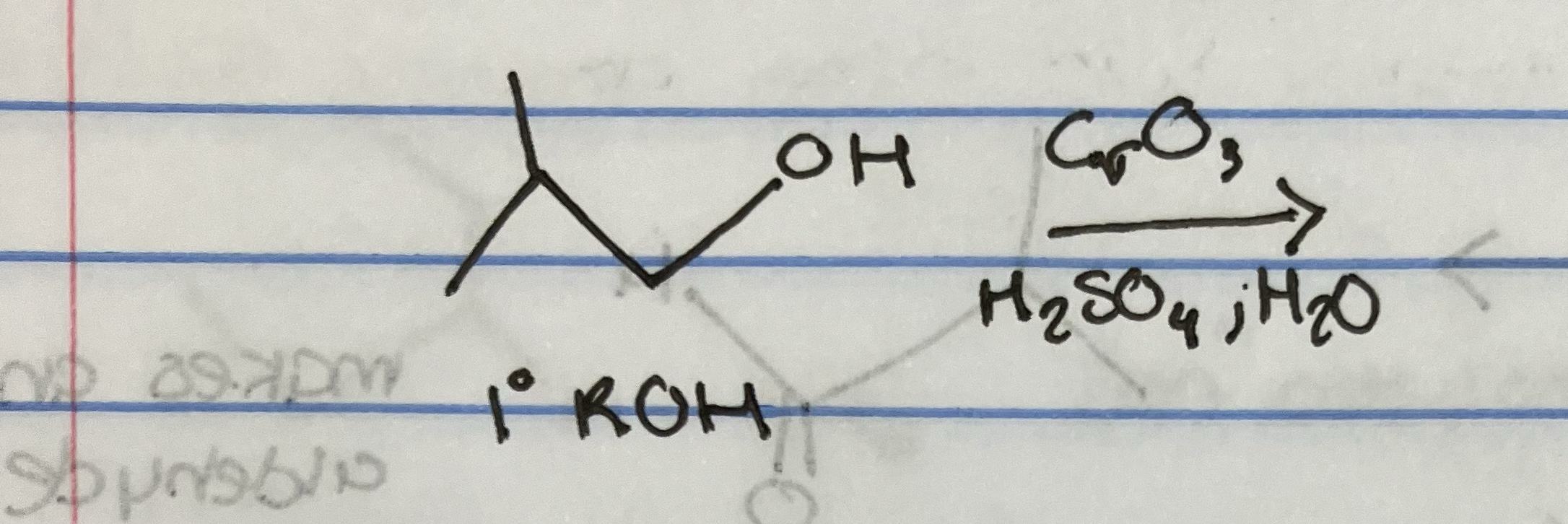
Jones Oxidation on primary alcohol
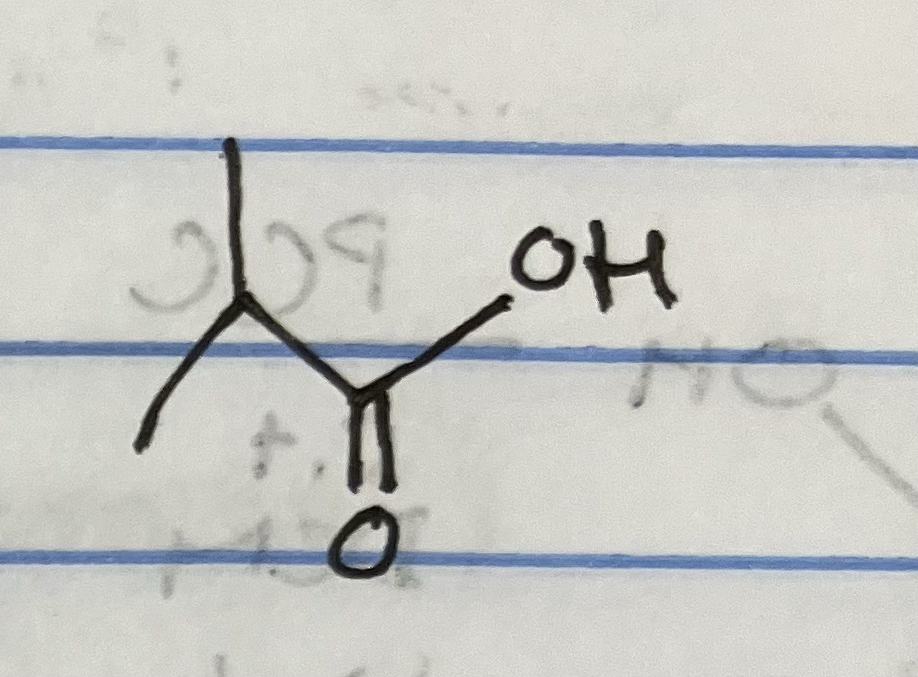
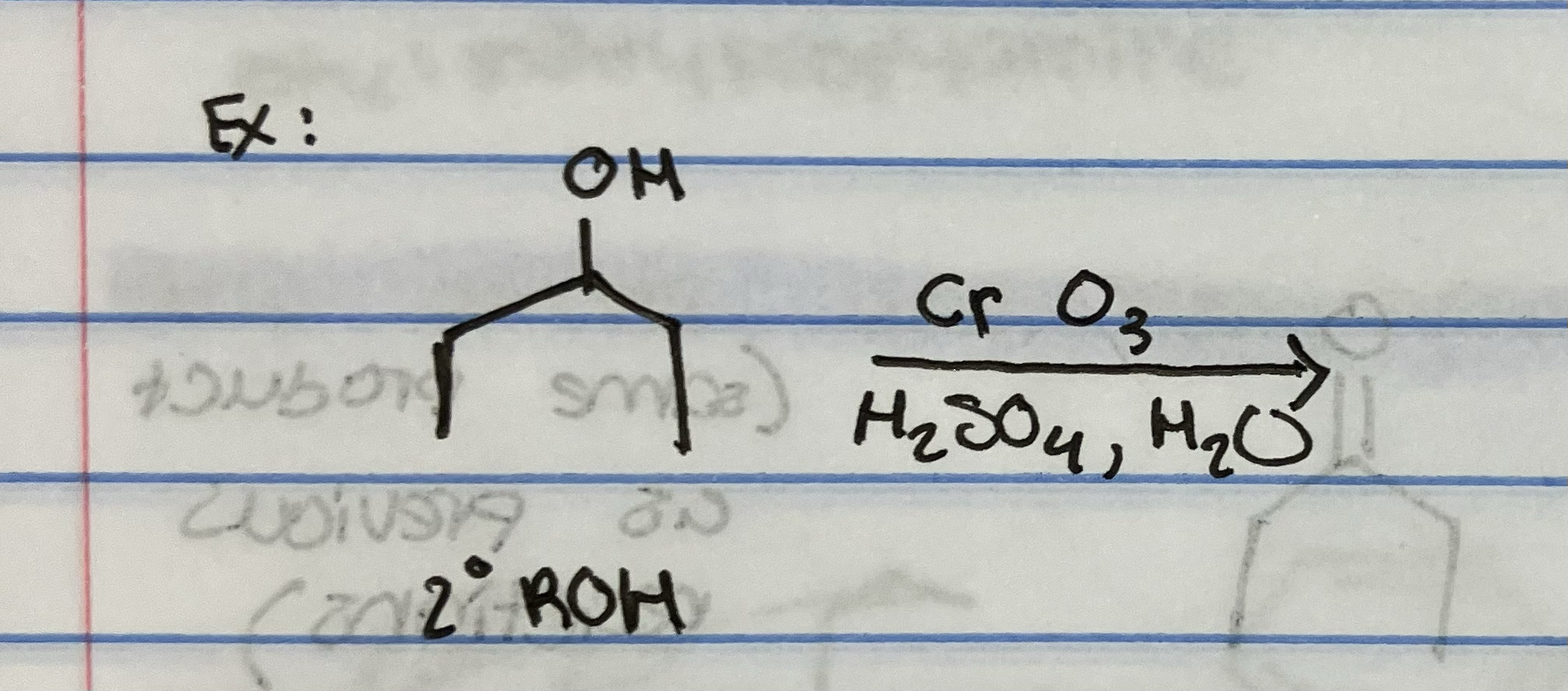
Jones Oxidation on secondary alcohol
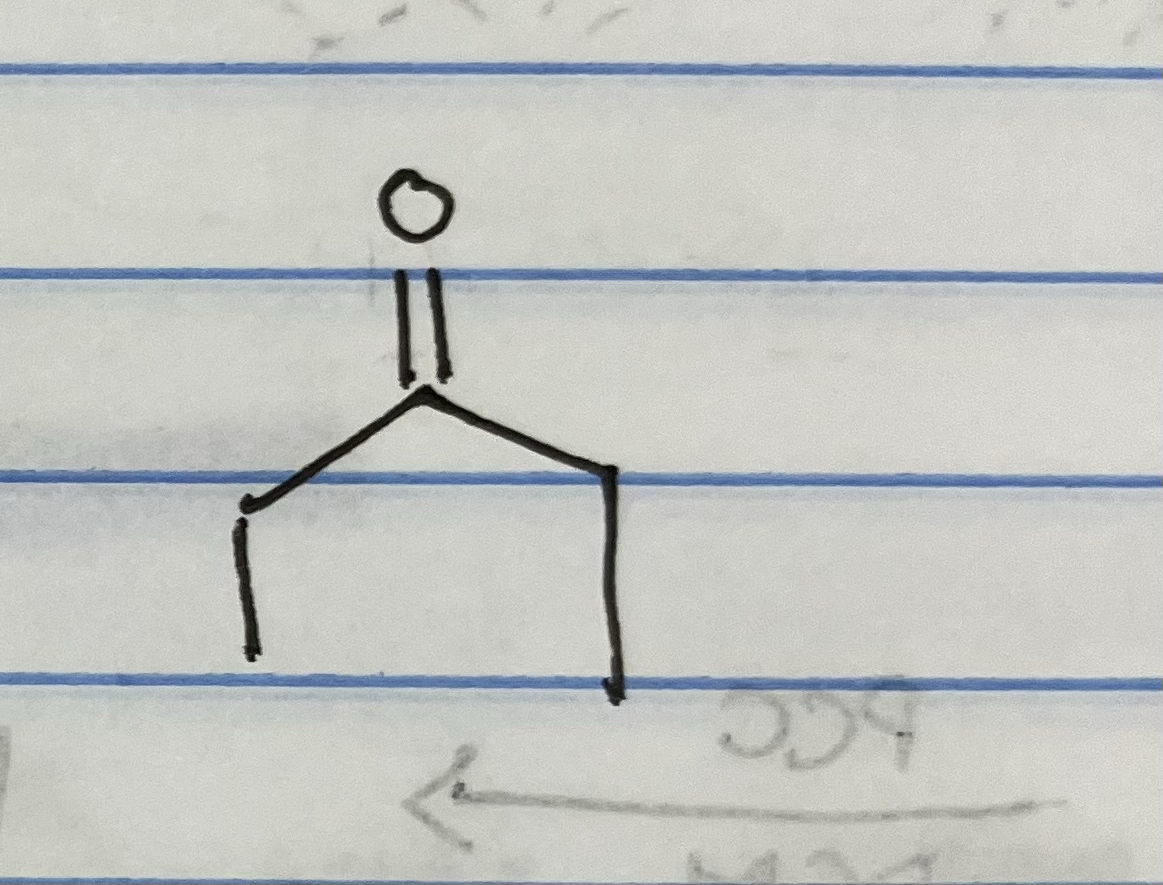
Jones oxidation is
Acidic, and full strong oxidation
KMnO4 oxidation is
basic conditions, full oxidation
Pyridinium chlorochromate (PCC) is
mild oxidation
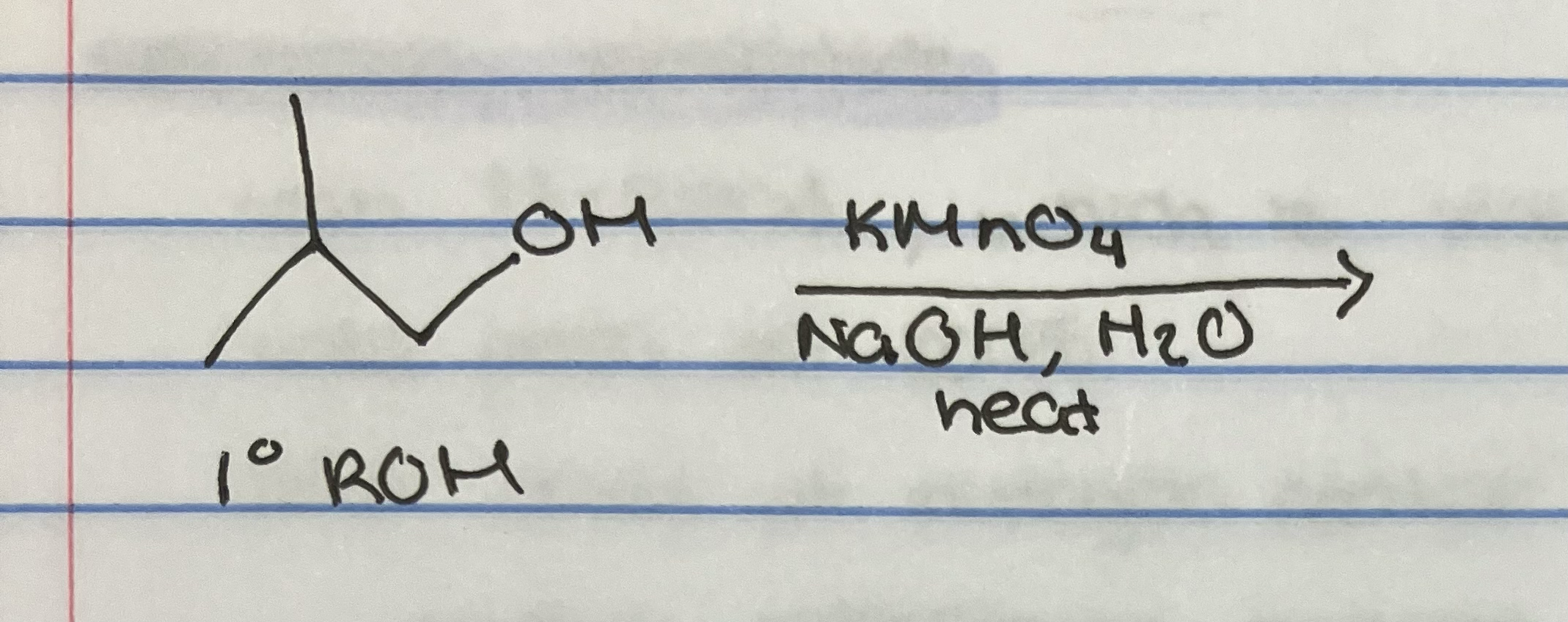
KMnO4 oxidation with primary alcohol
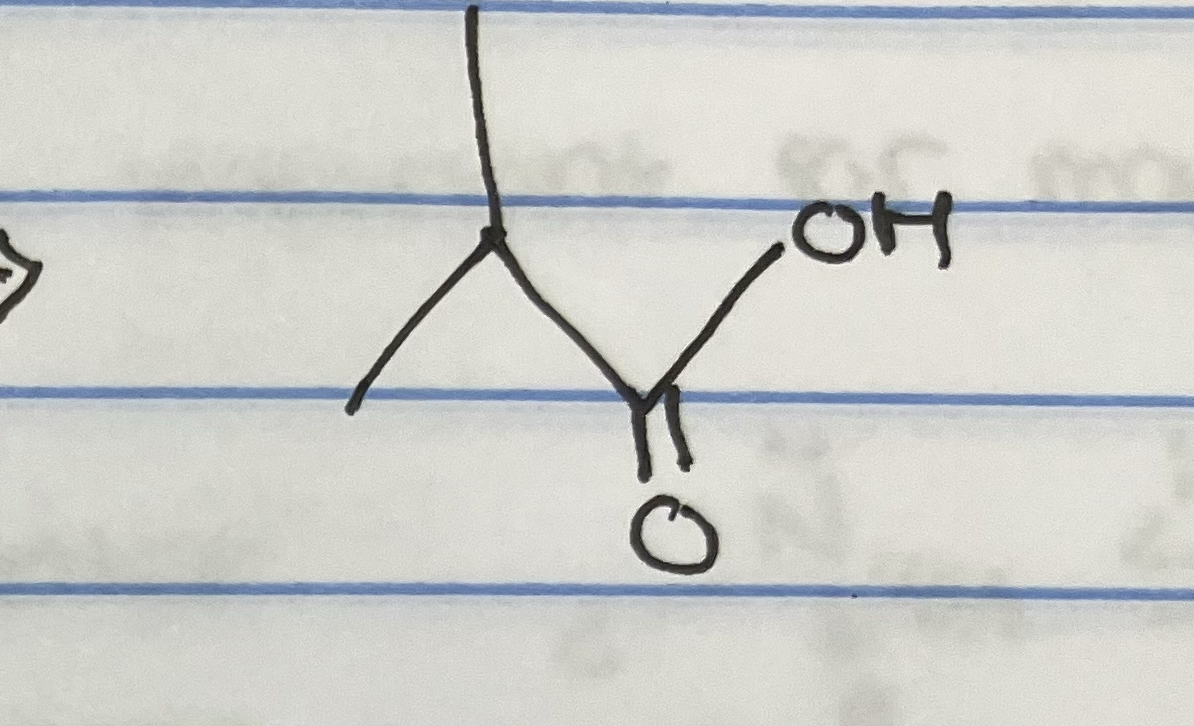
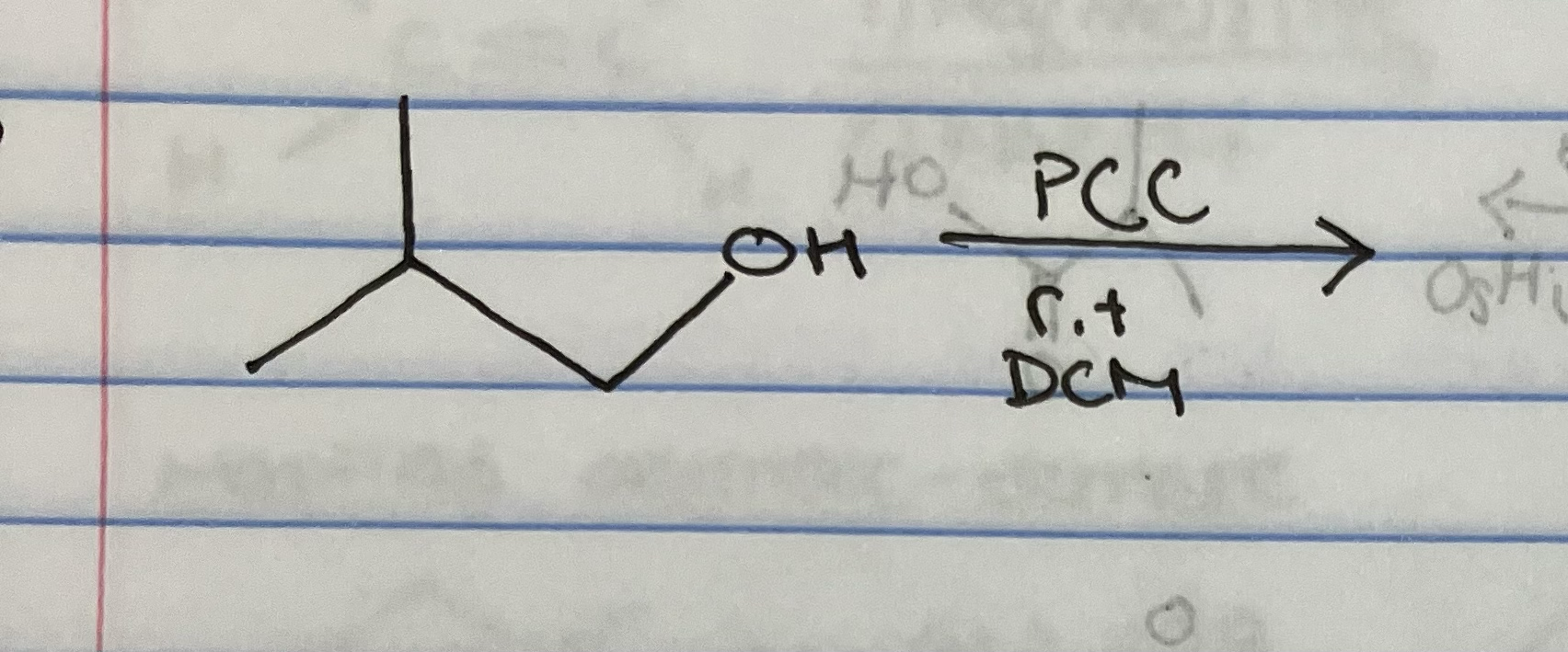
PCC oxidation of primary alcohol
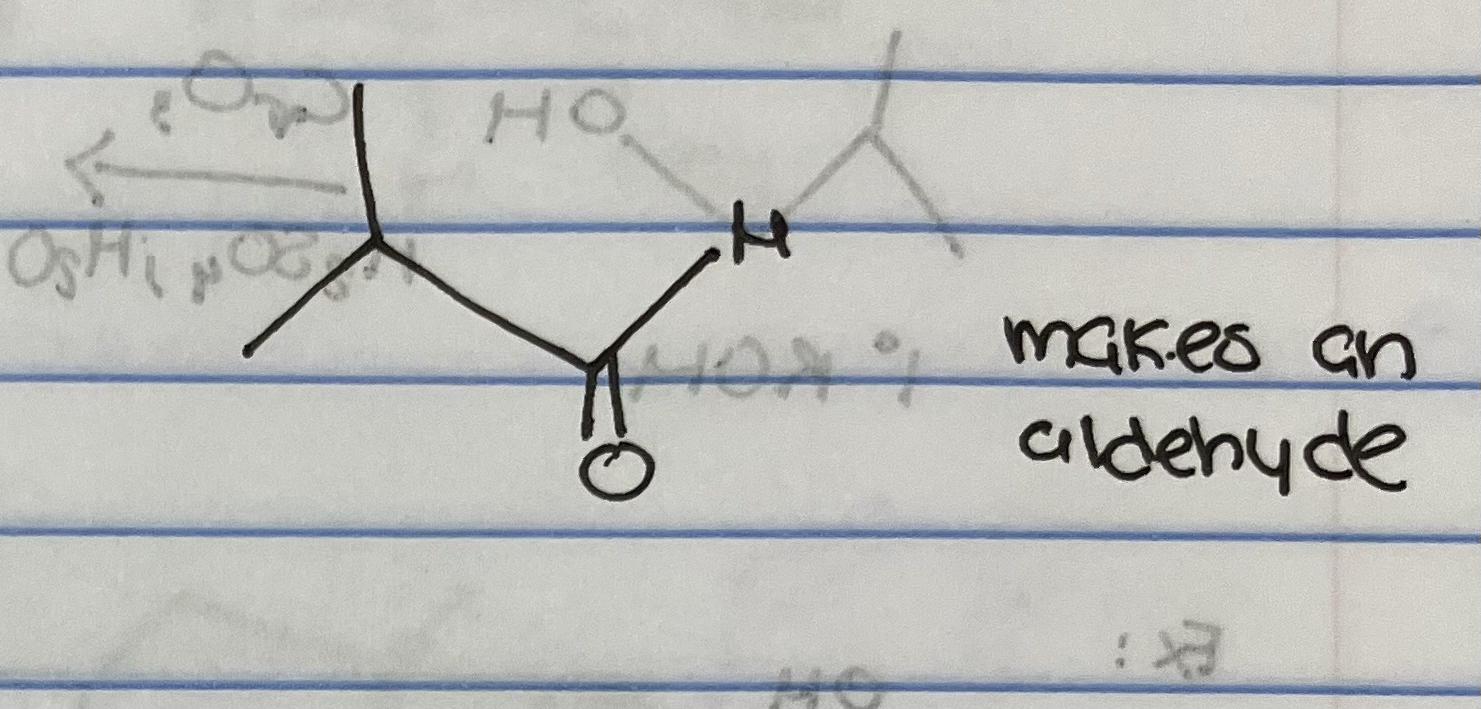
Why are carbonyls so reactive?
Due to it being sp2 carbon, flat, trigonal planar. It is very polar, and the partial positive charge on the carbon makes it very electrophilic.
Why is an aldehyde more reactive than a ketone?
Fewer sterics around the carbon. Less induction, so the partial positive charge is less stable
Reagents used to reduce carbonyls
NaBH4 or LiAlH4
What can generate carbon nucleophiles?
Organothium, Grignard, and Wittig pathway (the ylide)
Why are Grignard and organolithium so useful?
Creates carbon-carbon bonds

This is the same as…
(carbon nucleophile)
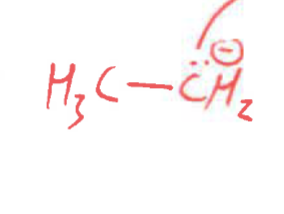
Are carbanions stronger or weaker nucleophiles than anionic oxygen nucleophiles?
More nucleophilic. Carbon is less electronegative, so it is less able to hold a negative charge
Strong oxygen nucleophiles
Are charged
weak oxygen nucleophiles
Are uncharged/neutral
What are acetals used for in synthesis
Used as protecting groups
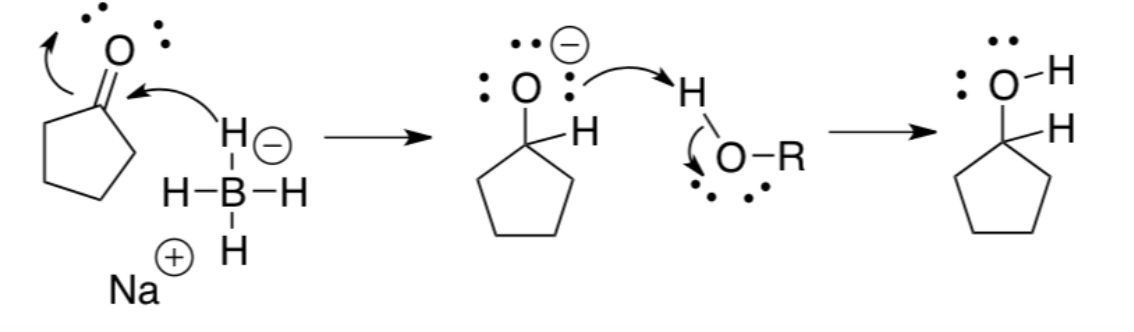
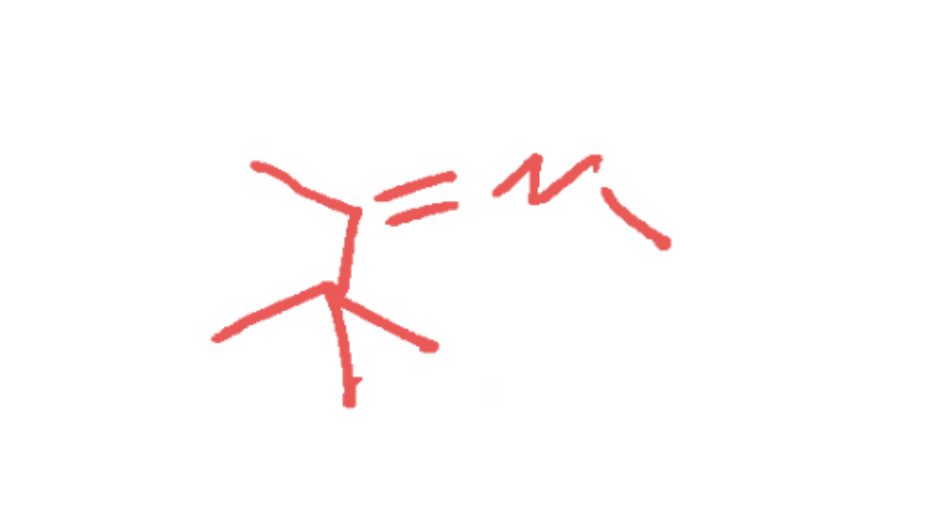
what reaction does this represent?
Carbonyl reduction reaction
-none
ketone
-ol
alcohol
-al
aldehyde
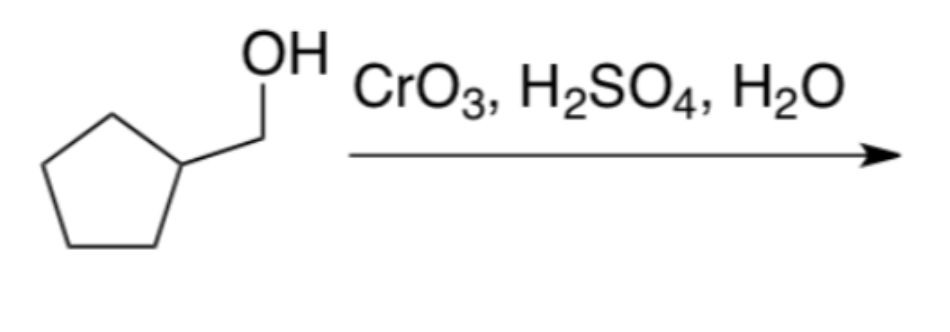
whats the product?
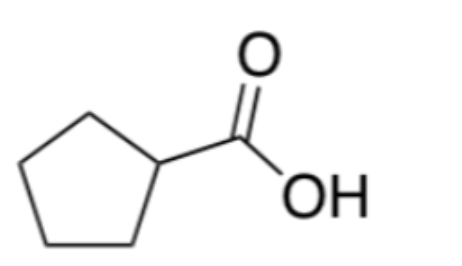
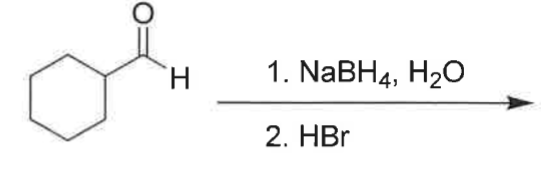
What's the product?
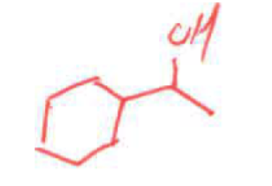
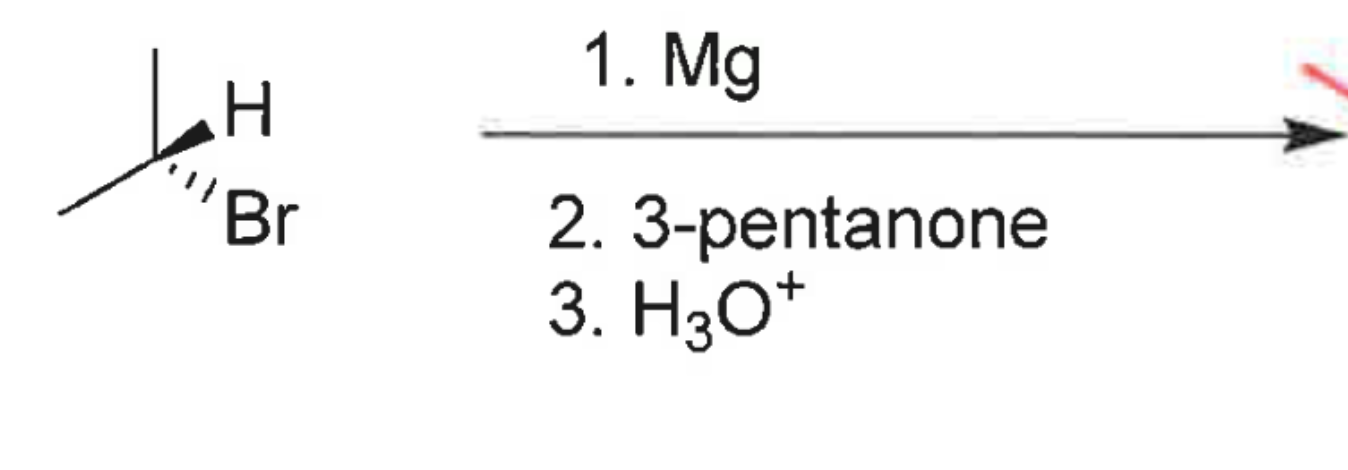
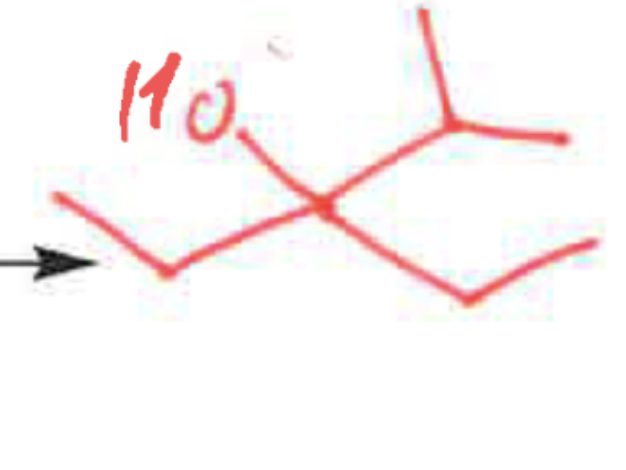
whats the product
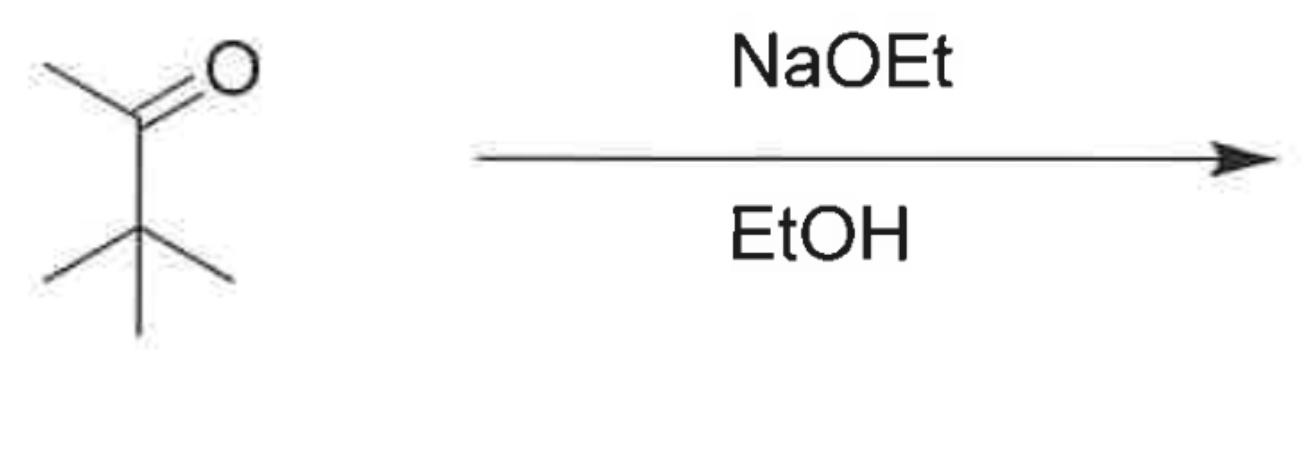
whats the product
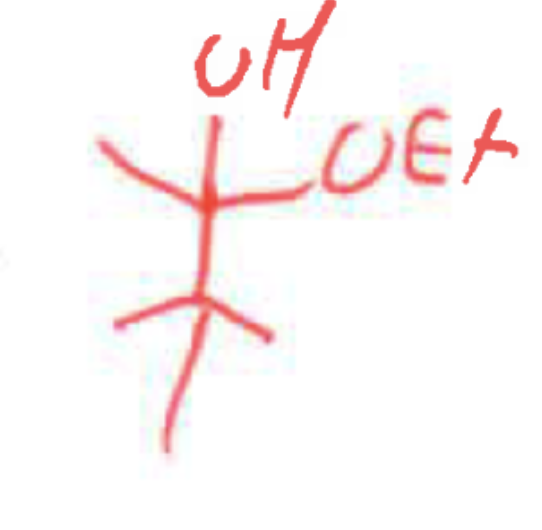
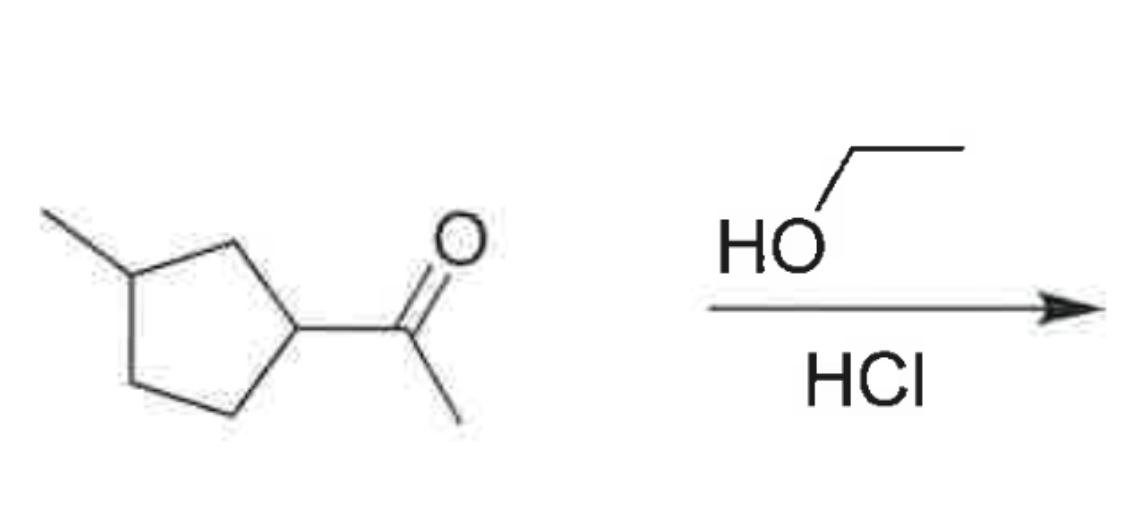
whats the product
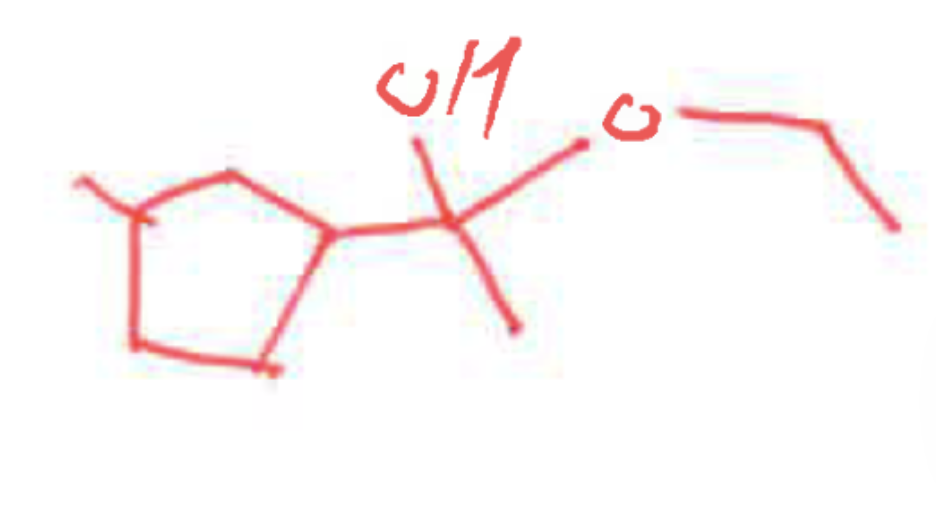
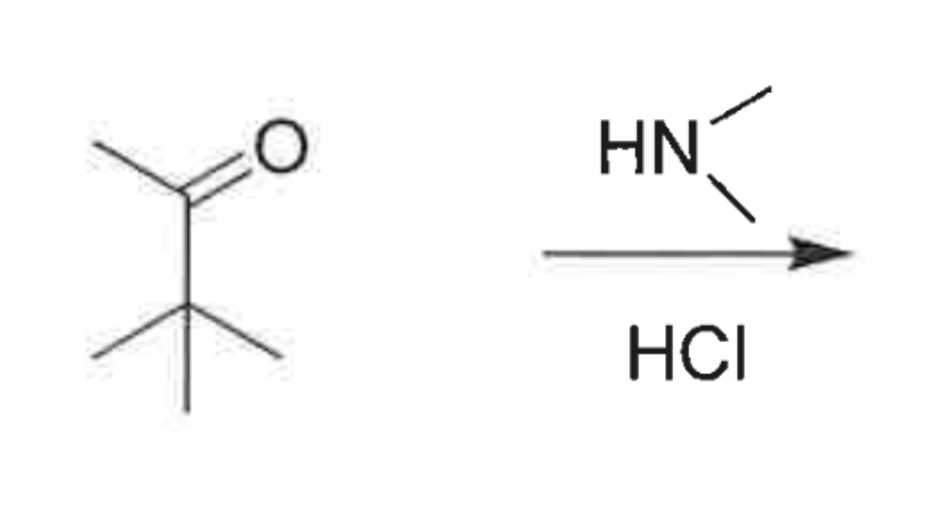
whats the product?

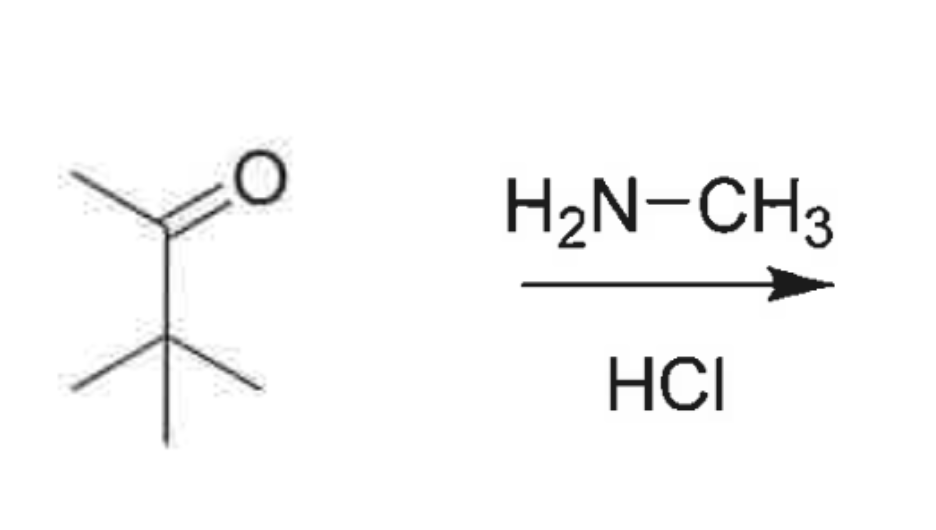
whats the product