BICD 158 Midterm 2
1/43
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
44 Terms
Briefly describe the components of an ERACR construct and what it is designed to do. Be sure to mention where these genetic element should be inserted into the genome.
ERACRs (Element for Reversing the Autocatalytic Chain Rxn)
are genetic elements inserted at the same genomic site as a gene-drive & they delete and replace the gene drive
They carry two gRNAs targeting sites immediately adjacent to the gene drive as well as a dominant marker
In the presence of the gene-drive, the two ERACR gRNAs combine with Cas9 and cut out the drive element. HDR-mediated repair then leads to the ERACR copying in place of the gene-drive to replace it.
Generate rare homology-based rearrangments
ERACRs were tested for neutralization of a gene-drive inserted into the X-linked yellow locus of Drosophila. What two distinct genetic phenomena contributed to the ERACR working as it did (hint: one of these mechanisms was the intended process and the other was not)?
1. intended: The ERACR deleted the gene drive and replaced it. In these cases the dominant marker of the ERACR (DsRed) replaced that of the gene drive.
2. Unintended: The ERACR deleted the gene-drive, but did not replace it. In the great majority of these cases, the targeted receiver chromosome originally carrying the gene-drive was damaged so that it could not survive when homozygous. This lethal mutation (m) was subject to strong negative selection. In these cases, the resulting allele carried neither of the dominant markers of the gene-drive or ERACR.
Describe an experiment that supports the interpretation for the mechanism underlying the unintended effect of ERACRs on the target chromosome?
crossing Drosophila melanogaster carrying an ERACR with a standard gene drive to observe the generation of recessive lethal mutations at the target locus
Heterozygous females (m/+) carrying a lethal allele generated by the ERACR were crossed to males carrying a duplication of the region of the chromosome targeted by the ERACR (the duplication is linked to a different chromosome, they translocated the deleted region to autosome)
Survivor progeny could be obtained that inherited the lethal allele from the X-chromosome (ex: become m/m female or m/Y hemizygous male) as long as duplication is inherited
duplication of the targeted chromosome region rescued lethality assoc w/ action of ERACR
What additional insight into this process was provided by experiments with so-called single-cut ERACRs?
Single-cut ERACR are inserted into the same site as the full ERACR but only carry one of the two gRNAs instead of both of them.
These single-cut ERACRs generated damage to the target chromosome at approximately the same frequency as a full ERACR.
suggests that the two gRNAs of the intact ERACR are likely not to cut the genome simultaneously and the damage to the target gene-drive chromosome is likely to reflect the fact that when only a single gRNA is acting one side of the ERACR lacks homology to the cleaved target (ex. On the side the gene-drive that has not yet been cut in the case of a full ERACR)
One of the ERACRs tested carried a recoded version of the yellow gene (ERACR-2) and the other (ERACR-1) did not. Which of these two ERACRs performed best in multi generational population cage experiments and why?
The recoded ERACR performed best. The yellow gene whose function is disrupted both by the gene drive and non-recoded ERACR is essential for male mating fitness (wildtype females strongly prefer to mate with wildtype males versus yellow mutant males). Because the recoded ERACR carries a functional copy of the yellow gene, males carrying this element do not suffer the same fitness cost as males carrying the non-recoded ERACR
In addition to the two actions of ERACRs addressed in question 2 above, what other two types of activities did they manifest when combined with the gene-drive (hint: one was a common outcome and the other rare)?
1. The ERACR sometimes failed to delete the gene drive. In these cases, the ERACR gRNAs generated NHEJ mutant alleles at both cut sites. Such double- resistant gene drive alleles were then immune to future cutting by an ERACR.
2. The ERACR sometimes generated mosaic elements resulting from illegitimate recombination between short elements present in the ERACR that were shared in common with the gene drive and were oriented in the same direction in both constructs
What ERACR design strategy reduced the frequency of the rare events and why?
inversion of the short seq in the ERACR relative to gene-drive —> frequency of the fake recombination events between these elements was quite reduced because they aren’t in same direction & cant easily recombine
Briefly describe the components of an e-CHACR construct and what it is designed to do. Be sure to mention where these genetic elements should be inserted into the genome.
An e-CHACR carries two gRNAs, one for copying the e-CHACR and another for mutation and inactivating Cas9
e-CHACR can be inserted anywhere in the genome and can both copy itself and inactivate a gene-drive inserted elsewhere in the genome
Once the Cas9 gene is mutated, the gene-drive is neutralized since its further drive potential is halted
A gene-drive in mosquitos was rendered less effective by an anti-drive expressing an anti-CRISPR protein (AcrIIA4). How does this anti-CRISPR protein work?
What design feature might the authors of that study have added to their anti-drive genetic construct to make it potentially more effective in reducing spread of the gene drive element?
AcrIIA4 binds to Cas9 and prevents it from binding to DNA (and then cutting it)
The anti-drive element carrying the transgene encoded AcrII4 could also have carried a gRNA cutting at its location of insertion (like a CHACR element). Then the anti- drive would have been able to actively copy itself instead of relying solely on its drive- inhibition phenotype associated with the expression of AcrII4.
Briefly describe two examples of evo-devo experiments that shed light on how changes in gene expression can alter morphological features of organisms.
CRM drives expression of kni gene in narrow stripe corresponding to L2 wing vein of D mel fly
lack this CRM —> lack L2 vein
D. mel L2 CRM repl w/ M Dom —> phenotype looks like M dom
M dom L2-CRM leads to broader zone of kni expression in L2 primordium —> shifting anterior border in anterior direction
entire 38kb hox gene locus pb in D. mel replaced with that D. min
d. mel: maxillary palps perpendicular to proboscis, doesnt have grill like structure
d. mim: maxillary palps parallel to proboscis, male proboscis hase grill like structure
D. mel replacement: mouth looks like D. mel, but palps parallel to proboscis, similar to D. mim
Overall: this reveals pb Hox gene provide abstract positional code that is read by downstream target genes in a species-specific fashion, one exception is reorienting of the maxillary palps reveals that pb also plays a more subtle instructive role in developmental parenting
What is a cis-regulatory module?
enhancer elements
typically short (<1-2 kb) DNA sequences containing binding sites for transcription factors that regulate a transcription of a neighboring gene
can be located upstream (5') , downstream (3') of the transcription unit or can reside in an intron
often act in an orientation and position independent fashion so long as they are within ~100 kb of the gene they regulate.
Briefly summarize two experiments that support the hypothesis that changes in gene expression underlie the 90° change in orientation of the maxillary palps (mouthparts) in Drosophila melanogaster versus its Hawaiian cousin Drosophila mimica
1. A transgenic strain of Drosophila expressing a cDNA encoding the D. mimica Hox Proboscipedia protein in the same pattern as the endogenous D. mel gene fully replaces the function of the D. mel gene
experiment reveals that the D. mimica protein product of the proboscipedia (pb) has the same function as that of D. mel. D mimica proteins work perfectly fine D mel to produce standard mel phenotype
2. A gfp transgene expressed under the control of the D.mimica pb locus exhibits reduced expression during early development of the progenitor cells giving rise to the maxillary palps compared to that of an equivalent gfp transgene expressed under control of the endogenous D. mel gene
experiment reveals that the pb gene is expressed at lower levels in D. mimica at a critical development timepoint than the D. mel gene. confirm lower expression of pb in D. mimica lineage during that critical window is what probably fails to trigger the 90 degree rotation, resulting in parallel orientation
In mice a split-drive system was developed in mice based on a Cas9 source expressed in various tissue-specific patterns using the CRE/LOX recombinase system. Briefly explain how this CRE/LOX system works. The gRNA cassette (inserted into the Tyrosinase locus) copied much better when transmitted via females compared to males, which is opposite to what is observed in insects. Explain why this might be and how the same overall biological principles apply to both organisms.
The CRE recombinase is expressed in a speicific tissue
tissue specific pattern of deleting Lox-Stop-Lox gene cassette (keeps ubiquitous CRM from expressing cas9)
tissue specific expression of cas9
Reason: timeline of germline specification & when they are set apart from somatic cell lineages
Female: germline develops in early embryo, Male: later in adult life
Males have more mitotic cell division in males —> cant be copying in germline
in second generation experiments where Cas9 expression was more precisely targeted to Meiosis 1, some copying of the gene cassette was also observed in males as well as in females
Give two examples of potential uses for gene-drive systems in mammals
Restore native habitats & Immunize endangered species
What feature of the CRISPR system is exploited in creating split-drives?
Ability to separately express the 2 required components for genome cleavage & drive, transgenes encoding cas9 & gRNA
What is the fundamental mathematical difference in the drive behavior of a split-drive system compared to a full gene-drive?
Full gene drive follow second order logistical dynamics (cas9 & gRNA)
Split drive follows first order additive dynamics (gRNA) (increase by adding fixed number of new alleles equal to originally seeded
Briefly describe two split-drive systems that can be converted genetically into full-drive systems. Give an example of unique advantages of each of these systems over a full drive?
What is an advantage shared by both of these systems?
When the two systems are in combined homology between the 2 (btwn DsRed & GFP half-genes) leads to copying of cas9 trans-gene & missing half of GFP transgene into the gRNA element.
Hackable drive:
gRNA bearing comp of this hackable drive carry a full functional copy of a transgene encoding a fluorescent marker (DsRed) and partial copy of a second marker gene (1/2GFP)
Cas9 bearing cassette: carry a full marker gene (GFP) and half of the marker from the gRNA cassette (1/2 Dsred) between which lies the Cas9 encoding transgene. The cas9 construct also carries a gRNA that can cut at a site located btwn the full-DsRed & partial GFp gene in the gRNAgRNA-bearing comp
When the 2 elements are combined homology btwn the 2 constructs btwn dsred & gfp half genes lead to copying of the cas9 transgene & missing half of the GFP transgene into gRNA element
An advantage of this hacking system is that the gRNA cassette is in exactly the sam eloc in genome in both fully hacked config. Any determinants of drive performance that depend on the site of genomic integration element are identical for the 2 constructs
Trans-complementing drive: consists of 1 gene cassette that encodes cas9 & second gRNA containing cassette that carry 2 grnas ( one cuts genome at sit where grna is inserted into genome & second grna cuts at integration site of the cas9 expressing cassette)
these 2 elements are inherited in standard mendelian fashion, but when crossed to e/o the cas9 combines in-trans w/ each of the 2 grna to copy both cassettes
Advantage of both systems:
- the 2 conditional systems permit experimentation to optimize components of the drive system under less stringent containment protocols & facilities than those that are required for studies with full drives. Gene drive experiments must be carried out in ACL-2 level barrier facilities, while split-drive experiments can be conducted under typical lab confinement (ACL-1)
Briefly explain the design of a split-drive experiment that revealed the origin of wide spread NHEJ induced drive-resistant alleles is caused by maternal deposition of Cas9/gRNA complexes in the egg?
An advantage of the trans-complementing system is that one can test 4 different inheritance configurations for the Cas9 and gRNA components. When Cas9 & gRNA are transmitted from separate F0 parents to generate test F1 females carrying both elements, both Cas9 & gRNA elements are transmitted in highly biased super-Mendelian fashion. The same efficient dual transmission was also observed when both Cas9 & gRNA comp were passed from male to the test mother
In contrast, if both element were transmitted from females (red box), the inheritance rates dropped back to nearly Mendelian levels
Findings & supporting DNA seq analysis reveal that co-inheritance of Cas9 & gRNA comp via grandmothers to mothers can generate a high freq of drive resistant NHEJ alleles that dramatically reduce drive efficacy
What single genetic component needs to be added to a gene-drive (full or split) to create an allelic-drive?
gRNA targeting non-preferred allele (ex mutant 1014F allele of VGSC that confers insecticide resistance)
Briefly explain the concept of an allelic-drive.
Preferred allele of a locus is located in another part in genome when the drive element is inserted.
As the drive copies itself at the site of cassette insertion, the second gRNA cuts the non-preferred allele if present.
The double stranded break at the second site is repaired from intact non-cleavable sequences from the preferred allele resulting in the conversion of the non-preferred allele to the preferred allele.
What is an important constraint on the gene targets for an allelic-drive? What is the reason for this condition (i.e., what might go wrong if this constraint was not applied)?
They must be essential for viability or fertility of the organism, essential genes must be targeted so if nhej mutations occur it wont spread
Describe a linked allelic-drive configuration. How does this strategy address the issue raised above?
To create an allelic drive, the gRNA targeting the non-preferred (wildtype or resistant) allele must be added to a full or split gene drive cassette
Describe what is meant by the term lethal (or sterile) mosaicism as it pertains to the performance of recoded split-drives or allelic-drives. What two fundamental conditions must be met to create this phenomenon?
refers to when females carrying a drive element produce Cas9/gRNA complexes during germline development that accumulate in their eggs. If enough cells in the body are so mutated and if the gene is required in a sufficient fraction of cells these individuals will either die (lethal mosaicism) or be infertile (Sterile mosaicism)
Conditions:
i. The target gene is essential for either survival or fertility.
ii. The target gene is expressed broadly and is required to be functioning in a large enough fraction of cells such that mosaic individuals will either die or be sterile.
Reconsider the following situation posed in Week 4, Question 9:
When mosquitos carrying the recoded kh-drive (which have normal fertility and black eyes) are placed in similar population cages, the drive increases in frequency from 10% to over >90% and remains stable thereafter. What process in addition to a standard Mendelian process contributes to the vastly superior outcome of the recoded versus non-recoded kh-drive systems? Which effect is more fast acting?
Conditions:
1. The target gene is essential for either survival or fertility. Thus, individuals carrying a non-functional maternally inherited will exhibit the deleterious phenotype in a dominant fashion.
2. The targeted gene is expressed broadly in the organisms and is required to function in a large enough fraction of cells such that mosaic individuals will either die or be rendered sterile.
3. The drive carries a coded allele or protected allele of the target gene so that any individual inheriting the drive element or the protected allele will have a wild-type phenotype.
The recoded kh-drive produces sterile mosaicism. If mothers carrying a recoded kh-drive allele male with a wild-type male but pass transmit a non-functional NHEJ-induced mutant allele of the target kh gene their eggs will nonetheless accumulate Cas9/gRNA complexes. The paternal wild-type kh allele in those progeny can be mutagenized in a mosaic fashion by those complexes leading to a pseudo homozygous female sterile phenotype. This dominantly acting phenomenon is referred to as lethal mosaicism acts like a catalytic filter to rapidly eliminate individuals inheriting loss of function copying errors. This process is much faster than the standard mendelian process wherein two carriers for a deleterious trait mate and then produce 25% mutant progeny.
Describe what is meant by the term "co-drive" in the context of an allelic drive. What is a likely explanation for this phenomenon?
This term refers to the phenomenon of correlated inheritance of the drive element and the preferred allele in an allelic drive system. In cells biasing for HDR, it likely that both double stranded breaks are repaired through HDR.
Briefly describe the difference between the following two forms of allelic drive: Copy cutting and Copy-grafting. Which process is more generally applicable and why (in one sentence)?
Copy-cutting: driving guide RNA directly cuts the non-preferred allelic variant leaving the preferred allele intact
Copy-grafting: preferred allele is adjacent to a sequence immune to cleavage by a gRNA that can cut WT or non-preferred alleles.
When the gRNA cuts either the WT or non-preferred allelic variants, the ds break is repaired by HDR using the cleave-resistant allele as a template, copying the favored allelic variant in the process.
Briefly describe four different biological mechanisms that can lead to insecticide resistance (one sentence for each)
1) Target site resistance: insecticide no longer binds or alters target site protein
2) Metabolic resistance: insecticide more rapidly metabolized and eliminated
3) Barrier resistance: insecticide less able to penetrate the exoskeleton
4) Behavioral resistance: insects adopt strategies to avoid insecticide exposure
Mutation of the amino acid residue 1014L of the voltage gated sodium ion channel (vgsc) gene in Drosophila results in resistance to DDT and pyrethroid insecticides. What is the basic mechanism by which this mutation provides resistance to these insecticides?
Insecticides no longer can bind with high affinity to the VGSC protein
Briefly describe the design and outcome of an allelic-drive system targeting the DDT (and pyrethroid) resistant 1014F allele of the vgsc gene in Drosophila. What two distinct processes contributed synergistically to the success of this drive?
A split-drive cassette carrying two gRNAs is inserted into the yellow locus:
one gRNA cuts at the yellow locus to copy the gene cassette
2nd gRNA cuts the vgsc 1014F allele but not the WT 1014L allele.
In the presence of Cas9 (prov from sep genetic element), the cassette is copied at yellow and the 1014F allele is converted to 1014L. The two independent processes that contribute to drive of the 1014L allele are: Allelic drive & Positive selection of the 1014L allele over the 1014F allele (carries significant fitness cost, ex: sensitive to elevated temp)
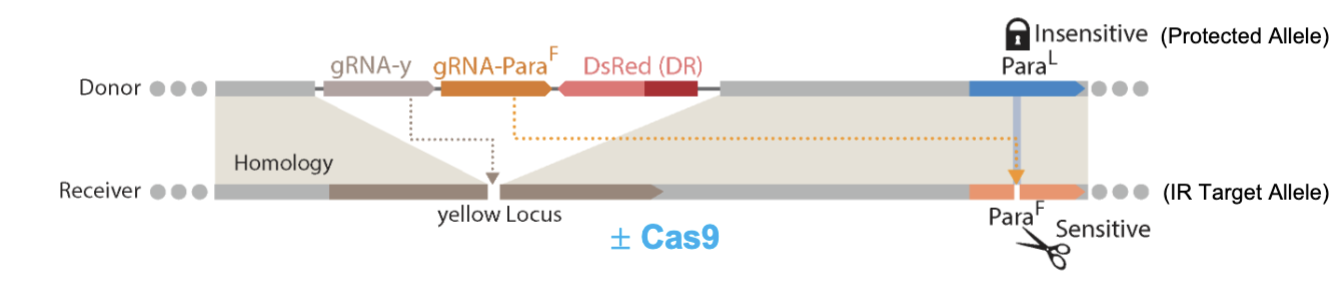
Describe the design and outcome of a unitary self-eliminating allelic-drive system targeting the DDT (and pyrethroid) resistant 1014F allele of the vgsc gene in Drosophila.
Explain how this system can be used in both self-eliminating and persisting (hover) modes. Which mode is most efficient in reversing insecticide resistance?
-The unitary self-eliminating allelic drive inserted into the yellow locus carries all the CRISPR comp from the split drive above in a single gene cassette.
-it lacks gRNA-y for copying gene cassette (hence its ability to perform in a self-eliminating mode)
-male mutants for yellow locus have severe disadvantage relation to WT males for mating with WT females & insertion of the gene cassette results in yellow mutant phenotype bc it disrupts yellow coding sequence
-if wt flies (m & f) are present in target population, the gene cassette imposes a string fitness cost & rapidly disappears from the population (self-eliminating mode). in contrast, if only yellow mutant flies are present in target popu, the gene cassette is fitness neutral & persists in the popu at its initial introduc freq (this hover mode is more efficient in reversing insecticide resistance since gene cassette remains for longer in popu
With the aid of a diagram, describe key features of a linked allelic-drive that biases inheritance of a protective host allelic variant of the mosquito FREP1 gene that is resistant to infection by malarial parasites (recall that in Anopheles stephensi mosquitoes carrying the rare FREP1-Q224 variant cannot be infected by malarial parasites, while those carrying the more common FREP1-L224 allele can be).
What is the key advantage of this linked FREP1 configuration as compared to a non linked system such as the vgsc1014F->L insecticide reversal allelic-drive system?
A linked allelic-drive physically couples: a protective allele (here, FREP1-Q224) a gene drive element (typically CRISPR/Cas9 + gRNA) on the same chromosome, so the drive preferentially copies the protective allele into homologous chromosomes during meiosis.
What is the defining essential feature of a CRISPR-based active genetic element inserted into the genome?
An active genetic element encodes for a nuclease (i.e HEG or Cas9/gRNA) that cleaves DNA at the genomic site where the element is inserted.
Provide a full definition for the term "active genetics". Based on this full definition, briefly explain why active genetic elements copy directionally from a "donor" chromosome to a receiver chromosome and why the reciprocal potential conversion event (i.e., a wild type receiver chromosome converting an active genetic donor chromosome) rarely if ever occurs.
Copying of a genetic element from one chromosome to its homolog in response to a DSB generated in homolog at the same genomic site the active genetic element is inserted in. Copying (results from directional gene conversion) is typically mediated in the germline by the SDSA/D-loop branch of HDR pathway.
SDSA repair pathway works by strand invasion of a SS- 3’ end of DNA generated at the cut site into the double strand template of the intact chromosome. DNA pol then copies seq from intact chromosome w/o modifying repair template. The repaired chromsome then acquires the same local seq as the repair template by directional gene conversion (intact chromosome → cut chromosome). However repair template retains it original sequence since it never gets cut
Define the term R0 as it applies to any infectious disease (e.g., Covid19). In the case of malaria, what are the two primary biological processes that contribute to determining R0?
R0 is the basic reproductive number of an infectious agent (like covid or parasite).
R0>1: infection spread & R0<1: infection recedes
2 primary biological processes that contribute to determining R0 is:
Ability of parasite to propagate in human host
Ability of parasite to reproduce & propagate in the mosquito vector
What are the two basic strategies for the concept of using gene drives to reduce the incidence of malaria vectored by anopheline mosquitos? What are the potential strengths and weaknesses of each of these approaches?
Population suppression: gene drive is designed as a trojan horse to either kill/sterilize the mosquito vector. The drive spreads until it reaches a high enough frequency so that mating pairs of mosquitos often carry the trait & pass on the recessive lethal of sterility traits to their offspring → population crash. drive lethal/sterile mutation to population.
Strength: it reduces the incidence of all of the different diseases that a given mosquito species can vector
Weakness: Impose a strong selective pressure for the evolution of resistance, not stable (needs to be repeated), could potentially do ecological harm
Population Immunization/Modification: gene drive is designed not to inflict a fitness cost on the mosquito but rather to have them express an anti-parasite factor that prevents development & transmission of the parasite. When these mosquitos bite an infected parent they dont transmit, this modifies the phenotype of population
Strength: stable since it does not reduce mosquito populations & does the least ecological harm possible
Weakness: immunization elements must be made for each disease & could drive a more virulent parasite
Austin Burt first proposed the idea of using a suppressive gene-drive element like a Trojan horse to reduce unwanted insect populations by spreading a deleterious trait into the population. What are two key constraints for this process to work effectively (hint: consider cell-specificity of the gene-drive element and the potential mistakes it can make during the copying process)?
1. Activity of an ideal suppressive gene-drive should be strictly confined to the germline. If it is also expressed in somatic cells, could impose a fitness cost when heterozygous and optimally the drive would behave in an ideal recessive fashion only incurring a cost when homozygous.
2. The gene-drive should not be able to generate any functional cleavage-resistant alleles. Such alleles would be under strong positive selection and therefore take over the population.
Among the deleterious traits that could be spread by a suppressive gene-drive Burt considered: lethality, sterility, and grand-childlessness. His technical modeling analysis revealed that the relative efficacy of such strategies could be ranked from most to least effective as: grand-childlessness> sterility> lethality? In non-technical terms explain the basic reason for this order.
delayed effect —> higher proportion of individuals with trait before its expressed (homozygous) —> more individs experience the fitness cost & population crashes more abruptly —> less chance to evolve resistance
Describe the basic features of a CRISPR-based modification gene-drive system designed to reduce the prevalence of malaria.
Cas9 & gRNA work tg to copy the gene to the germline
Anti-parasite effectors get expressed selectively in intestinal epithelial cells
A dominant marker gene (help track gene)
Question 5 asked what two key constraints potentially limit the utility of suppressive gene-drives. Why are modification gene-drives significantly less impacted by these two considerations? What is a concern specific to the modification strategy (e.g., consider gene-drives designed to reduce the prevalence of malaria as an example).
Germline Specificity:
Suppression drive: fitness cost could occur in heterozygotes which could select against the drive if there is leaky activity in somatic cells
Modification drive: does not give a homozygous fitness cost
Target site drive-resistant allele
Suppression drive: NHEJ induced mutations that prevent cleavage and maintain gene function have a fitness advantage over drive insertion alleles
Modification Drive: cleavage resistant alleles don’t have a predicted advantage
Some gene-drives carry a recoded version of the gene into which they are designed to insert (e.g., the kh-drive in Anopheles stephensi). These recoded transgenes restore function of the targeted gene drive elements that would otherwise be disrupted by the insertion event (e.g., female mosquitos homozygous for the kh-drive are sterile and have white eyes: recall that wild-type mosquitos have black eyes). Females carrying a non-recoded version of the kh-drive transmit the drive element to progeny (60%) much less efficiently than males do (~99%). What is the basis for this large difference between transmission from females versus males?
Female carriers:
Egg carries cytoplasm with proteins, including cas9/grna complexes produced during meiosis
Egg is fertilized by WT sperm with target allele, cas9/gRNA complexes in cytoplasm can act on chromosome to cut it before it has an opportunity to pair with the drive allele, generating cleavage resistant NHEJ alleles
Non-recoded allele carriers carry a fitness cost when homozygous, resulting in lethal or sterile mosaicism meaning have both a nonfunctional NHEJ allele & nonfunctional drive allele
Male carriers:
Sperm delivers dna to egg & paternal drive allele pair with WT maternal allele and get copied efificiently when Cas9 & gRNA transgenes are expressed zygotically
The difference observed are:
Eggs carrying NHEj can be produced in both cases
Recorded drives will not generate lethal or steril mosaic phenotypes since all somatic cells are at least heterozygous for functional allele
When mosquitos carrying the non-recoded kh-drive (consisting of both males and females) are tested for gene-drive activity in multi-generation population cages, the frequency of the drive in the population first increases substantially from the initial seeding frequency (e.g. 10%), but then either declines or results in the population crashing to zero. What are the reasons behind the two different outcomes?
Non-recoded kh-drive drive: insertion allele carries the same fitness cost as LOF NHEJ alleles
Non recoded element drives: results in homozygous females with white eyes that are infertile, resulting in a population crash. making population sterile, giving non-recorded kh drive act as suppression drive
When mosquitos carrying the recoded kh-drive (which have normal fertility and black eyes) are placed in similar population cages, the drive increases in frequency from 10% to over >90% and remains stable thereafter. What standard Mendelian process contributes to the vastly superior outcome of the recoded versus non-recoded kh-drive systems?
Recoded kh drive has no mutant phenotype or fitness cost when homozygous, when the recorded kh drive reaches a high frequency in the population it persists
Modeling results for suppression drives indicate that these drives will fluctuate in frequency when released in nature across large distance scales. The average population sizes of mosquitos are predicted to be substantially decreased (by up to 90% for optimally performing drives), but neither the wild-type nor the gene-drive mosquito populations ever disappear permanently. Why is this so?
Imagine that a modification gene-drive system can reduce the rate of parasite transmission by ~90%. What effect on malarial transmission (e.g., R0) would you expect if you treated an area first with a suppression drive (90% efficient as above) and then with the modification drive?
drive is efficient locally —> able to reduce population to essentially 0, being lethal or sterilizing the population, making them not able to produce more progeny
egg laying wt female mosquito flies in from a diff outside region outside of the local region & repopulate the region locally
when population is growing & stable, a drive-containing fly from an area where that wasn’t completely eliminated is going to come into this population again & reduce it
cycle & oscillate back & forth btwn above states, so it’s never going to completely eliminate
It reflects the dynamics of suppression across a large area, if the drive is efficient, it should behave locally as it does in population cages by reducing populations to zero.
When this occurs, the only type of mosquito that could fly in & repopulate the area is wt mosquito