1. Biological Molecules
1/149
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
150 Terms
What are proteins made of?
● Proteins are polymers made of the monomers amino acids
● Contain C, H, O, N, some contain S
● Contain different sequences of amino acids
Give examples of polymers and the monomers from which they're made from.
● Polynucleotide (DNA and RNA) → Nucleotide
● Polysaccharide (Starch, cellulose, glycogen) → Glucose
● Polypeptide (Protein) → Amino acid
Monomer
Smaller / repeating molecules from which larger molecules/polymers are made
Polymer
Molecule made up of many monomers bonded together
What are monosaccharides & give examples
Monomers from which larger carbohydrates are made
e.g. Glucose, fructose, galactose
What is a hydrogen bond?
A/an weak chemical bond between the positive charge on a hydrogen atom and the negative charge on the atom of an adjacent molecule.
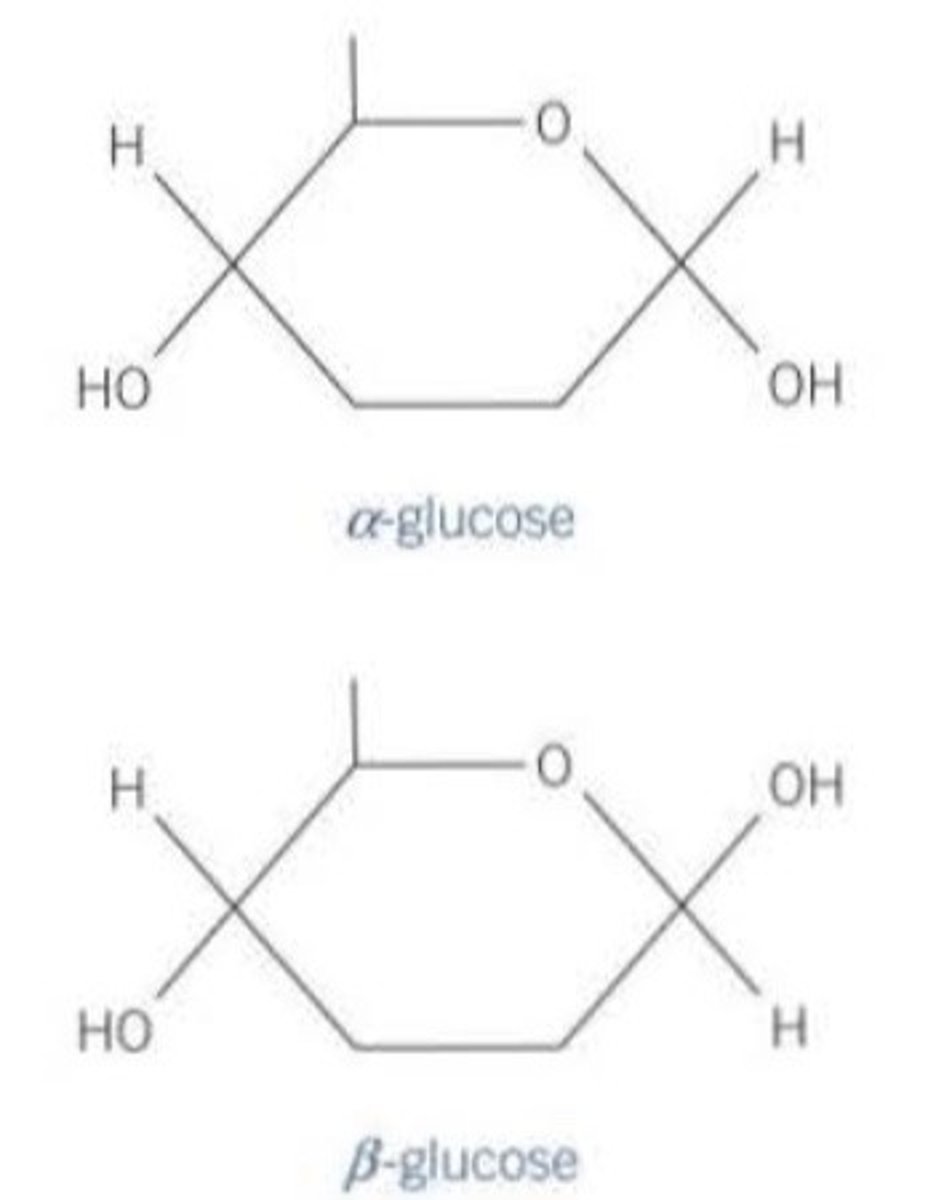
Draw alpha glucose and beta glucose
What is an isomer?
Same molecular formula, different structure
What is the OH group called?
Hydroxyl group
What is the difference between alpha and beta glucose?
In alpha glucose, the OH group is below carbon 1.
In beta glucose, the OH group is above carbon 1.
What do all carbohydrates contain?
carbon, hydrogen, oxygen (C, H, O)
Name the following products:
Glucose + Glucose -->
Glucose + Fructose -->
Glucose + Galactose -->
Glucose + Glucose --> Maltose + Water
Glucose + Fructose --> Sucrose + Water
Glucose + Galactose --> Lactose + Water
A bond is labelled as an alpha-1,4-glycosidic bond. 'Alpha' tells us that...
The first carbon involved in the reaction is on an alpha-glucose molecule.
What are disaccharides and how are they formed? Give examples.
● 2 monosaccharides joined together with glycosidic bonds
● Formed by many condensation reactions, releasing water molecules
● Maltose, sucrose, lactose
What are polysaccharides and how are they formed? Give examples.
● Many monosaccharides joined together with glycosidic bond.
● Formed by many condensation reactions, releasing water molecules
● Starch, cellulose, glycogen
Difference between polymers and polysaccharides
Polymers are large molecules made up of repeating units.
Polysaccharides are specific polymers made up of sugar units linked by glycosidic bonds.
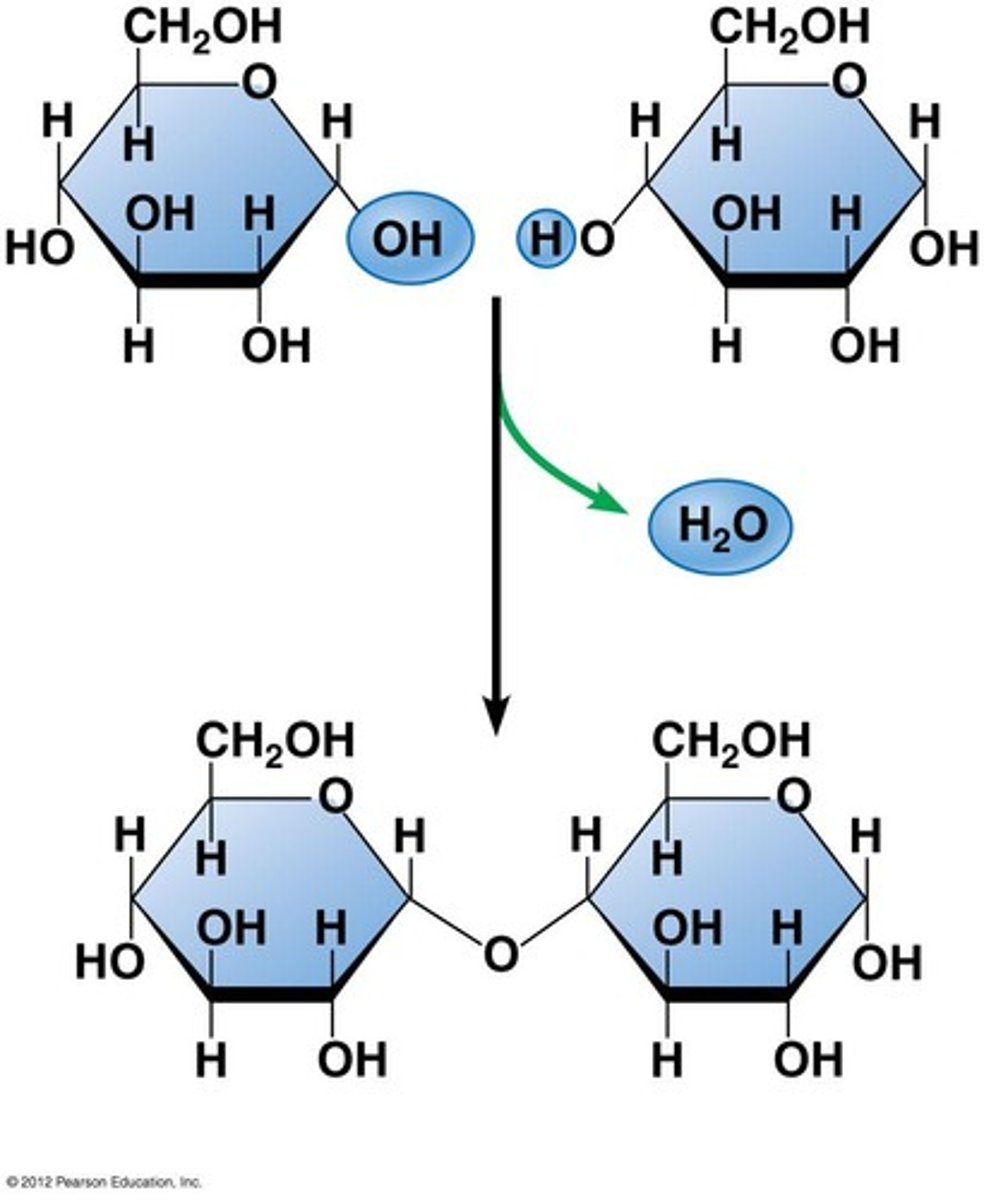
What is a condensation reaction?
Water is REMOVED
● 2 molecules join together
● Forming a glycosidic bond
● Releasing a water molecule
What is a hydrolysis reaction?
Water is ADDED
● 2 molecules separated
● Breaking a glycosidic bond
● Using a water molecule
What is a glycosidic bond?
A covalent bond between 2 monosaccharide molecules, resulting from a condensation reaction.
Draw a condensation reaction.
What is the general rule for solubility?
Like dissolves like.
(i.e, polar dissolves polar
non-polar dissolves non-polar)
During which processes are polymers hydrolysed in the body into monomers?
● Digestion: e.g. Starch (large & insoluble) hydrolysed (broken down) into glucose
● Glycogenolysis: Stores of glycogen hydrolysed back into glucose when blood sugar levels drop.
Catalysed by enzymes.
Describe the basic function of starch
● Found in many parts of a plant in the form of small grains.
● Especially large amounts occur in seeds and storage organs, such as potato tubers.
● It forms an important component of food and is the major energy source in most diets.
Describe the basic structure of starch
● Polysaccharide of α-glucose
● Formed from 2 polymers of alpha glucose: Amylose 1,4-glycosidic bonds → unbranched
● Amylopectin 1,4- and 1,6-glycosidic bonds → branched
Where is starch found?
Starch is found in starch grains inside plant cells e.g. inside chloroplasts.
Explain how the structure of starch relates to it's function.
● Long, straight, unbranched chain of amylose coils up to form a helix → compact for storage in cell
● Large, insoluble polysaccharide molecule → can't leave cell / cross cell membrane
● Insoluble in water → water potential of cell not affected (no osmotic effect)
● Branched → many ends for faster hydrolysis
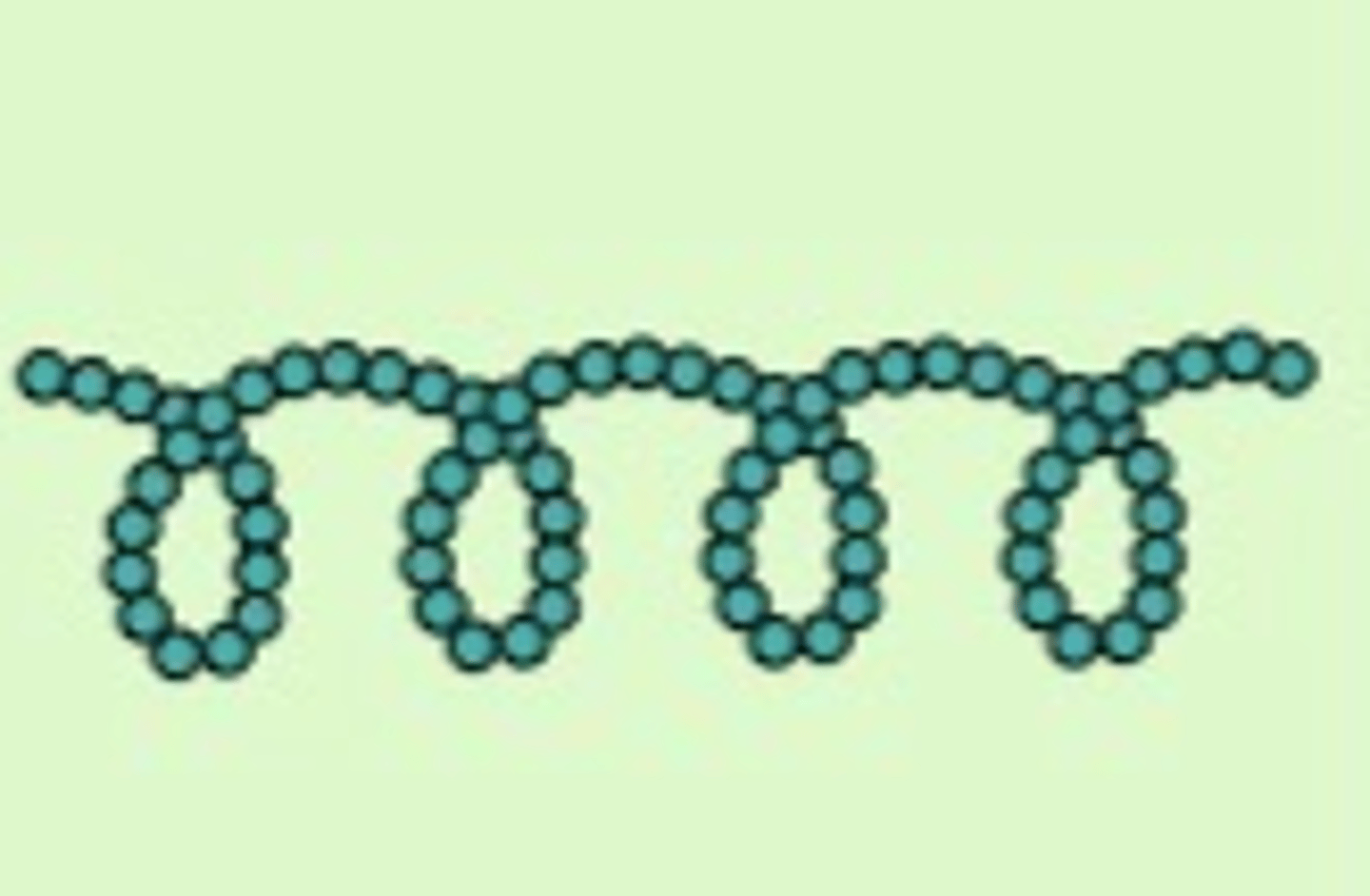
Describe the structure of amylose.
● Created by condensation reactions
● 1,4 glycosidic bonds
● Unbranched, coils up to form a helix
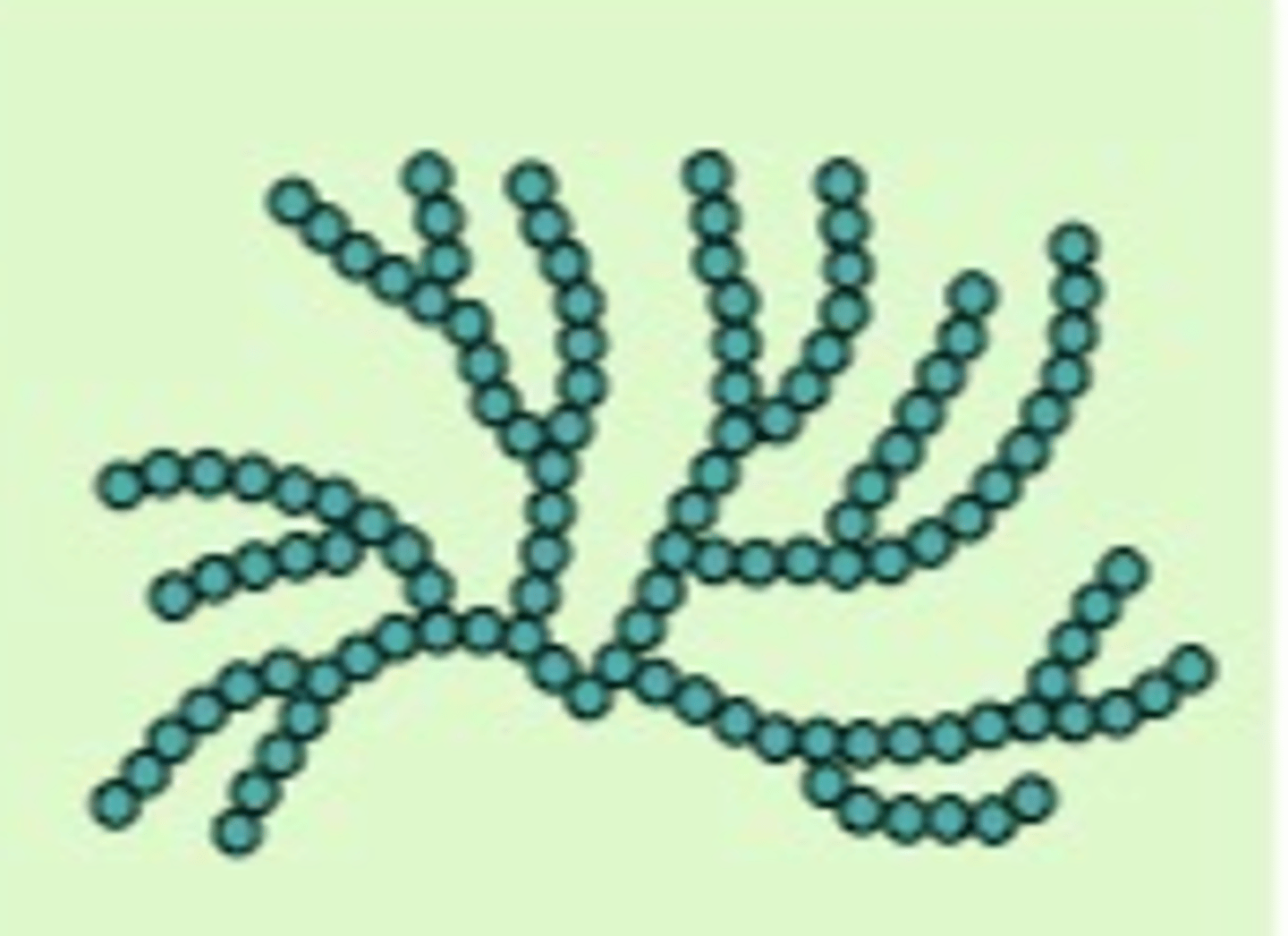
Describe the structure of amylopectin.
● Formed by condensation reactions
● Has 1,4 glycosidic bonds (straight) and 1,6 glycosidic bonds which creates branches
● Many ends for faster hydrolysis
Describe the basic structure of glycogen
● Polysaccharide made of α-glucose
● 1,4- and 1,6-glycosidic bonds →
● Highly branched (more than starch)
Describe the basic function of glycogen
Function: An insoluble store of glucose for animals
Where is glycogen found?
Muscle and liver cells
Glycogen fun fact!
We store an average of 500g of glycogen at one time, around the size/weight of a loaf of bread. So, needs to be compact to fit it all, so is coiled.
Explain how the structure of glycogen relates to it's function
● Branched → compact / fit more molecules in small area
● Highly branched → more ends for enzymes to attach to, faster hydrolysis → release glucose for respiration to make ATP for energy release
● Large, insoluble polysaccharide molecule → can't leave cell / cross cell membrane
● Insoluble in water so don't dissolve→ water potential of cell not affected (no osmotic effect. Won't cause water to move into cell)
What features do starch and glycogen share?
● Compact, with a coiled structure.
● Unable to cross cell membranes, as are large molecules.
● Do not affect cell water potential, as are insoluble.
● Many 'ends' for hydrolysing enzymes to attach to as a result of a highly branched structure.
Describe the basic function of cellulose
● Provides strength and structural support to plant cell walls
● Prevents cell from bursting when it becomes turgid
● Cellulose fibres are freely permeable which allows water and solutes to leave or reach the cell surface membrane
Describe the basic structure of cellulose
● Polysaccharide of β-glucose, formed from condensation reactions
● Only 1,4-glycosidic bond → long, straight, unbranched chains
● Chains linked in parallel by hydrogen bonds forming microfibrils. Number of hydrogen bonds means cell wall is strong.
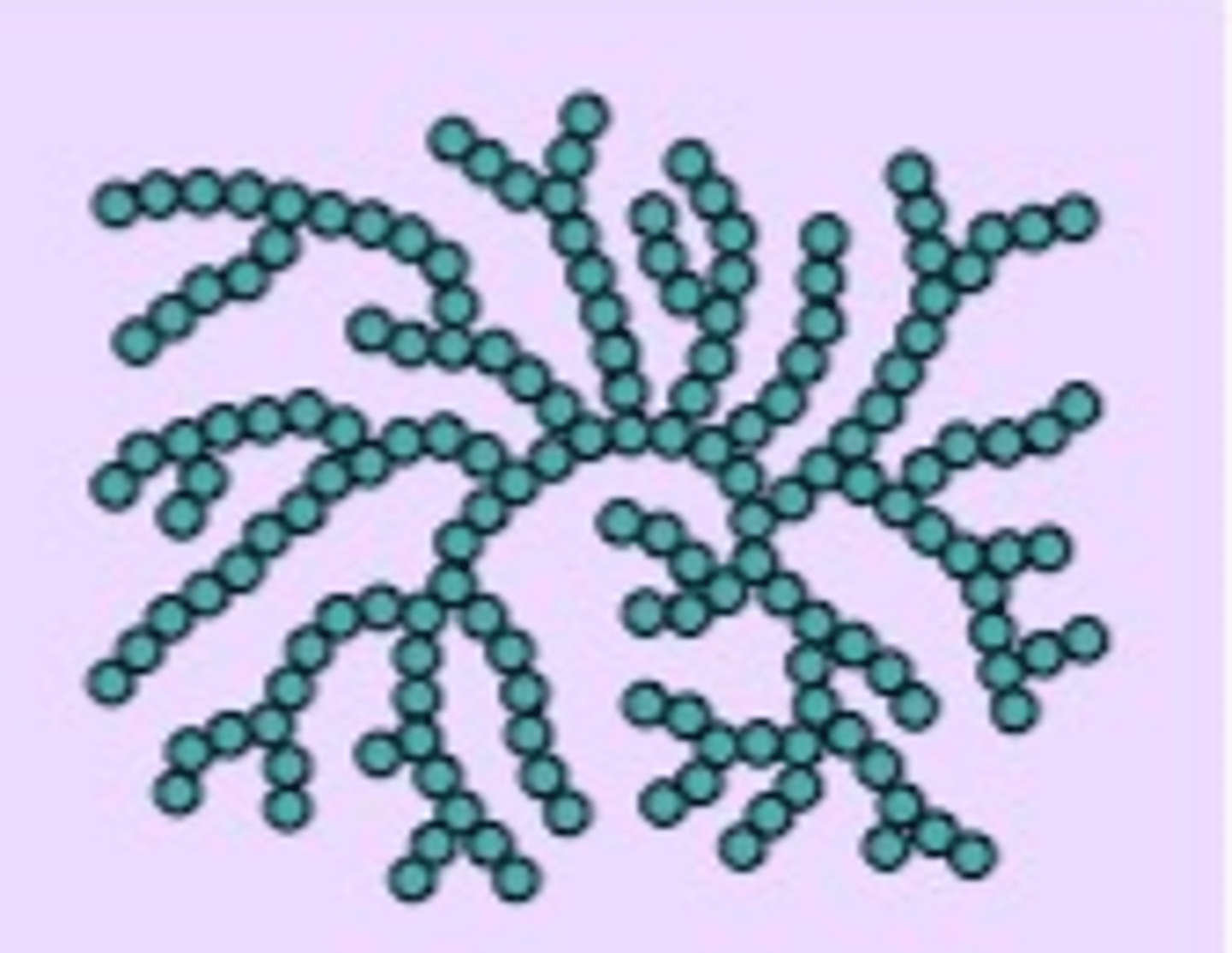
Explain how the structure of cellulose relates to its function
● Every other β-glucose molecule is inverted in a long, straight, unbranched chain
● Many hydrogen bonds link parallel strands (crosslinks) to form microfibrils (strong fibres)
● Hydrogen bonds are strong in high numbers
● So provides strength to plant cell walls
How are cellulose chains structured?
● Made of long, straight, parallel chains, with hydrogen bonds between them
● These chains are grouped into threads called microfibrils
● These are further grouped into macrofibrils
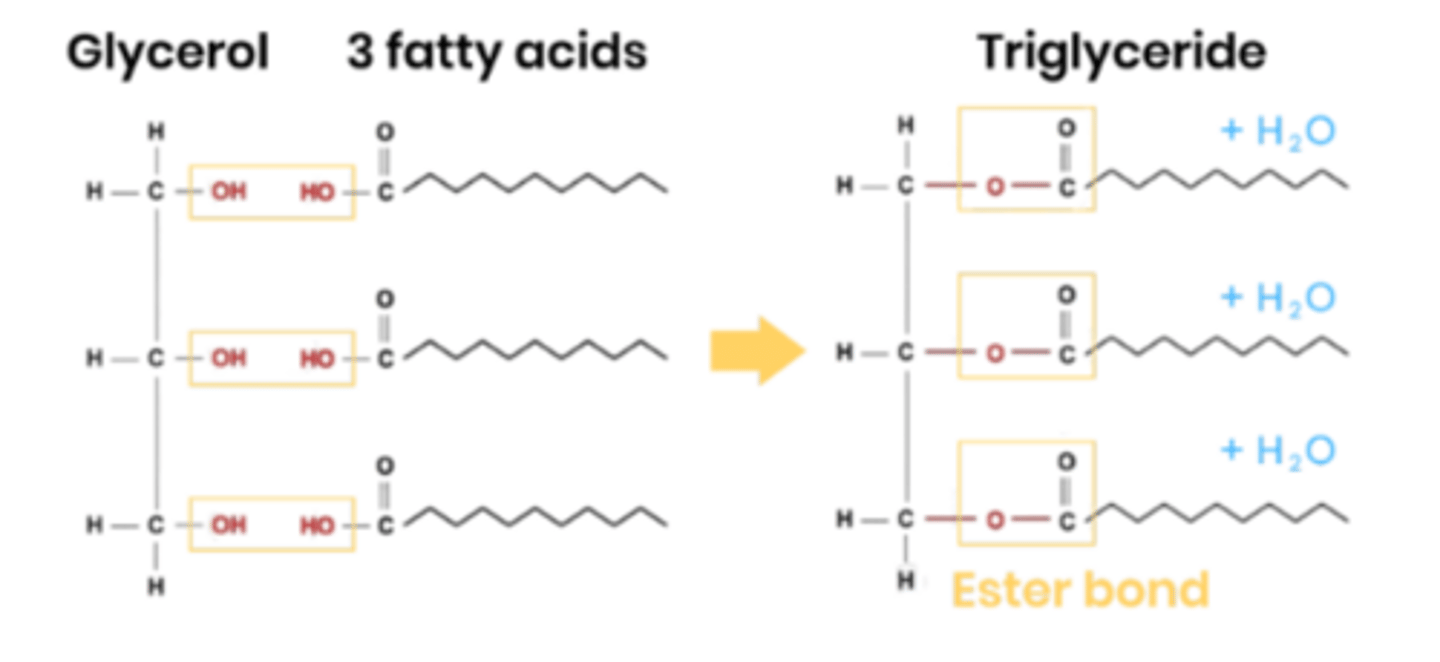
Describe the structure of triglycerides.
● 1 molecule of glycerol attached to 3 fatty acids
● Fatty acids made of hydrocarbons
● The tails are hydrophobic so makes triglycerides insoluble
● Non-polar (carboxyl group is polar, the hydrocarbon chain of fatty acids is non-polar.)
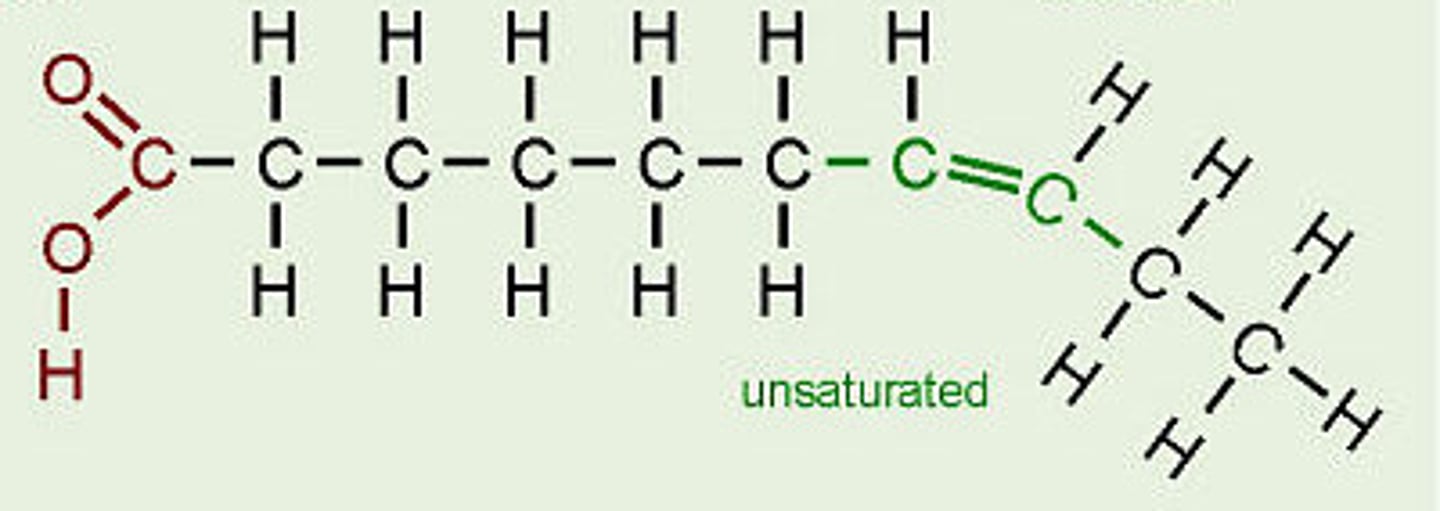
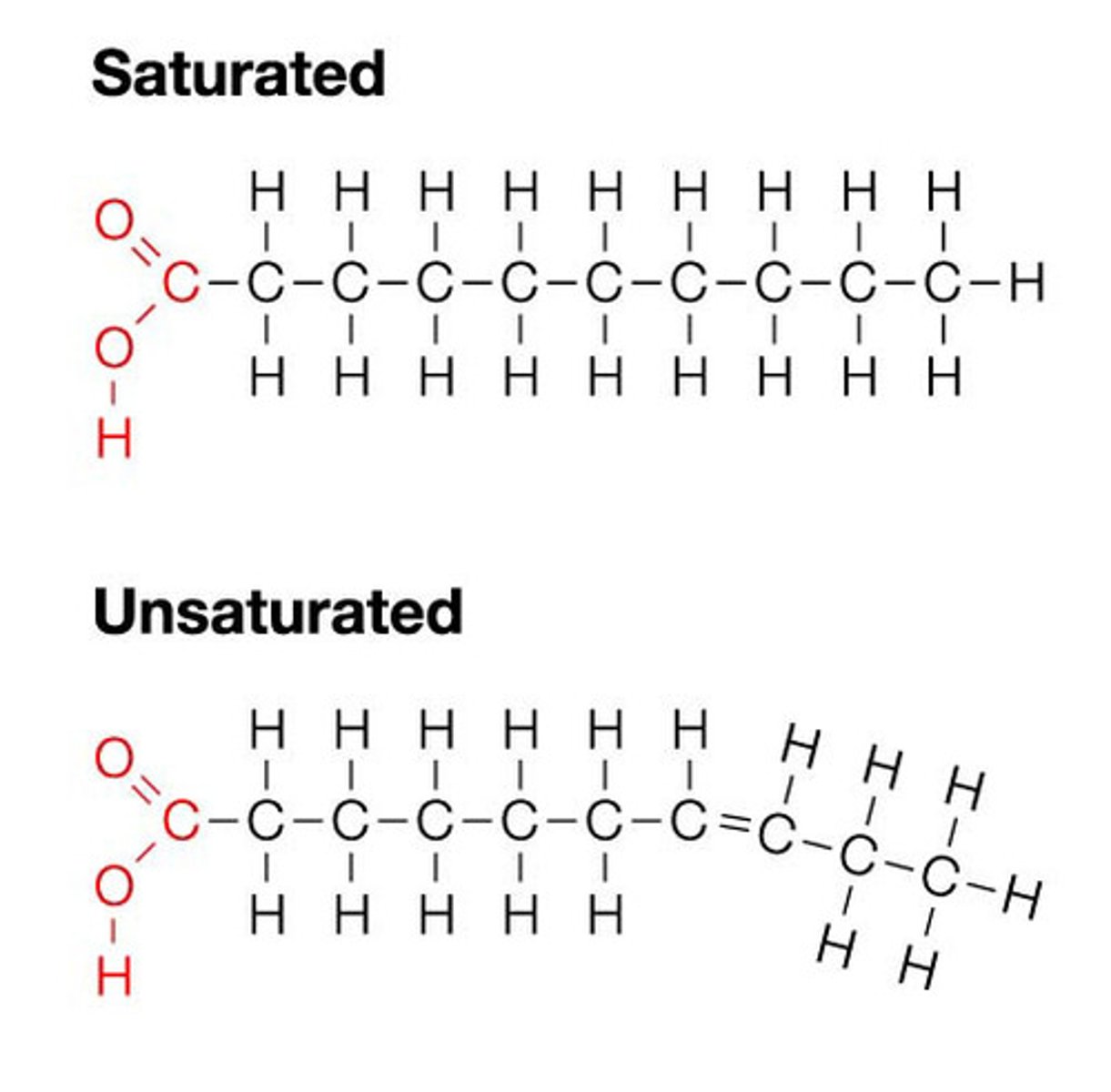
Describe the difference between saturated and unsaturated fatty acids.
● Saturated - no C=C double bonds in hydrocarbon chain → all carbons fully saturated with hydrogen
● Unsaturated - one or more C=C double bond in hydrocarbon chain (creating a bend / kink)
(MUST specify carbon-carbon!!)
How are triglycerides formed?
● Condensation reaction between 1 glycerol and 3 fatty acids
● H from OH group on glycerol joins with OH group on the COOH, to release a water molecule, forms an ester bond. Repeat for other 2 fatty acids.
● 3 condensation reactions, 3 ester bonds
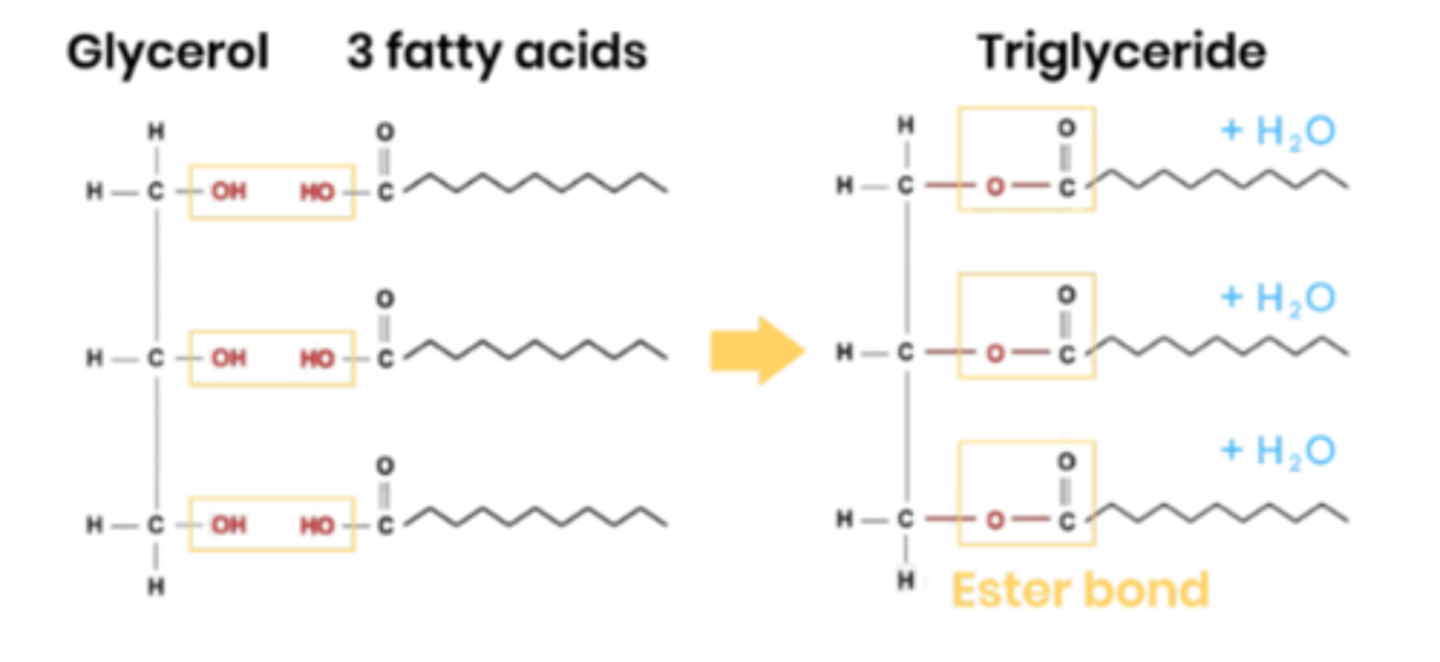
Name the reaction when 1 glycerol and 3 fatty acids join together.
Name the bonds formed during this reaction.
How many water molecules are formed?
● Condensation reaction
● Ester bonds
● 3 water molecules
What is the R group?
The R group is the hydrocarbon chain of fatty acids, can be saturated or unsaturated
(The variable group - the number of carbons and hydrogens can vary)
What is a common abreviations of fatty acids?
RCOOH
What are saturated fatty acids?
The hydrocarbon chain has only single bonds between carbons (bonding with as many hydrogen bonds as possible).
What are unsaturated fatty acids?
● The hydrocarbon chain consists of at least 1 double bond between carbons (not holding the maximum number of hydrogen atoms).
● Causing the chain to kink
How does the presence of a double bond in an unsaturated fatty acid alter how the chain looks?
Double bond causes the chain to bend.
Contrast saturated and unsaturated fatty acids.
Saturated:
● contain only single bonds
● straight-chain molecules have many contact points
● higher melting point = solid at room temperature
● found in animal fats
Unsaturated:
● contain C=C double bonds
● 'kinked' molecules have fewer contact points
● lower melting point = liquid at room temperature
● found in plant oils
Explain how the properties of a triglyceride are suited to its function as an energy source.
Triglycerides have fatty acid tails which have a high ratio of C+H to O, so they can release high amounts of energy via respiration to release large amounts of ATP.
Relate the structure of triglycerides to their functions.
● High energy:mass ratio = high calorific value from oxidation (energy storage).
● Insoluble hydrocarbon chain = no effect on water potential of cells & used for waterproofing.
● Slow conductor of heat = thermal insulation e.g. adipose tissue.
● Less dense than water = buoyancy of aquatic animals.
What are monounsaturated and polyunsaturated fatty acids?
Monounsaturated: Contains ONE C=C double bond
Polyunsaturated: Contains MANY C=C double bonds
True or false? Lipids are polymers.
False.
● Carbohydrates, proteins and nucleic acids are polymers.
● Lipids are not made up of repeating units of monomers.
Adding fatty acids into a solution causes the pH of that solution to decrease. Suggest why.
Fatty acids are acids.
A higher concentration of acid in a solution causes the pH to decrease (become more acidic)
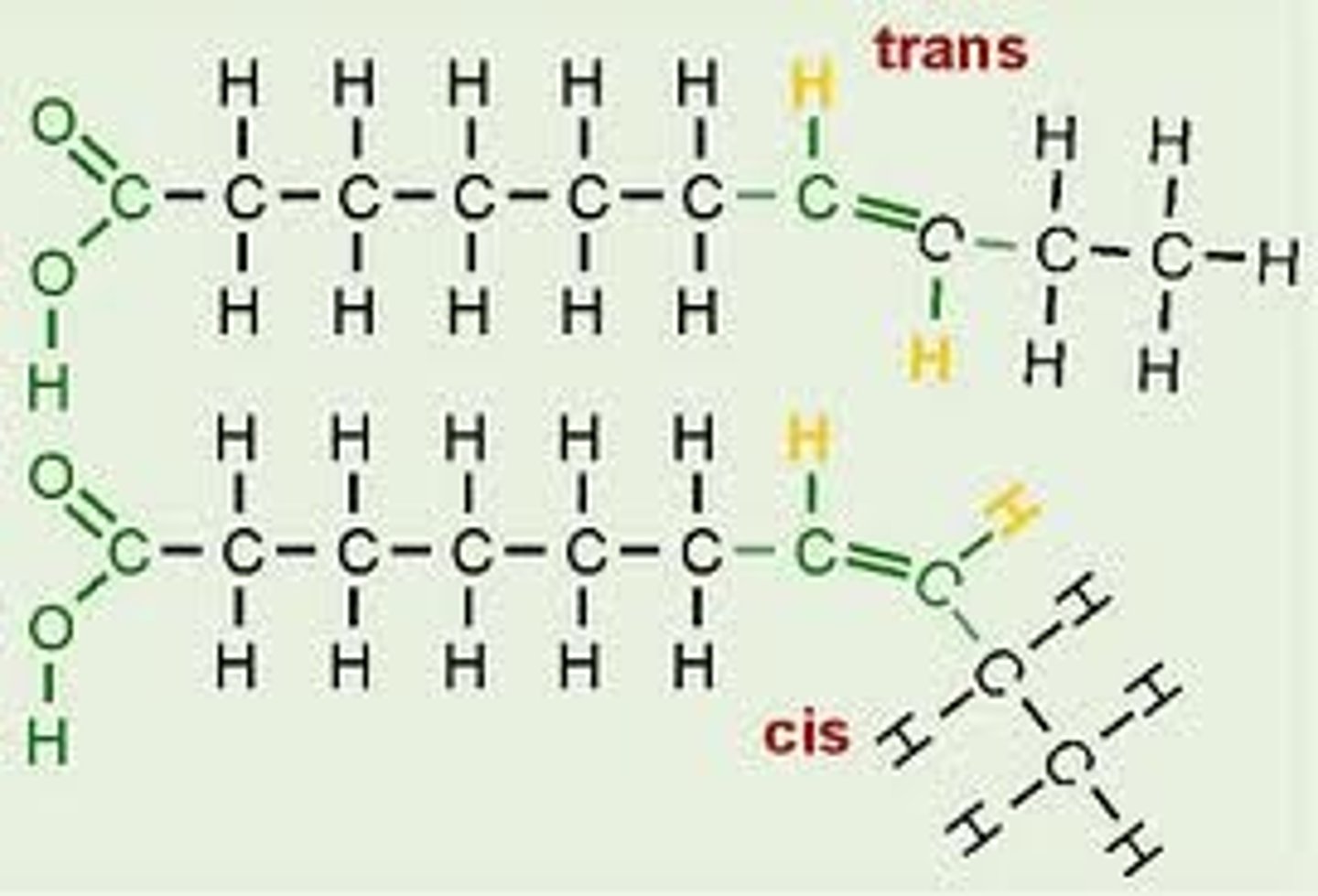
What is the difference between cis and trans fatty acids?
● If on double bond the H are on the same side it is cis
● if the H's are on opposite sides it is a trans unsaturated fatty acid.
What is the main function of triglycerides?
Energy storage
● High ratio of C-H bonds to carbon atoms in hydrocarbon chain
● So used in respiration to release more energy than the same mass of carbohydrates
● Hydrophobic / non-polar fatty acids so insoluble in water (clump together as droplets, tails inwards)
● So no effect on ψ of cell (or can be used for waterproofing)
a) Compared to carbohydrates, fatty acids have a...
b) Because of their long hydrocarbon chains, we say that fatty acids are...
a) Consistently high ratio of C + H to O
b) large, non-polar
Give the properties of triglycerides.
1. Large
2. Non-polar
3.Insoluble in water → hydrophobic
4. High ratio of C+H to O atoms
Explain the properties of triglycerides related to their structure.
● High ratio of C+H to O atoms → water source via respiration, energy source via respiration, storage molecule
● Hydrophobic, doesn't affect water potential and osmosis. Insoluble in water.
● Large → can't cross cell membrane and leave cell.
● Low mass:energy ratio → good storage molecule.
Why are fats (such as butter) and oils different although they are both lipids?
● Fats, such as butter, are made up of saturated fatty acids with straight, parallel hydrocarbon chains.
● So triglycerides with saturated fatty acids don't take up a lot of space and can be packed tightly together, forming a solid block at cool temperatures.
● Oils, such as vegetable oil, are made up of unsaturated fatty acids with bends in their hydrocarbon chains.
● So triglycerides with unsaturated fatty acids do take up a lot of space and can't be packed tightly together, forming a liquid at cool temperatures.
Explain how storage of fat in the hump of a camel can help it to survive in the desert.
● Triglycerides used to produce ATP during respiration, and also release water.
● So lots of ATP and water during respiration, since fatty acids have a high C+H:O ratio.
● The large amounts of water produced will enable camel to survive in desert, where water scarce.
● The large amounts of ATP produced useful, provides energy for essential functions even where food is scarce.
● Good storage molecule, have a low mass : energy ratio so are a good storage molecule, so a high amount of energy stored in a relatively small amount of mass, camel not weighed down and can roam easily.
What is the emulsion test for lipids?
● Dissolve the sample in ethanol
● Add distilled water, shake
● If a white emulsion appears, a lipid is formed.
State 2 functions of lipids. For each function, explain one feature of the lipid that makes it suitable for that function.
1. Energy store: High energy value / compact / insoluble / minimises mass
2. (Thermal) insulation: Poor heat conductor. Minimises heat loss from body.
3. Waterproofing: Non-polar / hydrophobic / insoluble / repels water
4. Buoyancy: Less dense than water
5. Protection / mechanical insulation: Flexible / protective layer around organs / helps cushion organs from external forces
6. Electrical insulation: Non-polar / hydrophobic fatty acids
What is the structure of phospholipids?
● Glycerol (Backbone) + 2 Fatty Acids (as tails) + Phosphate Group
● The 2 fatty acids bond to the glycerol via 2 condensation reactions, resulting in 2 ester bonds.
● Fatty acid tails = non polar, hydrophobic, insoluble in water
● Negatively charged head is polar, hydrophilic, soluble
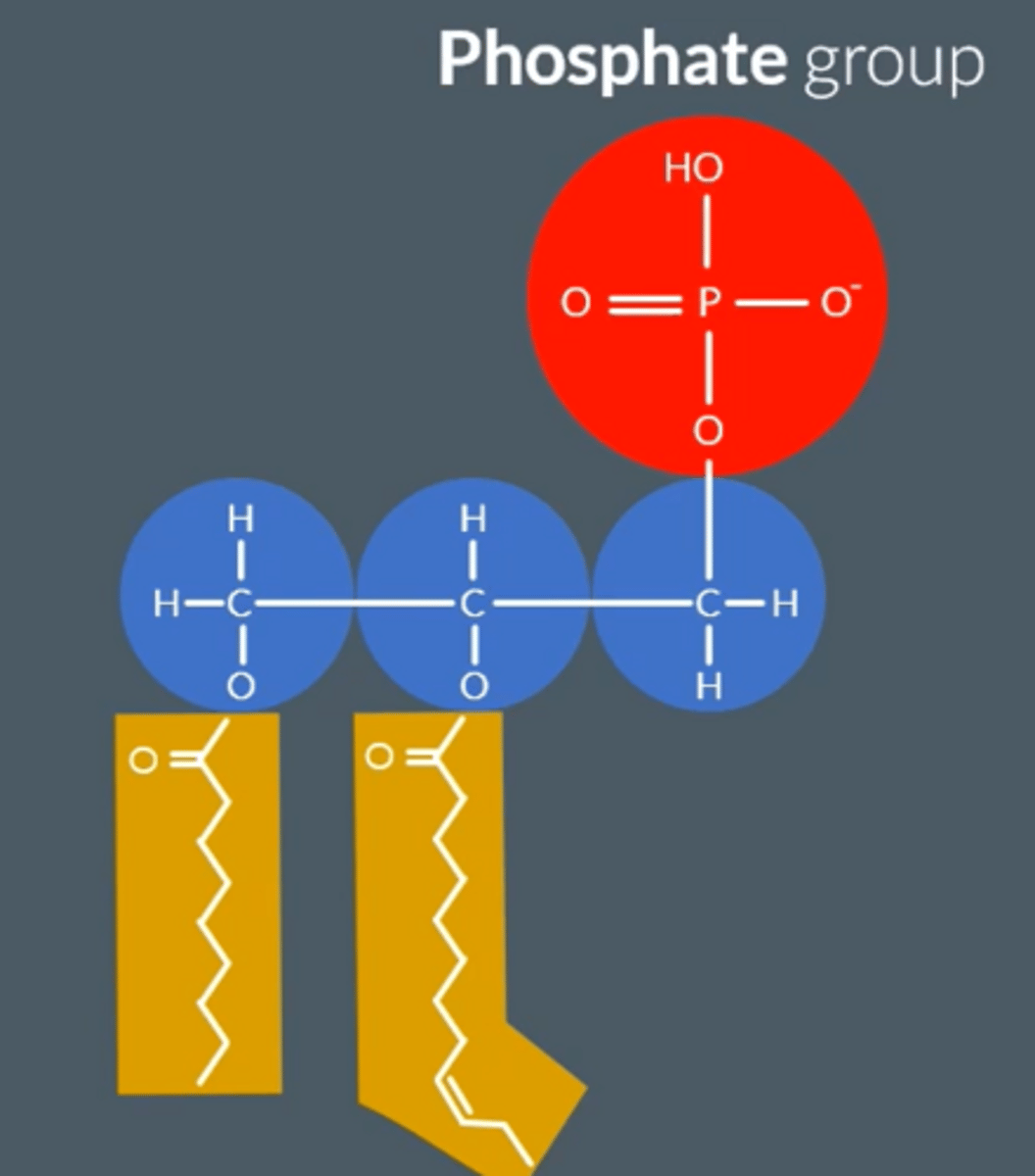
Describe the properties of phospholipids. (The head + tail)
● Fatty acid tails = hydrophobic (repels water), non-polar, insoluble in water
● Negatively charged phosphate head = hydrophilic (attracts to water), polar, soluble
● Amphipathic - both hydrophobic and hydrophilic regions.
Describe the properties of phospholipids.
● Have 2 charged regions, so they are polar
● In water they are positioned so that the heads are exposed to water, and the tails are not
● This forms a phospholipid bilayer membrane structure which makes up the plasma membrane around cells.
How do phospholipids form?
● Condensation reactions between glycerol and phosphate group forming phosphate ester bond
● and reaction between glycerol and fatty acids forming an ester bond, releasing water molecule per bond.
More properties of phospholipids
● Bilayer - hydrophilic heads attract water, tails repel, tails inwards shielded, heads outwards.
● Barrier - in cell membrane to water soluble molecules, ions, charged/polar molecules.
● Electrical insulator - ions can't enter as they are charged and repel fatty acid hydrophobic tails.
● Stability/fluidity - saturated fatty acids less fluid, can move past each other to keep membrane fluid to change shape and move but never expose hydrophobic fatty acid tails.
Describe the structure of fatty acids.
● All consist of a carboxyl group (COOH) and a hydrocarbon tail
● which can vary (R)
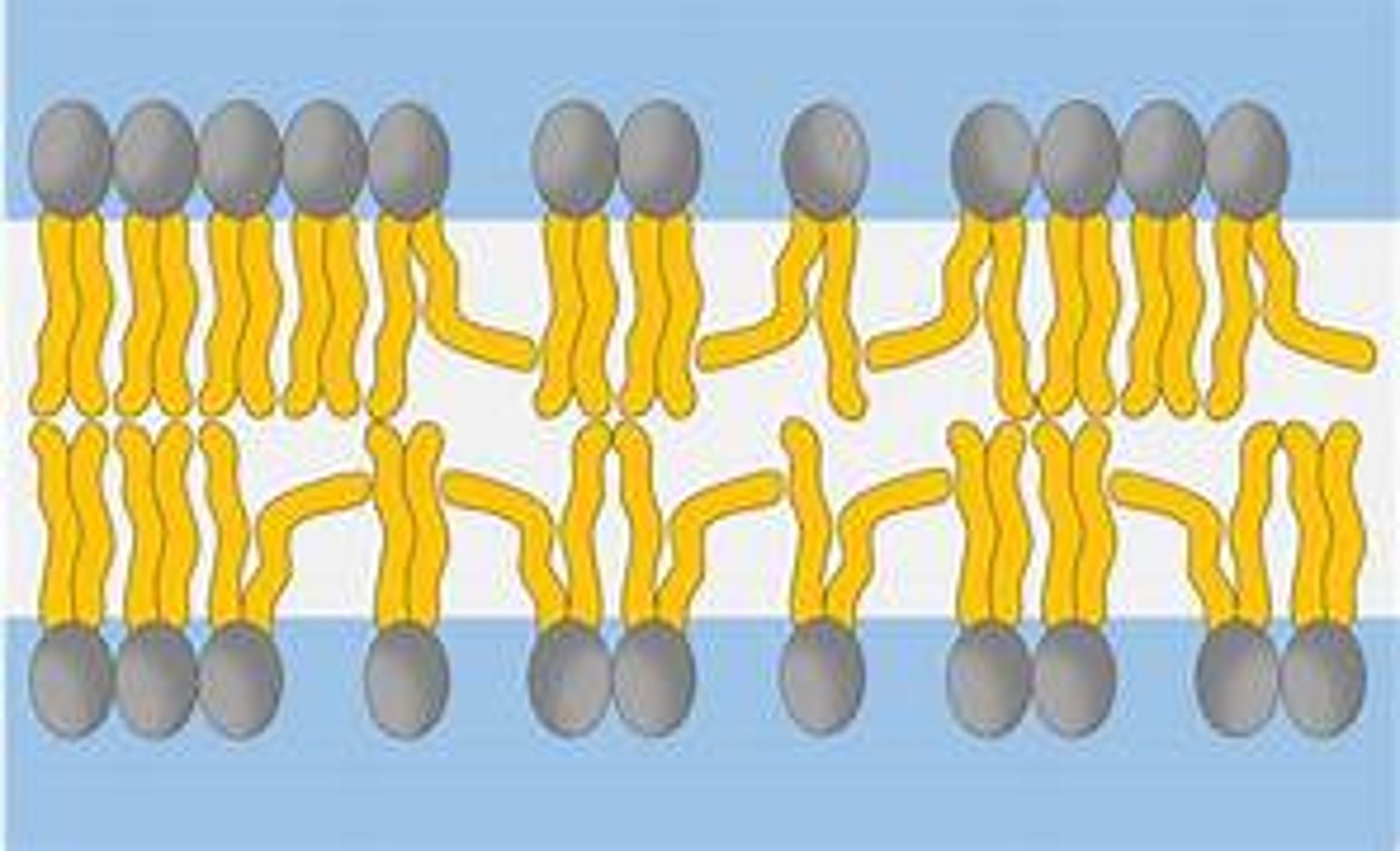
Name the 3 structures that phospholipids can form in water.
micelle, monolayer, bilayer
Describe how the properties of phospholipids relate to their structure.
Function: form a bilayer in cell membrane, allowing diffusion of lipid-soluble (non-polar) / small substances and restricting movement of water-soluble (polar) or larger substances
● Phosphate heads are hydrophilic
○ Attracted to water so point to water (aqueous environment) either side of membrane
● Fatty acid tails are hydrophobic
○ Repelled by water so point away from water / to interior of membrane
Why are O₂ and CO₂ able to enter and exit cells through the phospholipid bilayer?
Unsaturated fatty acids in the phospholipids create gaps which make the cell-surface membrane flexible and allow some small molecules like CO₂ and O₂ to pass through.
What are glycolipids and why are they important?
● Formed when sugars / carbohydrates bind to a phospholipid on the bilayer.
● Important for cell recognition as allow cells to be recognised as body's own cells.
Are triglycerides and phospholipids soluble in alcohol?
Yes.
Alcohols such as ethanol have both a non-polar and a polar region, so ethanol is able to mix with non-polar and polar substances.
What is the structure of an amino acid?
NH₂= amine group
COOH = carboxyl group
R = variable group
20 amino acids, only vary in R group
Glycine - H in R group
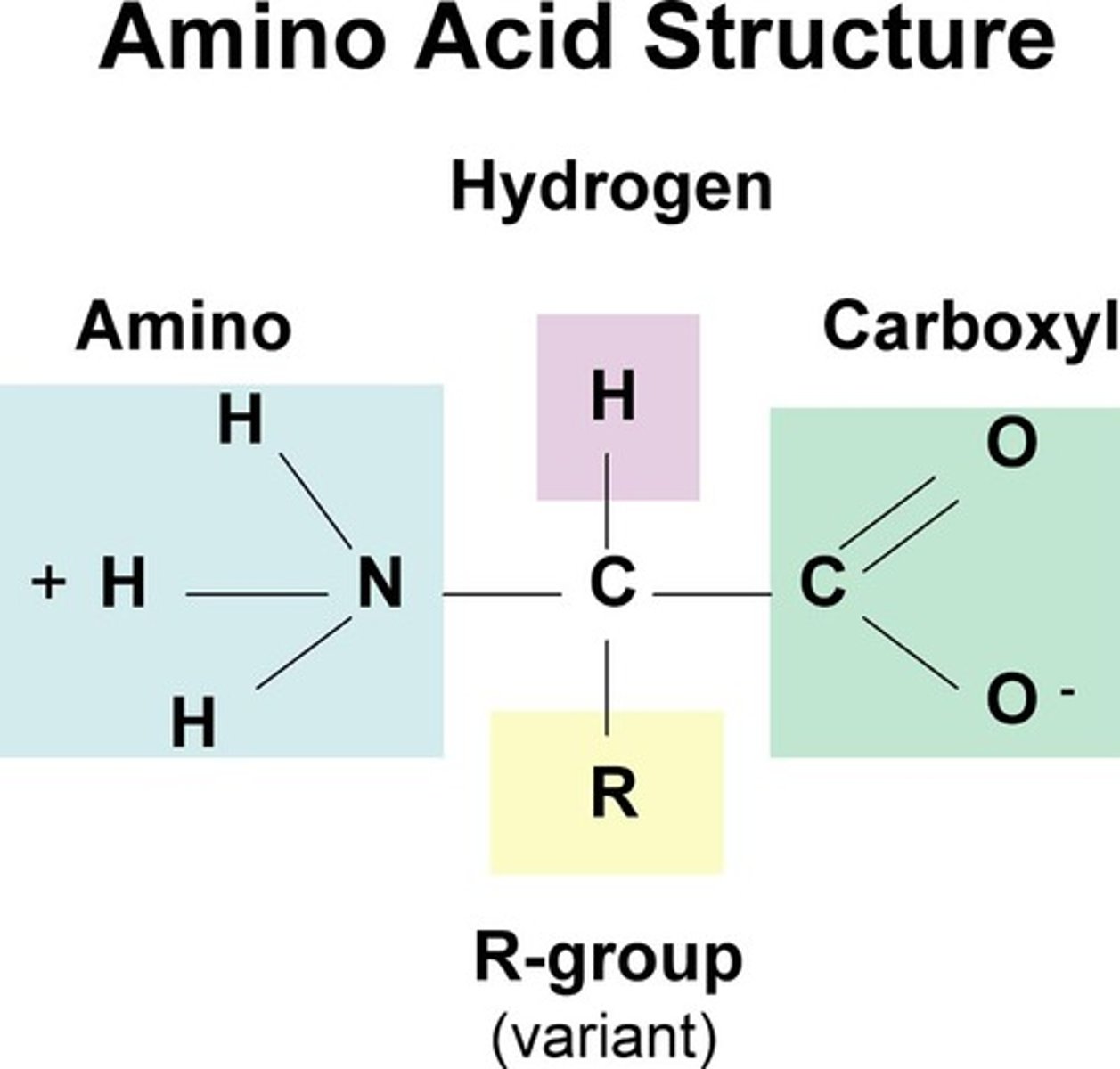
R group in amino acids
● The variable group
● 20 amino acids, only vary in R group
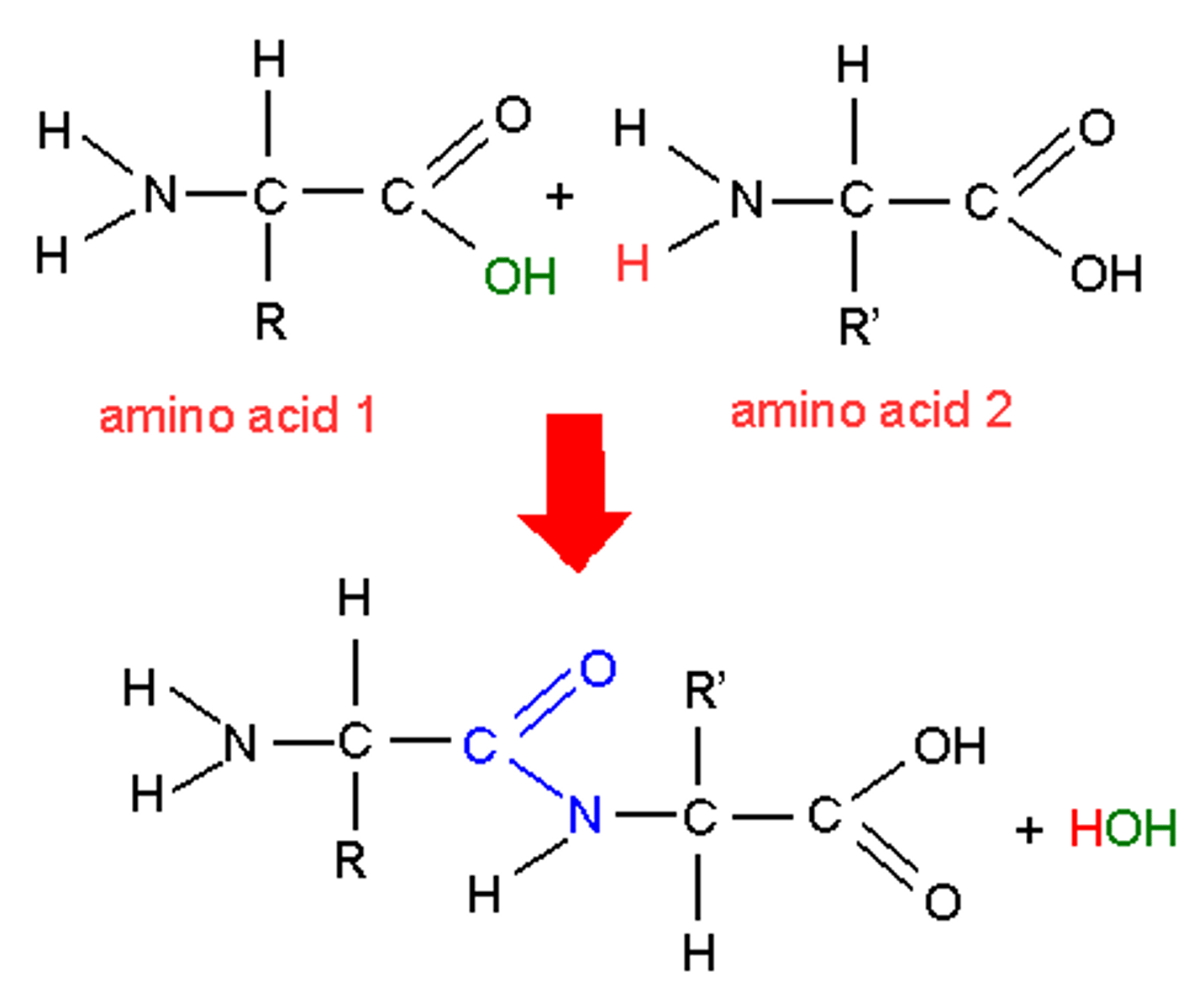
How are dipeptides formed?
● Condensation reaction between OH on carboxyl group
and H on amine group
● Water is removed (released)
● Forming a peptide bond.
What is the difference between dipeptide and polypeptides?
2 amino acids - dipeptide
2+ amino acids - polypeptide
1 or more polypeptide chains is a protein.
How are dipeptides/polypeptides (polymer) converted into monomers?
Hydrolysis reaction where water is added, breaking the peptide bond.
Draw the dipeptide formed between 2 amino acids, and label the peptide bond.
Primary structure of proteins
The sequence of amino acids in a polypeptide chain, joined by peptide bonds - this is a polymer
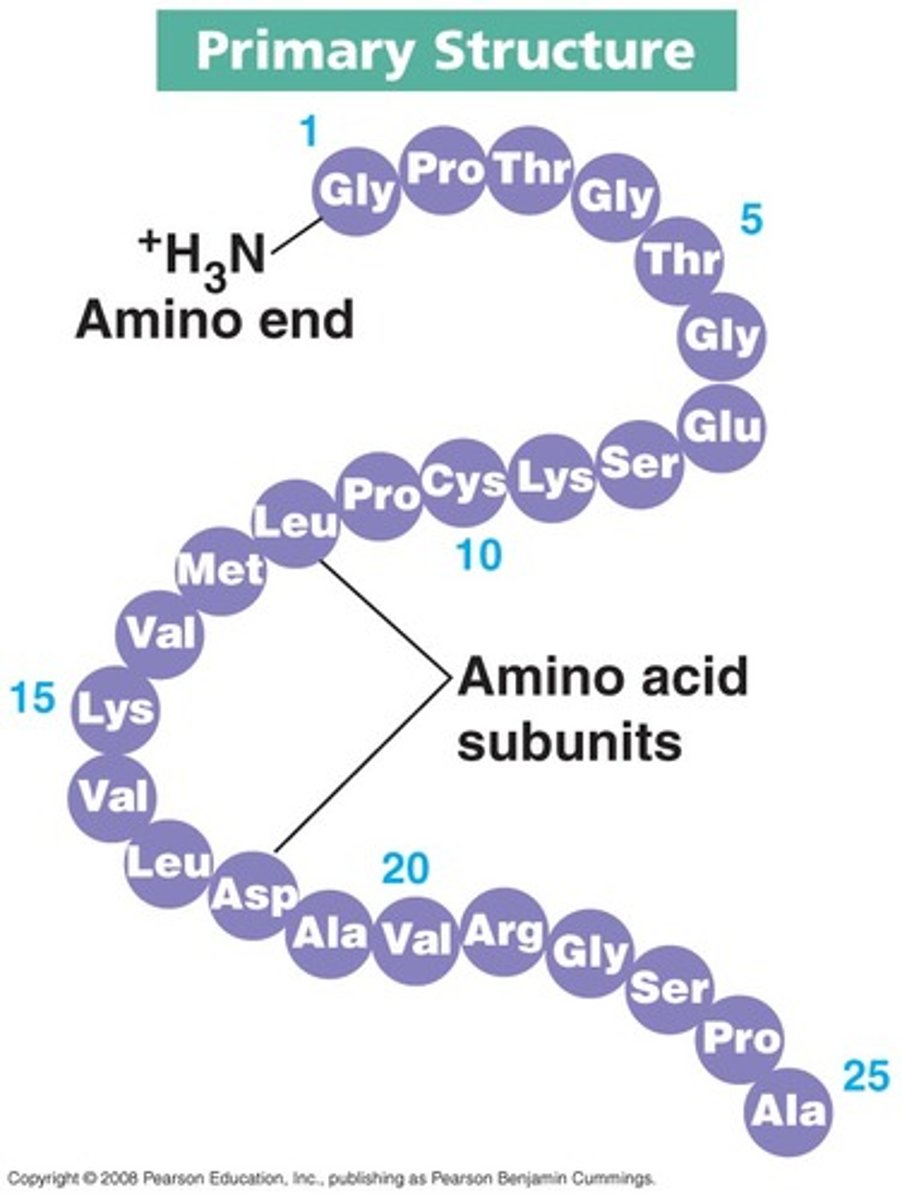
What is special about the primary structure?
The primary structure is specific for each protein (one alteration in the sequence of amino acids can affect the function of the protein)
What determines the primary structure of a protein?
● DNA of a cell; instructs the cell to add certain amino acids in specific quantities in a certain sequence.
● This affects the shape and therefore the function of the protein
What is a polypeptide chain?
A sequence of amino acids linked together by peptide bonds, joined together by several condensation reactions.
What would happen if one of the amino acids coded for in the sequence is different?
● Causes the ionic/hydrogen/disulfide bonds to form in a different location
● results in a different 3d shape
● enzymes will have a different shaped active site (non-functioning)
● carrier proteins will have a different shaped binding site (molecules no longer complementary and cannot be transported across membranes)
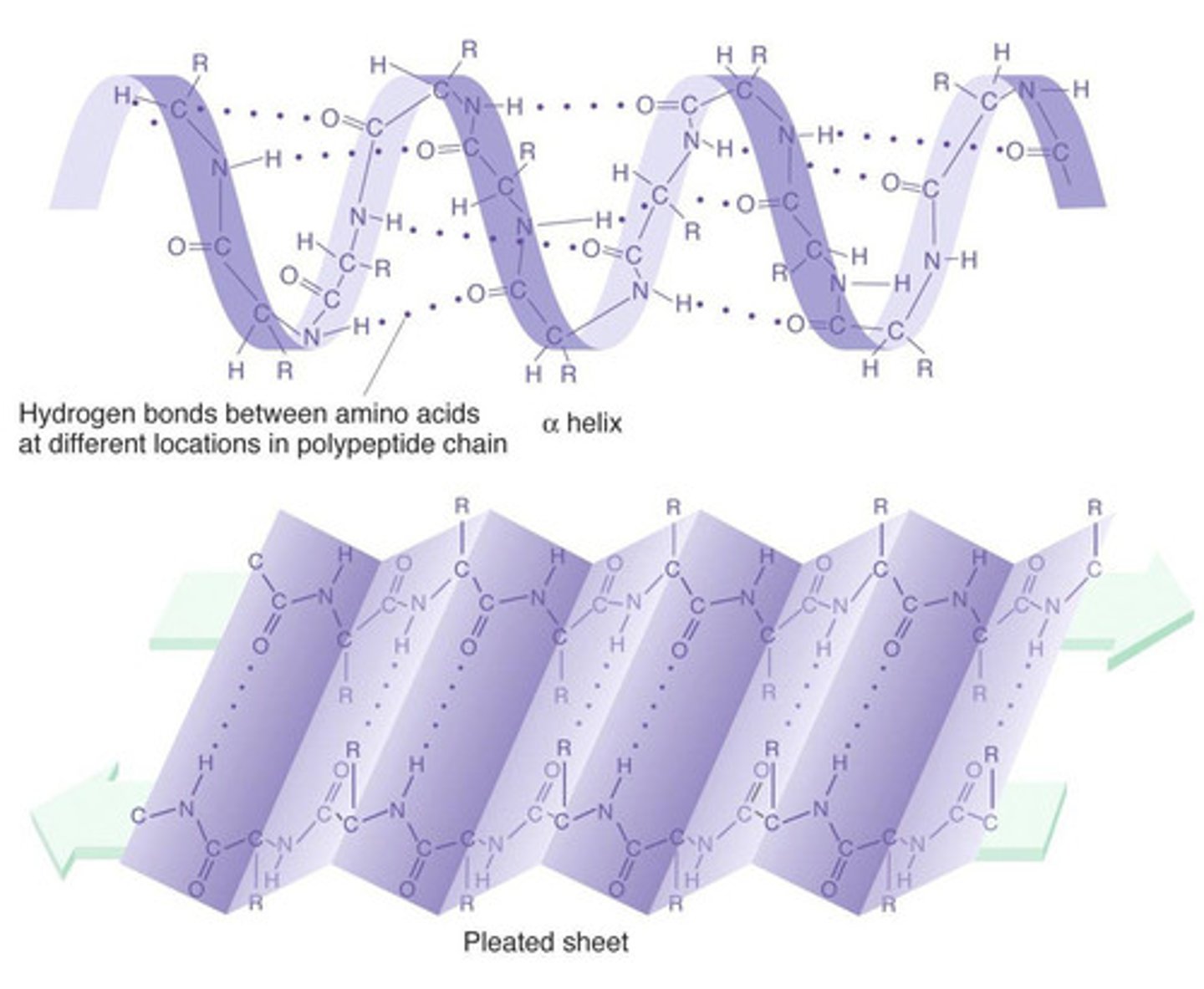
Secondary structure of proteins
● Simple folding (repeating patterns) of polypeptide chain e.g. alpha helix/beta pleated sheets
● Due to hydrogen bonding between amino acids
● Between NH (group of one amino acid) and C=O (group)
Why does the folding of the secondary structure happen?
Hydrogen bonds form between the groups of peptide bonds.
(From partial charges on H and O, so are polar)
Secondary structure of proteins
● Simple folded structure
● Held by hydrogen bonds
● That form between the amine group (NH) of one amino acid and the carboxyl group (C=O) of another
● Hydrogen bonds cause polypeptide chain to coil/twist
● Into alpha helix / beta pleated sheets
Where are the hydrogen bonds in secondary structures of proteins located?
Hydrogen bonds form between the C=O groups of the carboxyl group of one amino acid, and the H in the amine group of another amino acid
Tertiary structure of proteins
● Further folding of the secondary structure
● Due to interactions between amino acid R groups
● To form a unique 3d shape
● Held in place by ionic, hydrogen and disulphide bonds
Where do the bonds in tertiary structures form?
● Hydrogen bonds (between R groups with partial charges)
● Disulphide bridges (only occurs between cysteine amino acids)
● Ionic bonds (occurs between charged R groups).
● Hydrophobic interactions (between non-polar R groups)
The amino acids in 2 proteins are the same, but sequenced differently.
Explain why this might cause a change to the tertiary structure of the protein.
● The hydrogen, disulfide and ionic bonds could form in different locations.
● Meaning the polypeptide is pulled into a different 3D shape.
Quaternary structure
● Final 3D structure of proteins with more than 1 polypeptide chains.
● And a non-protein group.
● Formed by interactions between polypeptides (hydrogen bonds, ionic bonds, disulfide bridges)
Do all proteins have quaternary structure?
No. Only proteins with more than one polypeptide chain possess a quaternary structure.
E.g. haemoglobin, antibodies.
What is the non protein part of a quaternary structure called?
Prosthetic group → conjugated protein
Homodimer and heterodimer
Same polypeptide chains involved = homodimer
Different = heterodimer
What happens when a protein is denatured?
The bonds which hold the tertiary and secondary structure in shape break and therefore the unique 3d shape is lost (ie enzymes lose their unique active site shape)
Conditions that denature a protein
Too high temperature: too much kinetic energy
Too high/ low pH: too many H⁺ or OH⁻ gives imbalance in charge. Breaks the ionic & hydrogen bonds, changing the tertiary structure
How can a protein's structure be changed by changing the pH?
● Adding acid to the solution to make more protons (H+) available.
● Adding alkali to the solution would reduce the availability of protons.
Describe the test for proteins.
1. Add food sample to test tube. Add equal volume of sodium hydroxide solution
2. Add a few drops of copper sulfate solution to the test tube and mix gently.
3. A positive test observation = solutions from blue to purple
Describe the structure and function of globular proteins.
● Spherical & compact.
● Hydrophilic R groups face outwards & hydrophobic R groups face inwards = usually water-soluble.
● Involved in metabolic processes e.g. enzymes & haemoglobin.