PHRM 4180 Final Exam: Anti-Tumor Antibiotics
1/64
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
65 Terms
Anti-tumor antibiotics derived from:
Streptomyces species
How do anti-tumor antibiotics work?
They interact with DNA, generally through intercalation or insertion between DNA base pairs
This interaction either inhibits the enzymes of DNA and/or RNA synthesis or induces strand breaks in DNA
Bleomycin
Mixture of Bleomycin A2 and B2 as a copper chelate
Bleomycin is chemically considered:
A glycopeptide
Bleomycin can be divided into two regions; what are they?
Metal binding (pyrimidine) region
DNA binding (dithiazole) region
Metal binding region forms complexes with:
Divalent metals such as Fe++ and Cu++
Regions pictured
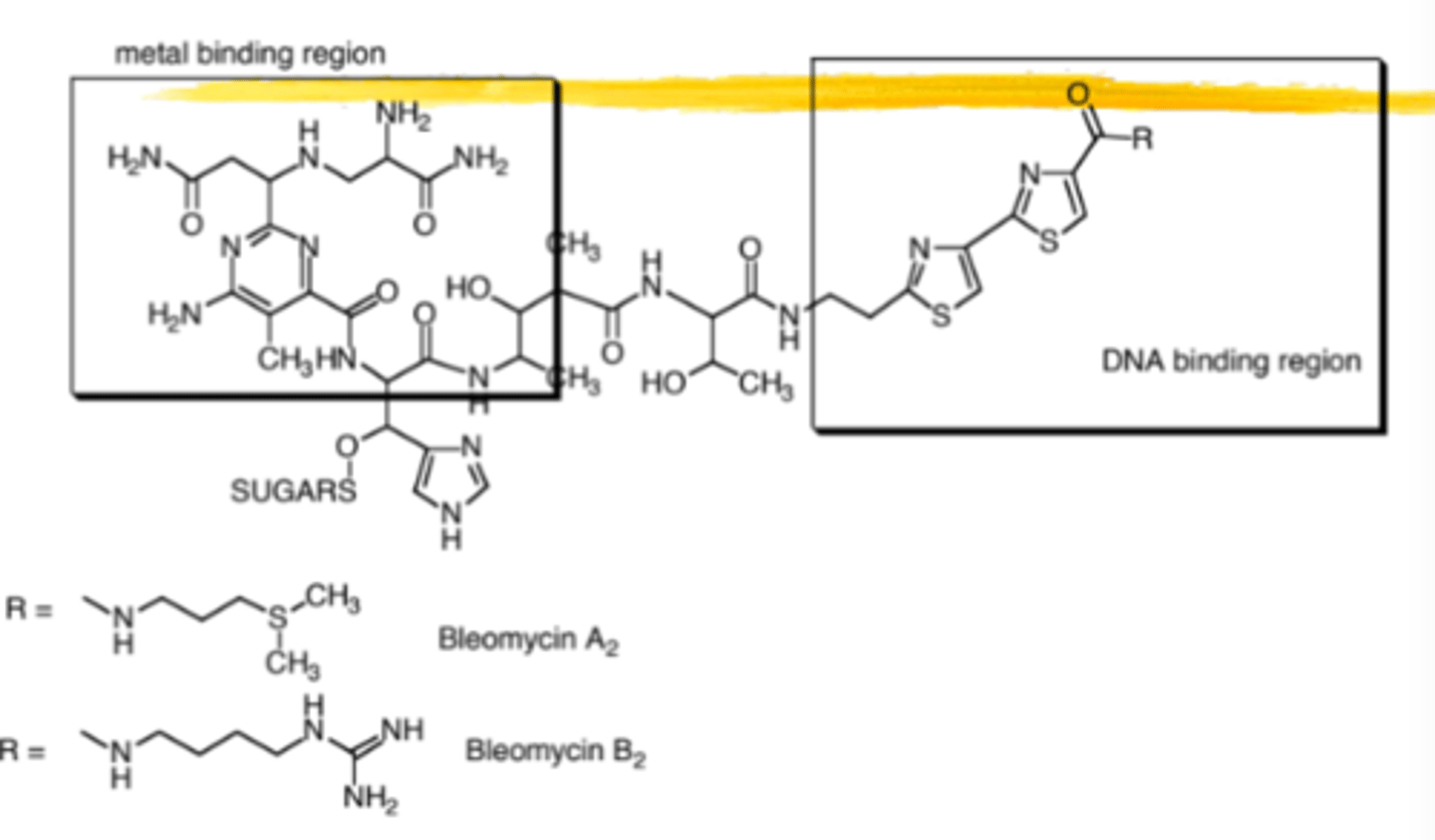
Bleomycin MOA
Dithiazole region intercalates into DNA and Fe is chelated by metal binding region
In presence of O2, iron is oxidized and the oxygen is reduced generating superoxide and hydroxyl radical, 2 ROS that cause DNA strand breaks
Bleomycin routes
IV and instillation into the bladder
Distribution of Bleomycin
Relatively high concentration seen in skin and lung after IV admin
Bleomycin uses
Ovarian cancer
Testicular cancer
Squamous cell carcinomas
Hodgkin's and non-Hodgkin's lymphoma
Bleomycin ADRs
Pigmentation of the skin and nail beds
Erythema and ulceration of elbows, knuckles, and other pressure points (cell memory)
Pulmonary toxicity (DL)
Little BMS
NV
Headache
Pulmonary toxicity
Begins with dry cough, fine rales, infiltrates on CXR
May progress to pulmonary fibrosis
Bleomycin sensitizes what?
Tumor and normal tissue sensitized to radiation
Actinomycin D structure
Composed of a planar (phenoxazone) ring system and two flexible cyclic peptides
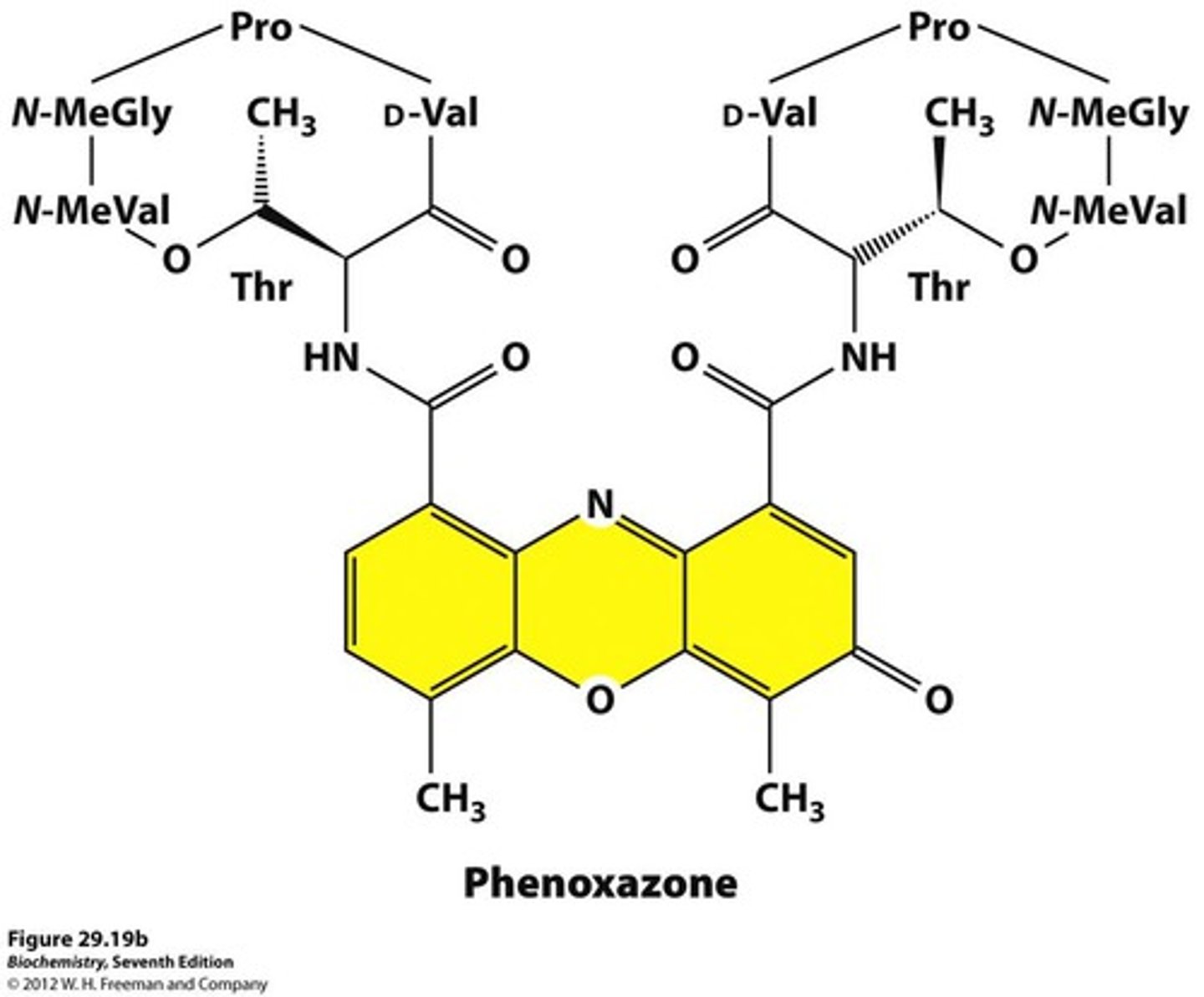
Actinomycin D MOA
Planar ring system intercalated between G-C base pairs of DNA, where G is on opposite strands
Cyclic peptides extend into minor groove
End result of MOA
Transcription of DNA by DNA dependent RNA polymerase is inhibited
Actinomycin D secondary MOA
Also causes single strand breaks in DNA either by free radical generation or by inhibition of topoisomerase II
Actinomycin D route
IV
Actinomycin D uses
Testicular cancer
Sarcomas
Wilm's tumor
Actinomycin D ADRs
NV (pre-medicate to avoid)
BMS (DL)
Sore throat and mouth ulcers common
Actinomycin dose adjustment
Reduce dose in renal and hepatic impairment
Anthracyclines structure
Tetracyclic planar ring system
Quinone and hydroquinone next to each other allow compounds to function as EDG/EWGs
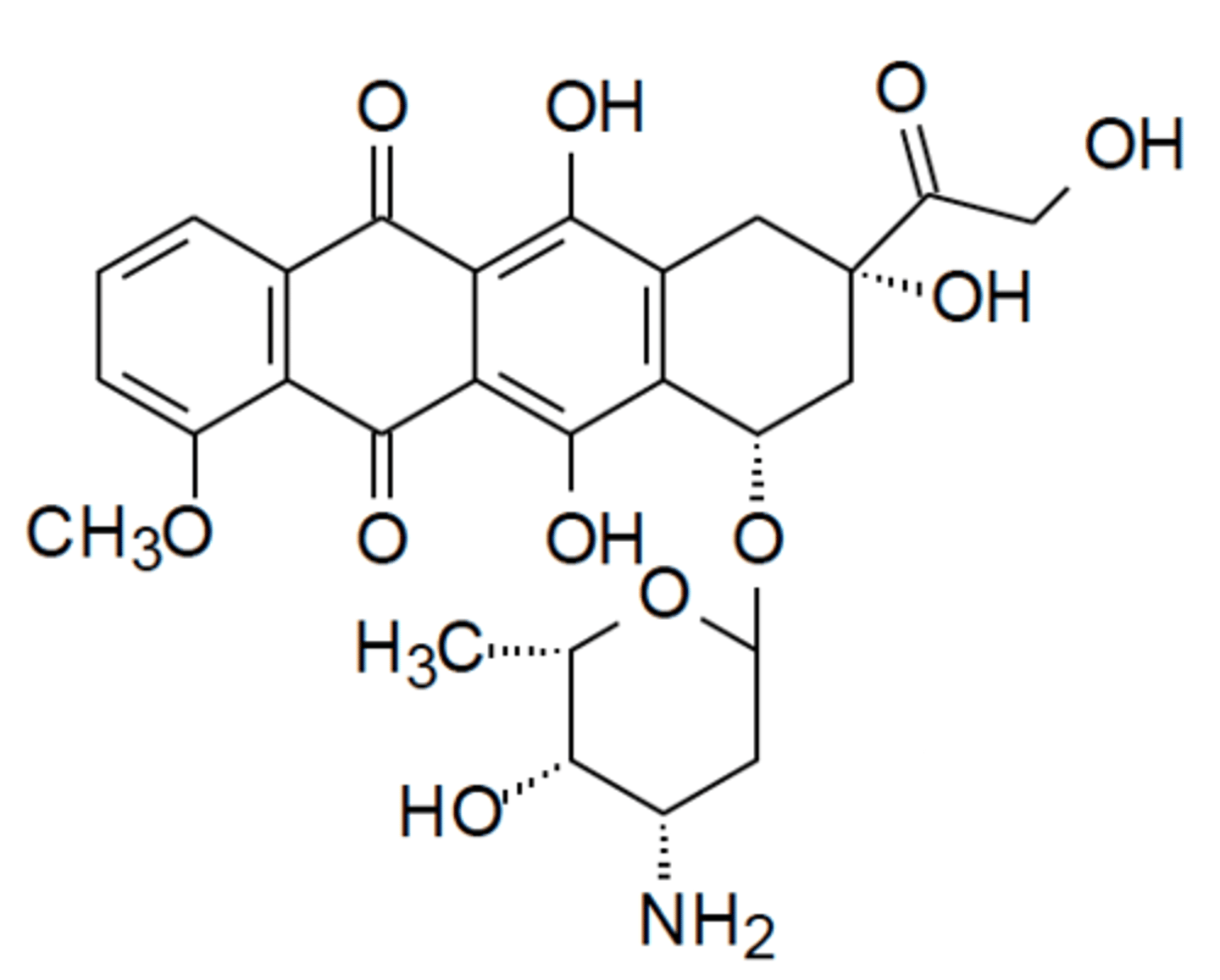
Anthracyclines MOA
Planar ring structure intercalates into DNA, unlike Actinomycin D there is no base pair preference
Sugar portion of Anthracyclines located where?
Minor groove
MOA interaction causes:
Inhibition of topo II's breakage reaction, which leads to DNA strand breaks, not totally known
Anthracyclines may also chelate:
Iron, and iron chelate can form complex with DNA to generate free radicals
Iron chelation MOA place in therapy
Mechanism probably minor in overall cytotoxicity
Con of drug/Fe complex
Primary reason cardiotoxicity is associated with anthracyclines
Two types of cardiotoxic changes seen with anthracyclines
Early transient ECG changes
Delayed progressive cardiomyopathy
Early cardiotoxic changes seen
Characterized by tachycardia, extra-systolic contractions and ST-T wave alterations
Duration of cardiotoxic ADRs
Arrhythmias reverse in hours
ST-T wave changes in a few weeks
Long-term cardiomyopathy manifests as:
Severe rapidly progressing CHF
Doxorubicin total cumulative life dose
>550 mg/m^2
Daunorubicin total cumulative life dose
>900 mg/m^2
Epirubicin total cumulative life dose
>900 mg/m^2
Dexrazoxane MOA
EDTA derivative that effectively competes for iron with anthracyclines, reducing incidences of CHF and increasing total cumulative dose allowed
Dexrazoxane indicated for:
Patients who have received 300 mg/m^2 of doxorubicin and who need to receive additional therapy
Dexrazoxane ADR
Increased BMS
Dexrazoxane dosing
Dosage 10:1 Zinecard:Doxorubicin
Doxorubicin uses
Wide range of tumors
Breast, lung, ovarian, thyroid, GI
Usually used in combo
Doxorubicin ADRs
CHF (DL)
Red discoloration of urine
Sensitization of normal and tumor tissue to radiation
Red/peeling skin
Daunorubicin uses
Primarily acute leukemia
Daunorubicin ADRs
CHF (DL)
Red urine
Mouth ulcers
Alopecia
Recall reaction
Epirubicin structure
Structurally similar to doxorubicin but sugar OH is up instead of down
Epirubicin uses
Breast cancer (combo)
CHF in Epirubicin
Low incidence of CHF if <550 mg/m^2 cumulative dose
Idarubicin uses
Acute leukemia
Idarubicin total cumulative lifetime dose
150 mg/m^2
Idarubicin issue
Metabolite has 55h half-life and is cardiotoxic
Valrubicin is different because
No IV use, instillation into bladder for those who have failed primary therapy for bladder carcinoma
Valrubicin ADRs
Red urine 24h after instillation
Mitoxantrone
Anthracycline analog (synthetic)
What makes mitoxantrone effect different?
Reduced free radical production (less cardiotoxic, less NV)
Mitoxantrone MOA
Intercalates into DNA and causes DNA strand breaks by inhibiting Topo II
Mitoxantrone uses
Breast cancer
Leukemias
Lymphomas
Mitoxantrone ADRs
BMS (DL)
Mouth ulcers
Blue discoloration of urine, fingernails, whites of eyes, and around inj site
Pilcamycin has high affinity for:
Ca++ and Mg++
Pilcamycin MOA
Intercalates into DNA with a preference for GC base pairs
Interferes with RNA synthesis
Plicamycin uses
Primarily for hypercalcemia
Testicular cancer (rare)
Plicamycin ADRs
Thrombocytopenia (BMS)
Bleeding disorders due to decreased Ca
Mouth ulcers
CNS depression
Mitomycin
Antibiotic but an alkylating agent
Note aziridine ring
Mitomycin MOA
Bifunctional alkylating agents causing cross linking of DNA
Inducing single strand breaks in DNA
Mitomycin uses
Stomach cancers
Pancreatic cancers
In combo for variety of other cancers
Mitomycin ADRs
BMS (DL)
Pulmonary fibrosis
Recall reaction