AP Bio Enzyme Mini-Unit 2024
1/35
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
36 Terms
metabolism
All of the chemical reactions that occur within an organism
Adenine Triphosphate (ATP)
Modified nucleotide used to store and release energy. This is because the bonding of the phosphate groups is unstable.
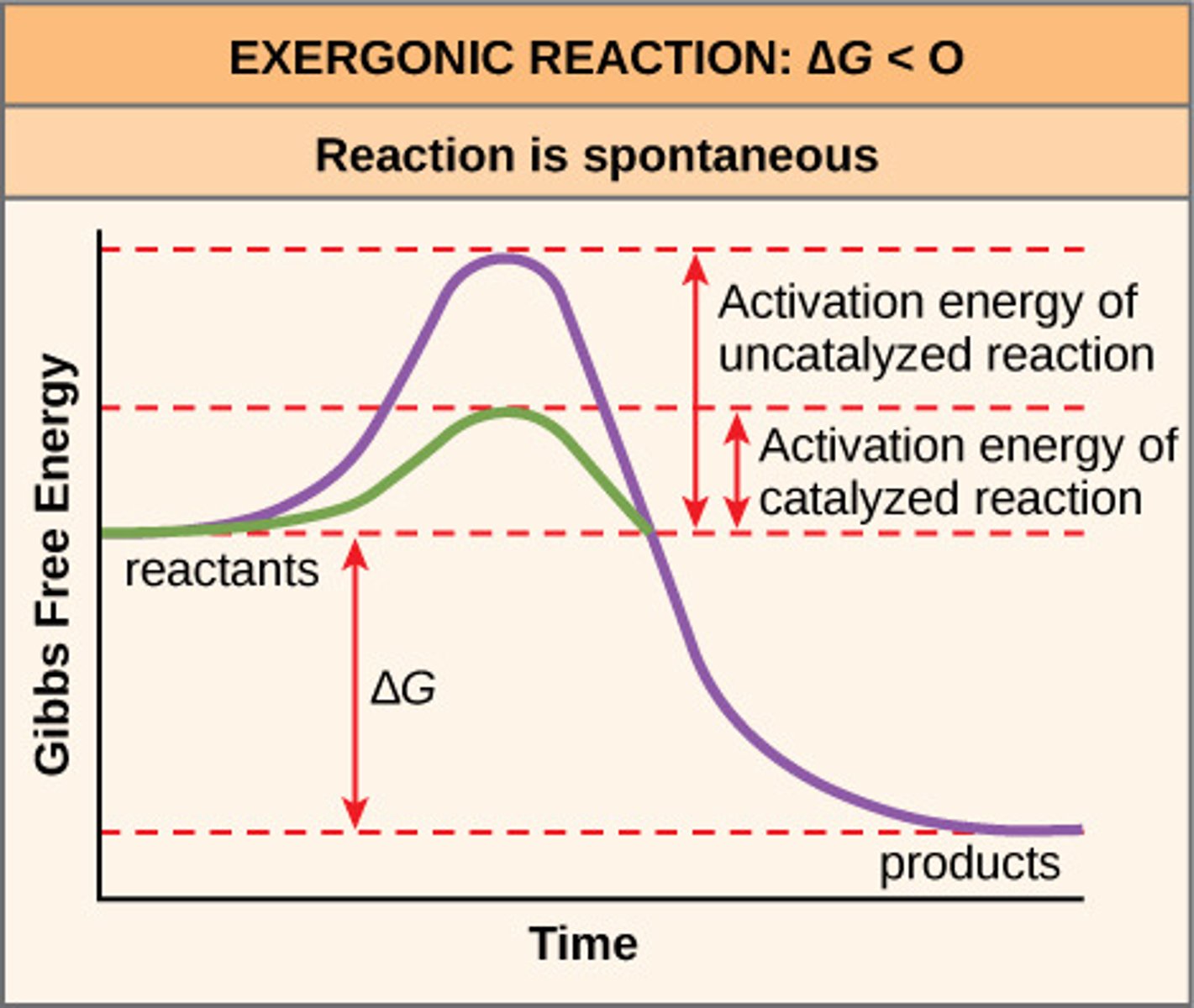
exergonic
releases energy, spontaneous
catabolic
To break down
endergonic
requires/absorbs energy
anabolic
To build up
energy coupling
Using exergonic (catabolic) reactions to fuel endergonic (anabolic) reactions
phosphorylation
Transfer of a phosphate group to a molecule
enzyme that phosphorylates
kinase
ADP
Lower energy molecule that can be converted into ATP. Also the compound that remains when a phosphate group is removed from ATP, releasing energy.
catabolic/exergonic pathways
Release energy by breaking down complex molecules into simpler compounds
anabolic/exergonic pathways
Consume energy to build complex molecules from simpler ones
Potential energy
Energy that matter possesses because of its location or structure
Chemical energy
Potential energy available for release in a chemical reaction
Change in free energy
∆G
∆G = ∆H - T∆S
Change in total energy or enthalpy (∆H) - change in entropy (T∆S)= change in free energy (∆G)
entropy
measure of disorder
enthalpy
total energy of a system
Only processes with ____ are spontaneous
negative ∆G
Exergonic reaction
Releases energy, spontaneous. Reactants > Products, so negative ∆G
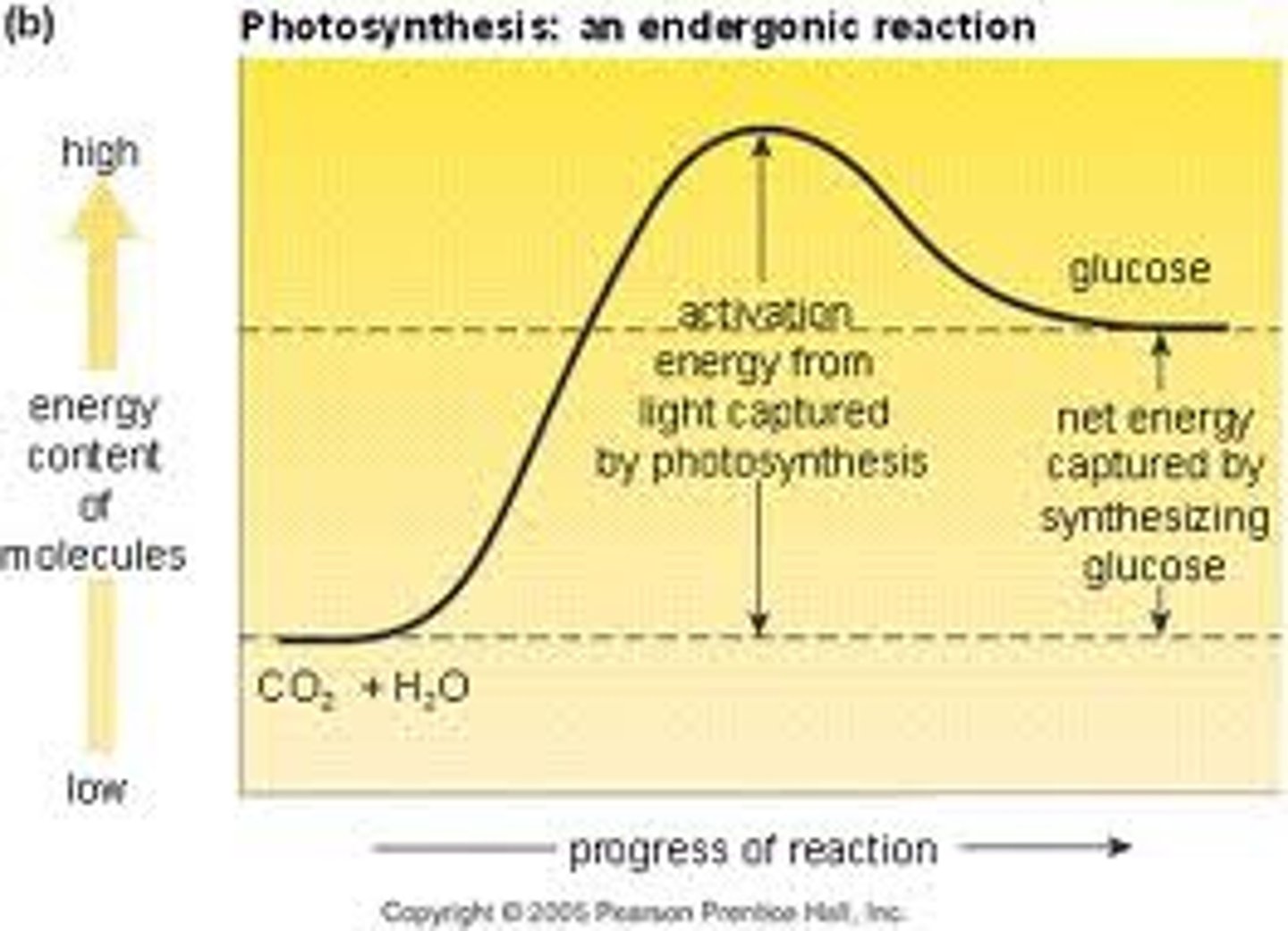
Endergonic reaction
Absorbs energy, nonspontaneous. Products > Reactants, so positive ∆G
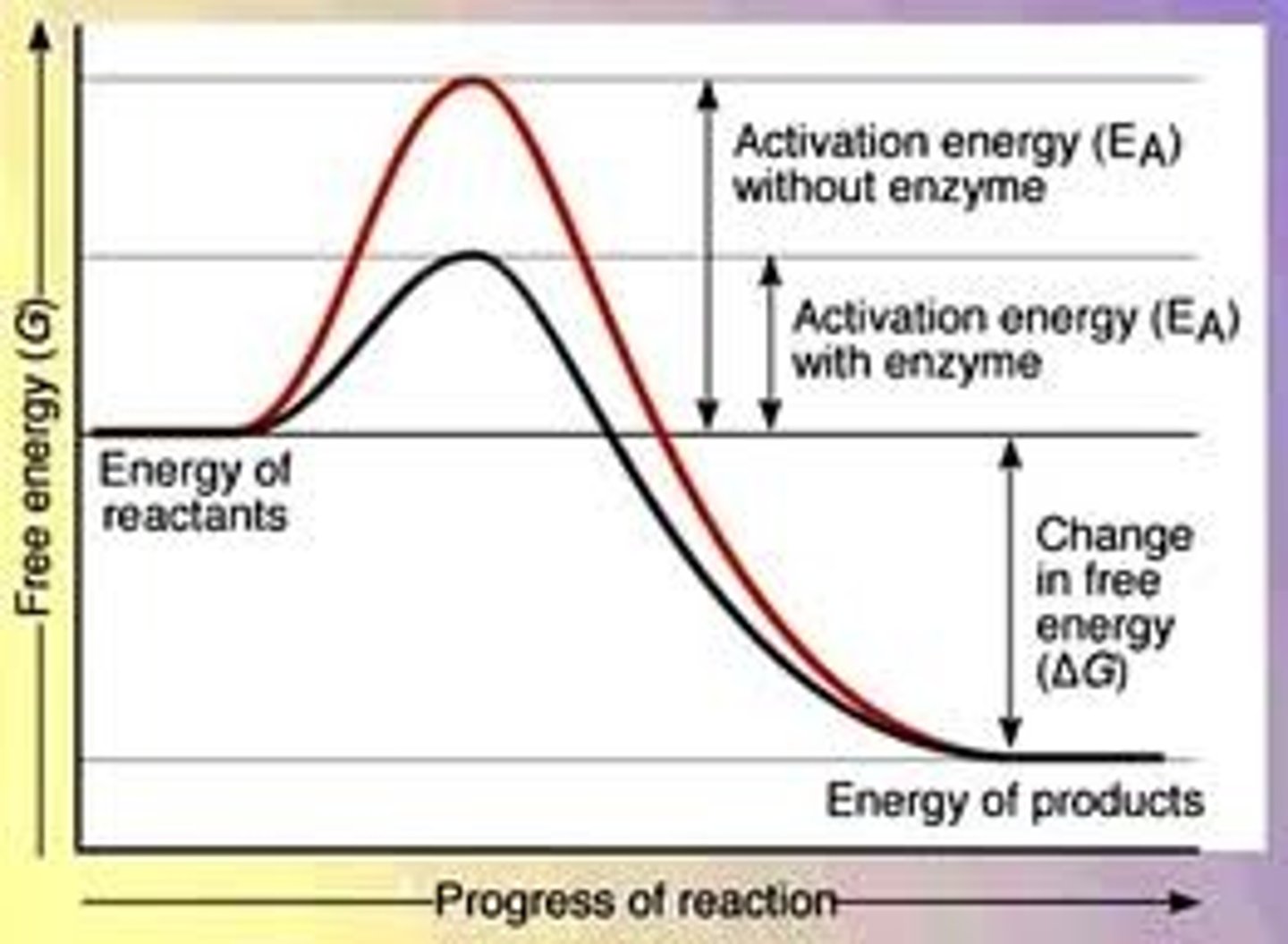
enzymes
proteins that speed up metabolic reactions without being consumed by the reaction, they lower energy barriers
enzymes do not
change ∆G, they just lower activation energy
substrate
reactant of an enzyme-catalyzed reaction
active site
the region on the enzyme where the substrate binds
induced fit of a substrate
brings chemical groups of the active site into positions that enhance their ability to catalyze the reaction

active site can lower an EA barrier by
Orienting substrates correctly
Straining substrate bonds
Providing a favorable microenvironment
Covalently bonding to the substrate
enzyme activity can be affected by
temperature, pH, chemicals (that specifically influence the enzyme), relative concentrations of substrates and products
When do enzymes denature?
too high or too low pH and high temperatures
Denature
Loses structure, thus losing its function
cofactors
nonprotein enzyme helpers
coenzyme
organic cofactors
competitive inhibitors
binds to the active site of an enzyme, competing with the substrate
noncompetitive inhibitors
binds to another part of an enzyme, causing the enzyme to change shape and making the active site less effective
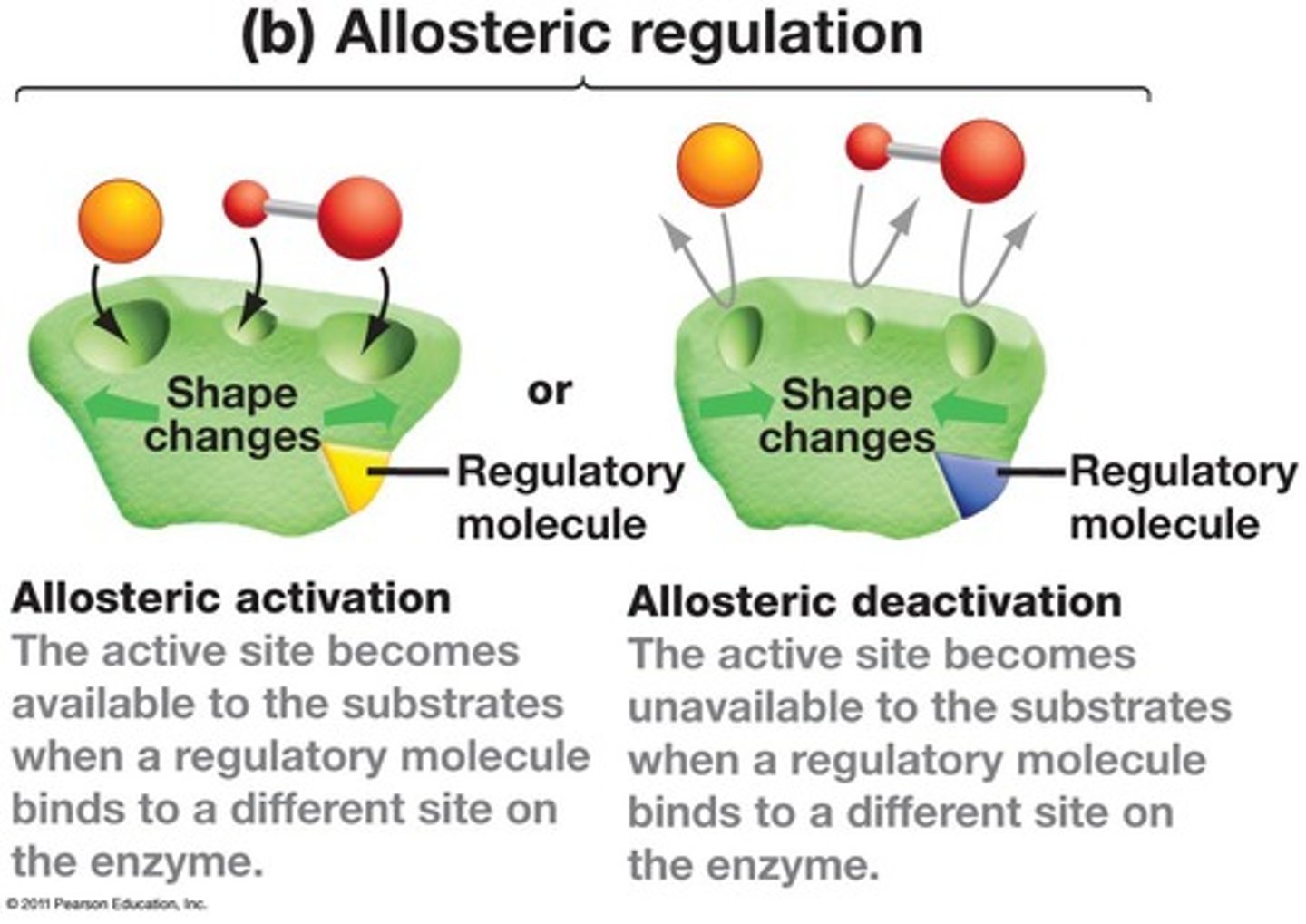
allosteric regulation
The binding of a regulatory molecule to a protein at one site that affects the function of the protein at a different site.
feedback inhibition
the end product of a metabolic pathway shuts down the pathway, this prevents a cell from wasting chemical resources by synthesizing more product than is needed