Reducing Benzil Using Sodium Borohydride
1/13
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
14 Terms
oxidation occurs when
oxygen is added or hydrogen is removed
why is chromate CrO4²-, dichromate Ce2O7²-, and chromium trioxide CrO3 good oxidizers?
Because chromium is in the 6th oxidation state. Chromate and dichromate are strong so they can oxidize an alcohol straight to a carboxylic acid and a secondary alcohol to a ketone
Collins reagent
CrO3-pyridine2/CH2CL2 can oxidize a primary alcohol to an aldehyde and stop because there is no water
What happens to chromium oxidation state after an oxidation
the oxidation state reduces to 3
Why didn’t we use chromate, dichromate, or chromium trioxide?
All chromium reagents are toxic.
how does KMnO4 work for oxidation
In basic conditions it will turn primary alcohol to potassium carboxylate salts. Then with the addition of a strong acid the carboxylate will turn to a carboxylic acid. THe oxidation state is reduced from +7 to +4. It is not used with alcohols that contain triple or double bonds and not with secondary alcohols because they may react further.
what did we use for oxidation?
NaOCl (bleach) It will oxidize aromatic and aliphatic secondary alcohols to ketones and primary AROMATIC alcohols to aldehydes. It selectively oxidizes secondary alcohols. In the mechanism the Na will deprotonate the OH and then the Cl will add to the O. Then water will eliminate a hydrogen and cause the O to become a carbonyl and then the Cl will leave, but the mechanism is not very well known.
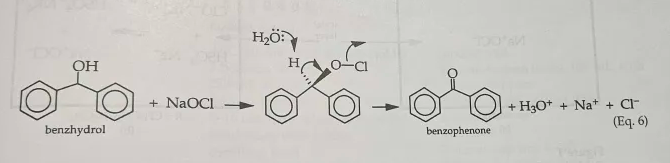
what did we use TBAS for (tetrabutyl-ammonium hydrogen sulfate) for
The alcohol is soluble in organic solvents and then NaOCl is soluble in water soluble solvents, so then they won’t mix and then they won’t react. The alkyl groups make it soluble in organic solvents and then at the same time it is also a salt so then it can shuttle between both layers. So then it will replace its HSO4 (hydrgoen sulfate) for the ClO-, which will then transfer to the ethyl acetate layer and then react with the alcohol. So it basically acts as a shuttle system.
ortho frequency range IR
735-770
meta frequency range
680-725 and 750-810
Para frequency range IR
800-840