Bio Enzymes
1/28
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
29 Terms
Carbohydrate
Organic compounds made up of carbon, hydrogen, and oxygen, typically in a 1:2:1 ratio.
Lipid
A group of organic compounds that are hydrophobic or amphipathic, primarily fats and oils.
Protein
Large biomolecules made up of amino acids, responsible for catalyzing reactions, structural support, and more.
Nucleic Acid
Polymers made up of nucleotide monomers, including DNA and RNA.
Carbohydrate monomer
Monosaccharide, the simplest form of carbohydrate (e.g., glucose).
Carbohydrate polymer
Polysaccharide, formed by the linkage of multiple monosaccharides (e.g., starch, glycogen).
Lipid monomer
Fatty acids and glycerol, which combine to form larger lipid molecules.
Lipid polymer
Triglycerides, which consist of three fatty acids attached to a glycerol.
Amino Acid
The building block of proteins, with a central carbon atom, an amino group, a carboxyl group, and a variable R group.
Polypeptide
A chain of amino acids linked by peptide bonds; it is a polymer of amino acids.
Peptide bond
The bond formed between the carboxyl group of one amino acid and the amino group of another.
Dehydration
A chemical process that involves the removal of water to form a larger molecule.
Hydrolysis
A chemical process that involves the addition of water to break down a larger molecule into smaller units.
Protein Function
The specific role of a protein, determined by its unique shape and chemical properties.
Unique Protein Shape
Determined by the sequence and chemical properties of the amino acids that compose the protein.
Hydrophobic Amino Acids
Typically found in the interior of proteins in aqueous environments, away from water.
Hydrophilic Amino Acids
Located on the surface of proteins, interacting with the aqueous environment.
Enzyme
A biological catalyst that speeds up a chemical reaction without being consumed.
Active Site
The region of an enzyme where substrate molecules bind and undergo a chemical reaction.
Substrate
The reactant molecule upon which an enzyme acts.
Product
The final product(s) of the reaction catalyzed by an enzyme.
Transition State
The temporary state during a chemical reaction when old bonds are breaking and new bonds forming.
Activation Energy
The minimum amount of energy required to initiate a chemical reaction.
Denaturing
The process by which a protein loses its shape and function due to environmental changes.
Optimal Temperature, pH, and Salt Concentration
The specific conditions under which an enzyme exhibits its greatest activity.
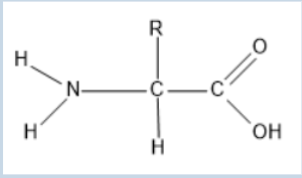
Protein Monomer
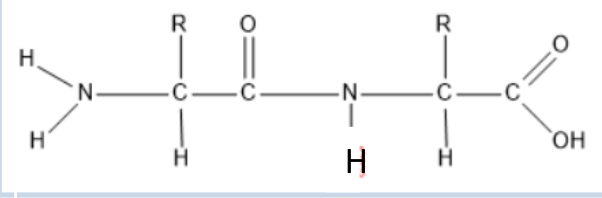
Protein Polymer
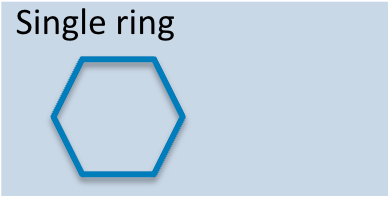
Carb Monomer
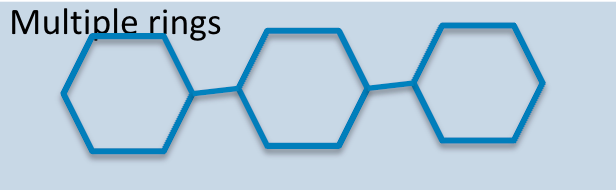
Carb Polymer