AP Chemistry: UNIT 1 ATOMIC STRUCTURES AND PROPERTIES
1/66
Earn XP
Description and Tags
7~9% of the exam
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
67 Terms
Frequency
number of waves that pass by in a given amount of time
Relationship between wavelength and frequency?
Inversely proportional
“Quanta”
Discrete packets of energy
Equations relevant to wave calculations
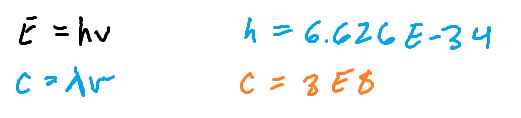
Higher frequency =
Higher KE (using ultraviolent light instead of yellow visible light)
Electrons can only be located in very specific _______________ around the nucleus
energy levels
Electrons cannot exist in _____________ energy levels
BETWEEN
Heisenberg’s Uncertainty Principle
Can never be certain about an electron’s location and momentum simultaneously
Explain what causes green light vs. red light to be emitted according to Bohr’s model

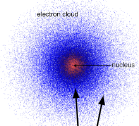
What do the dots in the electron cloud model represent
likelihood of finding electrons in surrounding parameter/at any given spot
s orbitals appear as…
spheres
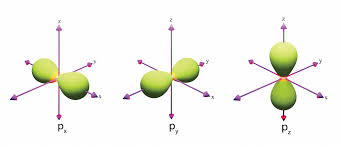
p orbitals look like…
3 dumbbells stacked together
Electron Configuration

Aufbau principle
In the ground state of an atom or ion, electrons fill subshells of the lowest available energy level first
Pauli’s Exclusion Principle
IN a given atom, no two electrons can occupy the same orbital WITH the same spin
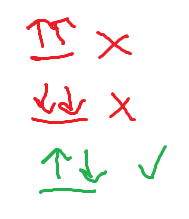
Hund’s rule
the lowest energy configuration for an atom is the one having a maximum number of unpaired electrons
For orbitals of same energy, add one electron per orbital before pairing

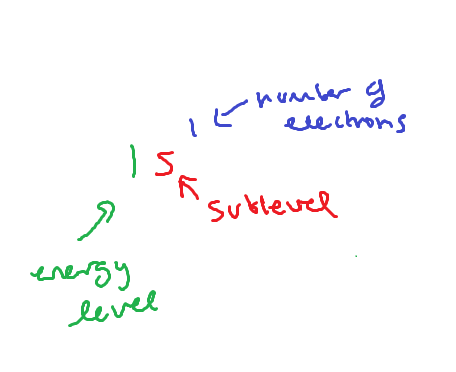
FORMAT FOR ELECTRON CONFIGURATIONS
Electron configuration of ions
figure out if the ion has lost or gained electrons
+ charge = LOST electrons
- charge = GAINED electrons
Isoelectronic
Two ions have the same number of electrons, therefore the same electron configuration
Why do some electron configurations no follow the normal pattern due to stability reasons?
ONLY occurs in large atoms, DUE TO CLOSE PROXIMITY OF SUBSHELLS, electrons move to increase stability
MOVE FROM S
Stability of all orbital types
Completely filled orbital- most stable
Half filled- less stable
Partially filled- least stable
ORDER OF FAVORABLE ELECTRON CONFIG.
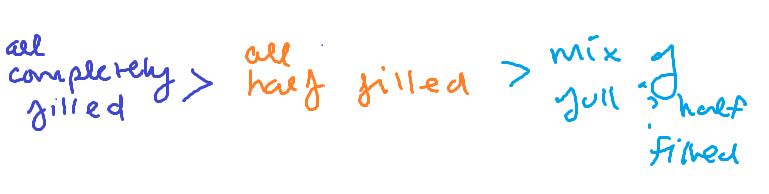
Paramagnetic
Attracted by a magnet because of unpaired electrons
Diamagnetic
Slightly repelled by magnets because all electrons are paired
Coulomb’s Law
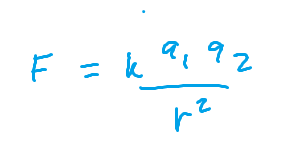
IONIZATION ENERGY IS
FORCE OF ELECTROSTATIC ATTRACTION
Ionization energy
the amount of energy required to remove an electron from an atom
IE1
1st ionization energy
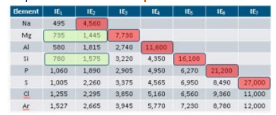
Red spaces on IE graph
Ionization energy of core electrons; MUCH harder to remove from an atom
What do the peaks in a PES graph represent?
Number of electrons
On PES graph, what does a BE closer to y axis mean? (use normal legend)
greater nuclear charge/sometimes more electrons
BIGGER GAPS IN PES GRAPH
greater electron repulsions
Shielding
Electrons closer to the nucleus “shield” some of the positive charge of the nucleus from the electrons further from the nucleus
Core electrons
all electrons inside valence shell
ZEFF
Effective nuclear charge = the effect of the nuclear charge on a specific electron
Nuclear Charge
Charge of nucleus
Zeff calculations
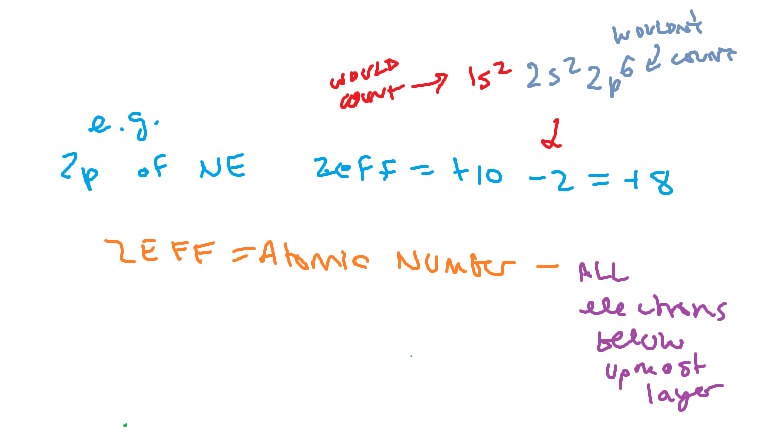
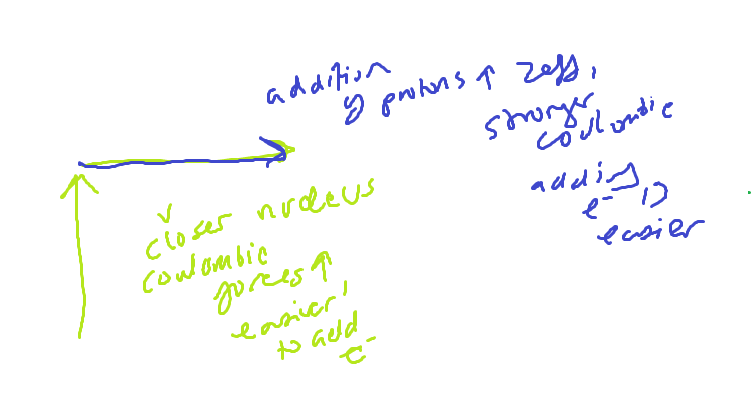
Atomic Size Trend AND explanation

Why are anions bigger than parent
More electron repulsions, valence electrons are tightly held by the nucleus causing the ion to increase in size
Size trend for isoelectronic ions
All have the same electron configuration, and therefore the same number of energy levels
LARGER AMOUNTS OF + CHARGE FROM THE NUCLEUS = LARGER COULOMBIC ATTRACTIVE FORCE, MAKING THE ION SMALLER
IE trend
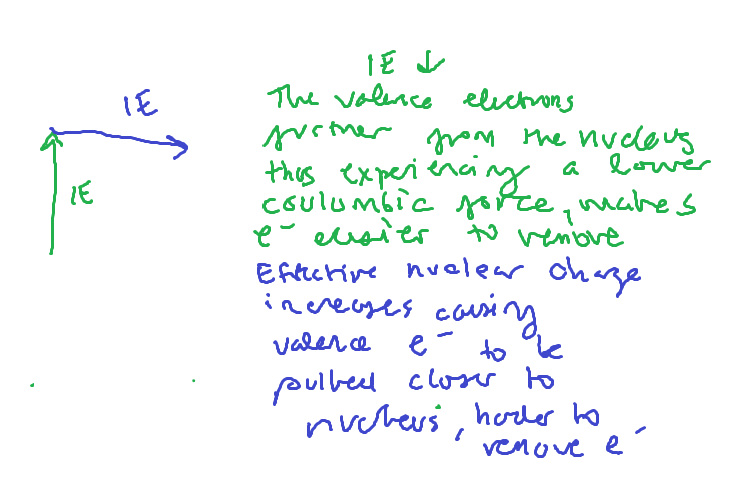
Be to B (IE)
AN electron gets added to the 2p orbital in B. This electron is slightly shielded by the 2s orbital making it easier to remove
N to O (IE)
O’s electron configuration ends in 2p4 There is repulsion between the paired electrons of the 2p orbital. This repulsion makes it easier to remove an electron
When does energy do a huge jump?
When all valence electrons have been removed
The first electron(s)______________ are from the _______ principal energy level
removed, HIGHEST
EA (Electron Affinity) trend
Electron affinity releases more energy as you go left to right
EXCEPTION: Noble Gases have a positive EA because it would require energy for them to gain an electron since this process is making the noble gas less stable, N ALSO HAS A + FOR SAME REASON
Electronegativity
Ability of an atom in a molecule to attract electrons to itself
Electronegativity trends
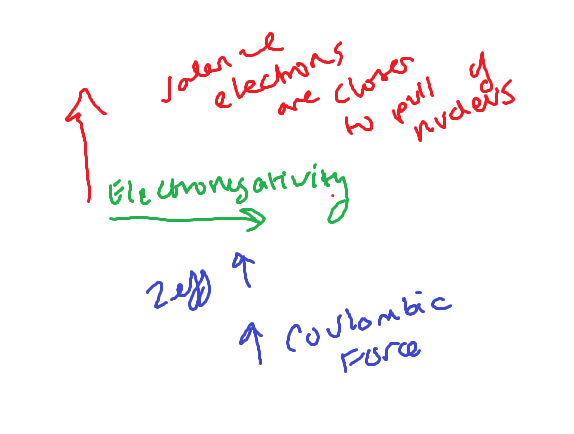
High electronegativity
High ionization energy, high negative electron affinity
Electron affinity
Energy change accompanying the addition of an electron
PROPOERTIES OF METAL
lustrous, malleable, ductile, good conductors of heat and electricity, form cations due to low ionization energy
NONMENTAL properties
dull, brittle, poor conductors of heat and electricity, can be solid, liquid, gas at room temp., form anions due to their large exothermic Electron Affinity
Metalloids
some characteristics of metals some of nonmetals, are semiconductors
Electrical Conductor how does it work
Electricity is the flow of electrons
-towards positive charge
-away from negative charge
IN CONDUCTORS, electrons will move away from their atom, conducting electricity
Insulators
do not allow electrons to flow because electrons that get excited REMAIN IN THEIR ATOMS
In conductors, electrons……
will move away from their atom and flow towards a positive charge thus conducting electricity
Metals form ____________ because they have __________, meaning its easy for them to lose electrons
cation, low ionization energy
Nonmetals have _____________ values meaning they will ___________________ to increase stability
larger exothermic electron affinity values, gain electrons
Metal reactivity trend
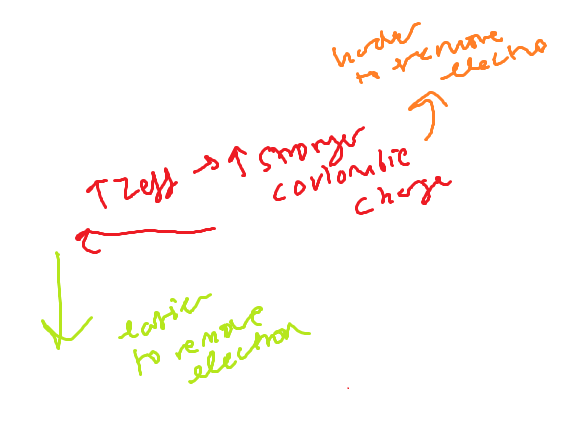
Nonmetal reactivity
Allotrope
existence of an element in two or more forms, which may differ in arrangement of atoms in crystalline solids
Why is IE of noble gases so high?
Atom form is so stable due to filled valence, largest effective nuclear charge = high IE, OVERALL UNREACTIVE
Spectroscopy
study of matter interacting with light
3 parts:
SOURCE OF LIGHT
SAMPLE OF MATTER
A DETECTOR
Spectrophotometry
Quantitative measure of transmitted or absorbed light, Specific to visible light and some areas of UV and IR
Mass Spec. can be used to separate…
atoms, isotopes, fragments of molecules BASED ON MASS
5 postulates of Dalton’s Atomic Theory
1) elements are made of extremely small particles called atoms
2)atoms of a given element are identical in size, mass, and other properties. Atoms of different elements DO differ in those properties
3)Atoms cannot be subdivided, created, or destroyed
4) Atoms of different elements combine in simple whole number ratios to form chemical compounds
5) In chemical reactions, atoms are combined, separated, or rearranged
Isotopes
atoms of the same element with different mass due to differing number of neutrons