Chem Exam 3
1/21
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
22 Terms
Entropy is favorable when…
ΔS > 0 (messy bedroom happens naturally, nature prefers high disorder)
Gibbs free energy is spontaneous when
ΔG < 0 (Products are lower in energy than reactants, reaction proceeds)
Transition state
Fleeting state between reactants and products where activation energy is the highest
What states of matter are omitted from equilibrium expressions?
Solids and Liquids
Keq >>> 1 means…
reaction is extensive/it goes completely to products (Keq > 1.0×10³)
Keq<<<1
Products are not favored, solution mostly has reactants (Keq < 1.0×10³)
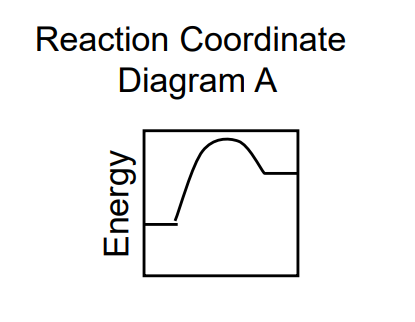
ΔG > 0
Ea forward > Ea reverse
Keq << 1
Not extensive (reactants preferred)
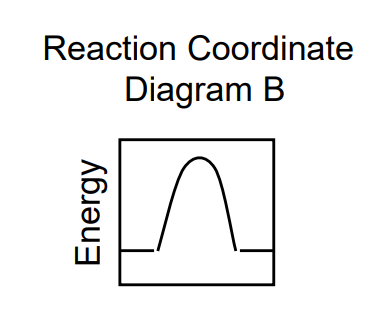
ΔG = 0
Ea forward = Ea reverse
Keq ~ 1
Not extensive
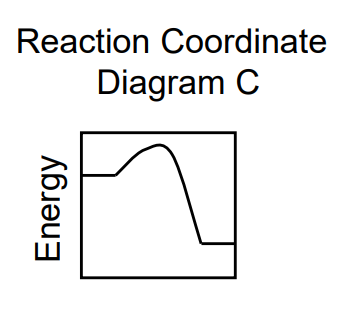
ΔG < 0
Ea forward < Ea reverse
Keq >> 1
Extensive (products preferred)
Le Chatelier’s Principle
if a system at equilibrium is disturbed by changing conditions, the system will respond by bringing itself back to equilibrium
Endothermic equilibrium reactions
Heat is a reactant
When heated, converts reactants into products (shift L→R)
When cooled, converts products into reactants (shift R→L)
Exothermic Equilibrium Reactions
Heat is a product
When heated, converts products into reactants (shift R→L)
When cooled, converts reactants into products (shift L→R)
A higher energy species is
More reactive
Less stable
Strong acid/base
A lower energy species is…
Less reactive
More stable
Weak acid/base
pKa (inverse of Ka)
the stronger the acid the smaller the pKa
the weaker the acid the larger the pKa
The more electronegative the central atom…
the more stable the conjugate base is and the more acidic the acid it came from is (pKa gets smaller the more EN^-)
As atom size (going down the column) increases, pKa…
decreases (acid gets stronger), this is because larger atoms hold their electrons loosely making it easy to lose H^+
Also more stable
When should we look at resonance structures and what should we look for?
When the negative charge is on the same central atom, use resonance structures
If there is a negative charge adjacent to pi bonds, there are multiple resonance structures
What do more resonance structures mean
The more stable the conjugate base is and the more EN^- the central atom
Keq and Stability
Negative Keq means the reactants are favored, this makes them weaker and more stable (THE WEAKER THE ACID THE MORE STABLE)
Extensive reaction means
The reacting acid is stronger than the resulting conjugate base (small to big pKa)
For ΔG, if entropy and enthalpy are in agreement… (ex: both unfavorable)
Temperature doesn’t affect whether it is spontaneous or not