Lec 6: Lutein & Garlic Exam 2
1/28
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
29 Terms
Lutein
naturally occurring yellow/orange carotenoid obtained in the diet, commonly found in fruits and vegetables, dark-green vegetables, kale, spinach, turnip greens, and collards
Lutein is only synthesized in
plants, and animals obtain lutein through eating plants
For plants to resist oxidation they...
produce bioactive compounds like lutein
Lutein is an isomer of ...
zeaxanthin, is present un plants as fatty acid esters (lipophilic)
Lutein absorbs
light due to the presence of conjugated double bonds, it absorbs blue light and appears yellow at low concentrations and orange/red at high concentrations
Lutein also exists in
photoautotrophic organisms like algae-spirulina, chlorella
Lutein production in microalgae is .... compared to plants
significantly, 3-6X, higher
Lutein structure
extended conjugated double-bond system, polyene chain exists in cis or trans form. Light, oxygen, heat, pH can convert trans carotenoids into cis carotenoids. This is not ideal because trans-lutein has higher bioavailability
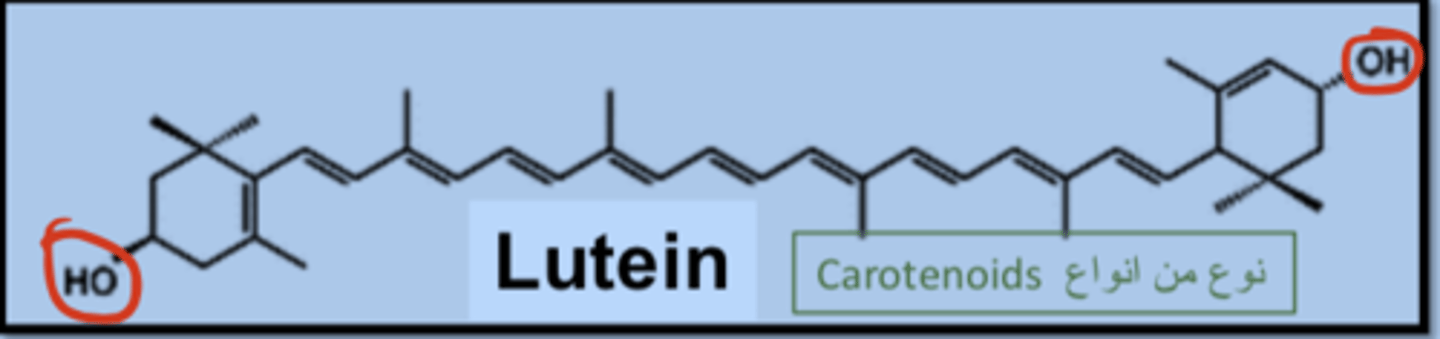
Lutein hydroxyl group position
at both ends of lutein and they react with fatty acids to form mono-esters or di-esters
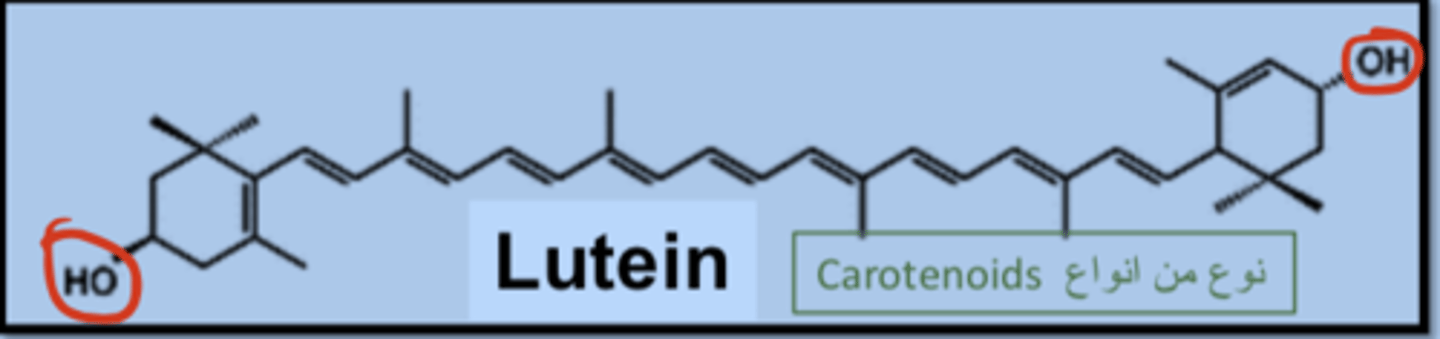
Lutein is an ester within plants and
can be converted to free lutein via saponification with alkali, addition of a base converts ester into free form
Lutein esters are
more stable when subjected to heat/light and maintain stable color profile compared to free lutein
Bioavailability of lutein ester is..... and the biological effects
higher compared to the free form and the biological effects are different for ester and free form
Biological activities of lutein
selective distribution within tissues, macular retina, adrenal gland, liver
prevents age-related macular degeneration, improves skin hydration and elasticity
reduces blood pressure and cholesterol, stimulates immune function and cancer prevention
Lutein safety
not known to be toxic at higher amounts of ingestion >30mg, even consumption at 400mg/kg bw/day does not cause adverse effects
Garlic (Allium sativum) possesses
valuable medicinal properties and is rich is phytochemicals
about 85% of garlic production in the US is in
california
Garlic supplement examples
essential oil, garlic oil macerate, garlic powder, and garlic extract
garlic contains
sulfur containing compounds such as allicin
Precursors to sulfur compounds
gamma-glutamyl-S-alkenyl-L-cysteine (more stable, umami flavor, concentration increases during storage because S-alkenyl degrades) and S-alkenyl-L-cysteine sulfoxides (alliin)
Garlic also contains
micronutrients (calcium, magnesium, phosphorus, potassium, selenium, vitamin C, and folate)
Garlic odor arises from ... when the bulb is damaged
allicin and oil-soluble sulfur compounds, membrane destruction results in organosulfur degradation due to enzymatic action
Alliinase converts
alliin to form odiferous thiosulfinates (allicin)
Bioavailability of allicin
between 65-95%
Cooking degrades
alliinase enzyme so there is no strong smell form allicin (allicin not produce if enzyme is denatured), heat/drying of garlic for capsule supplement with no odor/flavor
In oil form allicin reaches the blood flow in ...
10 minutes
Allicin degrades into
sulfides, ajoene, and dithiins
strong garlic flavor is associated with
higher amounts of volatile sulfur compounds
Biological activities of garlic
antihypertensive, anti-Alzheimer's, anticancer, anti-inflammatory, antimicrobial, antidiabetic
Difference in lutein and zeaxanthin structure
Both consist of long chains of conjugated double bonds with ionone rings on the ends. Zeaxanthin has two beta-ionone rings and lutein has one beta ionone ring and one epsilon ionone ring