Chem Paper 1 Exam Qs
1/90
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
91 Terms

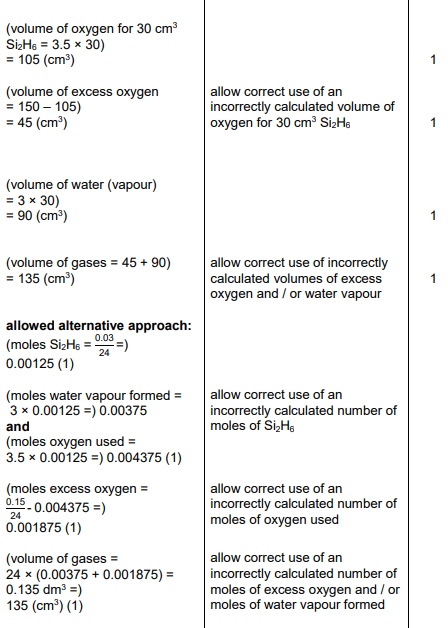
2024: Gove one reason for filtering the mixture in step 5 (1)
to remove excess copper carbonate
2024: Some salts can be produced by reacting sulfuric acid with a metal.
Neither copper nor sodium is used to produce a salt with sulfuric acid.
Give one reason why each metal is not used (2)
Copper: Unreactive
Sodium: Reaction is dangerous/ could explode/ could get too hot
Observations made when alkali metals react with water
effervescence
metal floats
metal moves around
metal becomes smaller
metal melts
flame
2024: A student investigated the energy change of the reaction between zinc and box copper sulfate solution.
Explain why using a polystyrene cup gives more accurate results than using a glass beaker (2)
polystyrene is a better insulator
so there is less energy/heat transfer/loss to the surroundings

2024: The results show random errors
The student did not make any measuring errors
Suggest one reason for the random errors in this experiment (1)
any one from:
starting temp may be different
inconsistent stirring

2024

2024: Describe how a hydrogen fuel cell produces a potential difference (2)
hydrogen is oxidised (electrochemically)
to produce water
2024: Iron is a metal.
Describe how iron conducts thermal energy (2)
energy/heat is transferred
by delocalised electrons
2024: Pure iron is too soft for many uses.
Explain why mixing iron with other metals makes alloys which are harder than pure iron (3)
2024: Explain why propane has a low boiling point (3)
propane is a small/simple molecule
with weak intermolecular forces
which require little energy to overcome

2024


2024:

2023: The atom Tennessine was first identified by a small group of scientists in 2010.
Suggest one reason why tennessine was not accepted as a new element by other scientists until 2015 (1)
needed peer review
2023: Plan a method to make pure, dry crystals of zinc chloride from zinc carbonate and a dilute acid (6)

Name the substances that can be reacted with a dilute acid to make zinc chloride
zinc
zinc carbonate
zinc hydroxide
zinc oxide
2023: The reaction between hydrogen and chlorine is exothermic.
Explain why this reaction releases energy to the surroundings (2)

Indicators and colour changes possible in titration
methyl orange
yellow to red/orange/pink
phenolphtalein
pink to colourless
litmus
blue to red
2023: Which element has atoms that have the same electronic structure as the chloride ion?
Use the periodic table (1)
argon
2023: Describe how metals conduct electricity (3)
delocalised electrons
electrons carry charge
electrons more through the structure
2023: Suggest why alloys do not conduct electricity as well as pure metals
Answer in terms of structure and bonding (2)
in alloys different sized atoms distort the layers/structure
so the movement of (delocalised) electrons is restricted
2023: Cryolite contains Na+ ions as well as Al 3+ ions. Suggest one reason why sodium is not a product of the electrolysis of aluminium oxide (1)
sodium is more reactive than aluminium
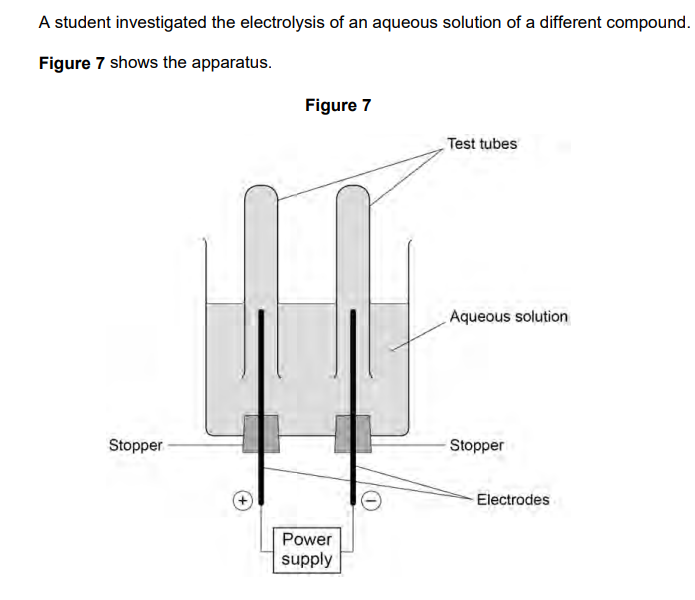
2023: Hydrogen was produced at the negative electrode and oxygen was produced at the positive electrode.
Explain how oxygen was produced from water during the electrolysis of this aqueous solution (4)
2023: Argon has the atomic number 18
Explain why argon does not form compounds.
Answer in terms of electrons (2)
argon has a stable arrangement of electrons or a full outer shell
so argon atoms do not SHARE/TRANSFER electrons

2023 (2)

Metal + acid →
salt + hydrogen

2023 (2)


2022: (4)


2022

2022: Describe the structure and bonding of diamond (3)
giant structure
covalent bonds
four bonds per carbon/atom
2022: Explain why diamond has a very high melting point (3)
bonds are strong
bonds must be broken
lot of energy is required
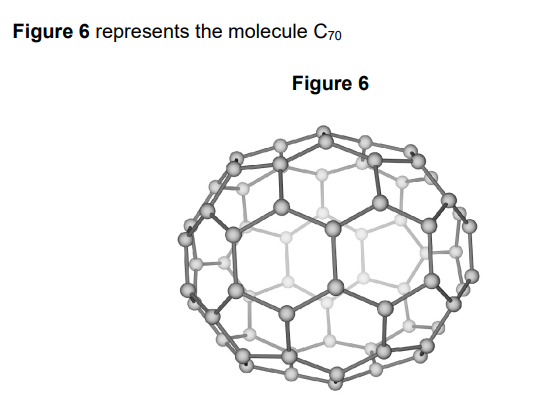
2022: Molecules such as C70 can be used in medicine to move drugs around the body.
Suggest one reason why the C70 molecule is suitable for this use (1)


2022 (3)


2022
4.1. solid remaining
4.2. zinc oxide can be filtered off
2022: Describe how the student should obtain crystals of zinc chloride from a solution of zinc chloride (2)


2022
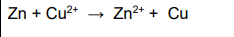

2022
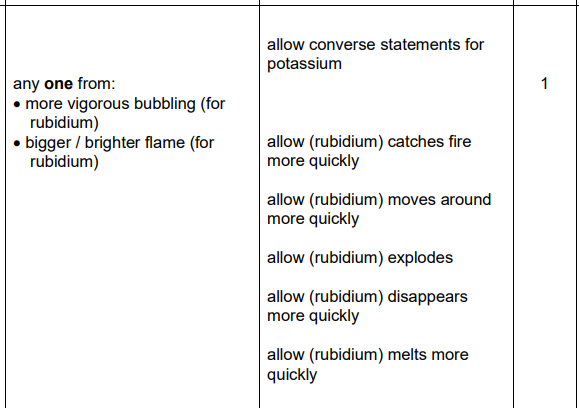
2022: Explain why rubidium is more reactive than potassium (3)

Alkali metals + water
metal hydroxide + hydrogen
acid + alkali
salt + water
acid + metal carbonate
salt + water + carbon dioxide
How do boiling points change down group 0?
increase
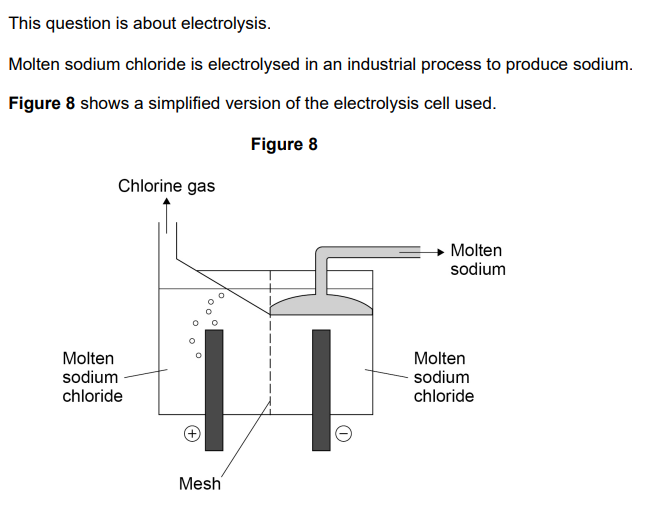
2022: Which type of particle passes through the mess in the electrolysis of molten sodium chloride? (1)
Atom
Electron
Ion
Molecule
ion
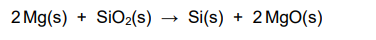
2022: Magnesium is used to reduce silicon dioxide
(see picture for equation)
Give one reason why the products are difficult to separate if magnesium is used to reduce silicon dioxide (1)
both products are solid

2022 (4)
2022: Explain why the pH of an acid depends on:
the strength of the acid
the concentration of the acid (4)

Observations when alkali metal is added to water
metal floats
metal melts
metal moves around
metal becomes smaller or disappears
flame
effervescence
2021: Why does sodium oxide have a high boiling point? (3)
giant structure
strong electrostatic forces of attraction between oppositely charged ions
large amounts of energy needed to overcome the forces
2021: Explain why alloys are harder than pure metals (3)
atoms are different sizes
so layers are distorted
the layers slide over each other less easily than in a pure metal
2021: Explain the difference between the processes in electrolysis and in a chemical cell (2)
electrolysis uses electricity to produce a chemical reaction
but cells use a chemical reaction to produce electricity
How to improve titration method:
swirl solution
white tile under flask
add acid dropwise near endpoint
repeat and calculate mean

2021 (3)

(4)

2021: Why are reactions with a high atom economy used in industry
any one from:
for sustainable development
for economic reasons
to produce high(er) percentage of useful product
to reduce waste
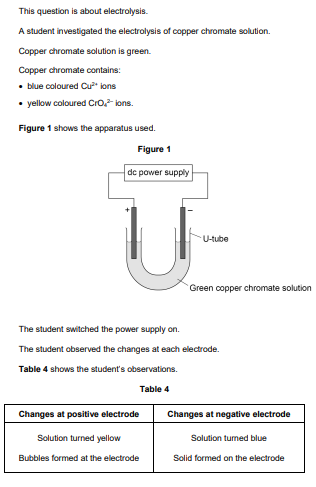
2021: Describe how the solid forms at the negative electrode (3)

Why is the maass of products not same as mass of reactants?
unexpected side reactions
gas: lost to surroundings
products lost between reaction vessels
reversible reaction did not go to completion
2020: Reason why reactions with high atom economy are used in industry (1)
for sustainable development
economic reasons
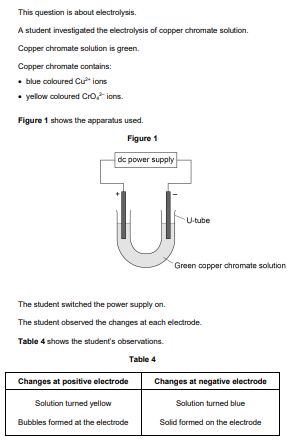
2020: Describe how the solid forms at the negative electrode (3)

2020: Describe the change that Bohr made to the nuclear model (2)
electrons orbit the nucleus
electrons are at specific distances from the nucleus
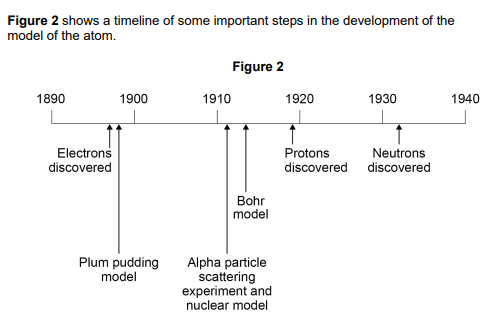
2020:
Mendeleev published his periodic table in 1869.
Mendeleev arranged the elements in order of atomic weight.
Mendeleev then reversed the order of some pairs of elements.
A student suggested Mendeleev’s reason for reversing the order was to arrange the elements in order of atomic number
Explain why the student’s suggestion cannot be correct
Use figure 2 (2)
atomic number is the number of protons
protons were not discovered until later

2020: (2)

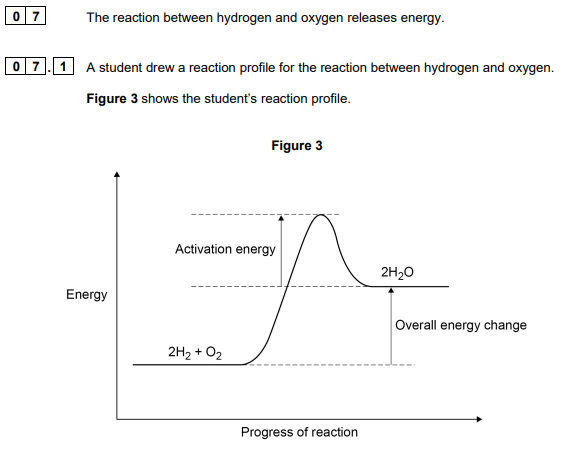
2020: The student made two errors when drawing the reaction profile
Describe the two errors (2)

2020: Advantages of hydrogen fuel cells compared to rechargeable cells

2020: Half equations hydrogen fuel cells

2020: Limitations of particle model of matter (for gas)
particles shown as spheres
particles shown as solid
does not show forces (between particles)
does not show movement/speed (of particles)
only two dimensional
2020: The hydrogen gas needed to power a car for 400 km would occupy a large volume.
Suggest one way that this volume can be reduced (1)
any one from:
under (higher) pressure
cool
absorb/adsorb in a solid
store as a liquid/solid
2020: Why is it not correct to say that the boiling point of a single bromine molecule is 59oC? (1)
boiling point is a bulk property
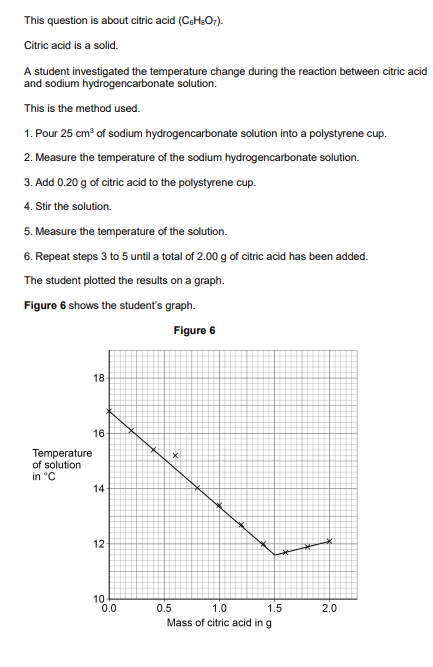
2020: Explain the shape of the graph in terms of the energy transfers taking place. You should use data from Figure 6 in your answer (3)

2020: Give two reasons why a burette is used for titration (2)

Difference between boiling and evaporating
Evaporation:
Occurs at any temperature below boiling point
Occurs at the surface
Slower than boiling
No bubbles are formed
Only the fastest (highest-energy) particles escape the surface
Boiling:
Occurs at a specific, fixed temperature (boiling point)
Occurs throughout the whole liquid (bulk property)
Faster than evaporation
Bubbles of gas form within the liquid and rise to surface
Requires a constant input of heat energy to maintain the temperature
Describe the structure of a metal (4)
giant lattice structure
of positive ions/atoms
with delocalised electrons
positive ions and electrons attract each other and hold the structure together
Explain why diamonds are hard (4)
giant covalent structure
covalent bonds
4 bonds per carbon atom
which need lots of energy to overcome
Why can graphite conduct electricity (3)
each carbon atom forms 3 (Covalent) bonds
one electron per atom becomes delocalised
delocalised electron can travel through structure and carry charge/electricity
Why are fullerenes good lubricants?
spherical shape means it can roll/slide
Uses of fullerenes
lubricants
drug delivery
catalysts
reinforcing materials
The surface area of some metals corrode when exposed to air
Suggest why their conductivity is reduced (2)
a metal oxide compound is formed
so there are fewer electrons to carry charge
Explain why (giant ionic compound) has a high melting point
giant lattice structure
strong electrostatic forces of attraction between oppositely charged ions (acting in all directions)
which need a lot of energy to overcome

2025 (4)

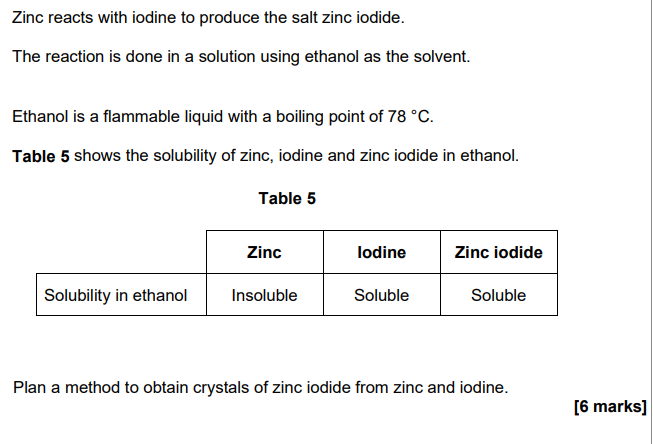

2019: Mendeleev left spaces in his periodic table
He left these spaces because he thought missing elements belonged there.
Why did Mendeleev’s periodic table become more widely accepted than previous versions? (3)
mendeleev had predicted properties of missing elements
elements were discovered that filled the spaces/gaps
properties (of these elements) matched Mendeleev’s predictions
2019: What is the modern name for atomic weight? (1)
relative atomic mass
2019: Describe what you would see when alkali metals react with chlorine (2)
flams
(white) solid forms
colour of gas/chlorine disappears/fades

2019: (6)

2019: Limitation of dot and cross diagram (1)
does not show the shape
only two dimensional
2019: Explain why a reaction is exothermic (using bond energy calculations) (2)
More energy is released in making bonds than used in breaking bonds
so energy is released (to surroundings)
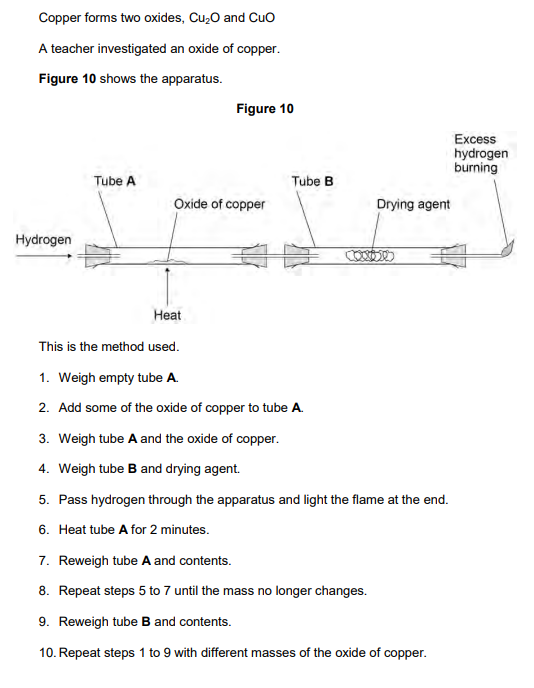
2019: Suggest one reason why step 8 is needed (1)
to make sure all of the oxide has reacted or to make sure all water is removed
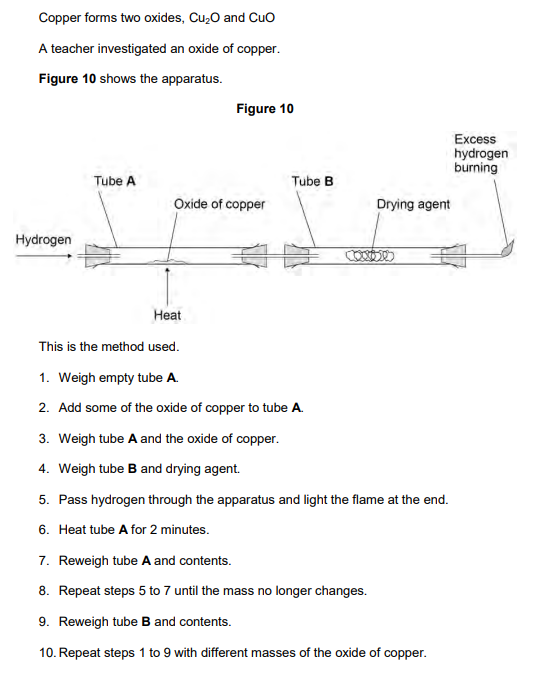
2019: Explain why excess hydrogen must be burned off (2)
to prevent hydrogen escaping
(because) hydrogen is explosive
2019: How to convert from mol/dm3 to g/dm3
concentration in mol/dm3 x Mr
2018: Why do alkaline batteries eventually stop working? (1)
reactant is used up

2018 (3)
2018: Explain why an acid can be described as both strong and dilute (2)
strong because completely ionised
dilute because small amount of acid per unit volume
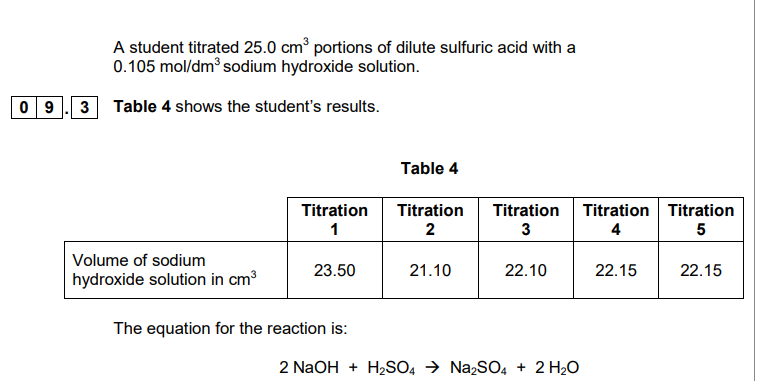
2018: Explain why the student should use a pipette to measure the dilute sulfuric acid and a burette to measure the sodium hydroxide solution (2)
