Biochem 1B - Biological Catalysis
1/76
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
77 Terms
what can we see from thermodynamics
By looking at thermodynamics we can see if the reaction is feasible. Can see its exothermic and find the entropy and enthalpy and work out the Gibbs free energy to see if the reaction is spontaneous or not (if it can happen or not)
Thermodynaics tells us whether it can happens
what does kinetics tell us
Kinetics tells us how fast that reaction occurs
spontaneous doesnt mean fast!
why is kinetics important
Many important processes involve kinetics
what do we mean by rate
The rate of a reaction tells us how fast the reaction occurs.
how do we calculate rate
Proportional to the concentration of the species
Rate = ΔC / ΔT
what will
Rate = ΔC / ΔT give us
But this gives us a curved line not a straight line so a better way to do this is differentiation. Differentiating this line will give us a gradient, the gradient will be our rate of reaction
what does differentiation do
Differentiation allows the gradient of a line to be measured at any particular point
how do we solve differentiation
y = ax^b
dy/dx = a(b)x^b-1
how do we calculate instantaneous rate
Rate (v) = d[concentration]/dt
units of concentration
Concentration can be measured in many different units. E.g. mol dm-3, mg ml-1, molecules ml-1, Torr(pressure for a gas).
units of rate
Units of rate are concentration/time.
mol dm-3 s-1
mg ml-1 min1
molecules ml-1 min-1
Torr s-1
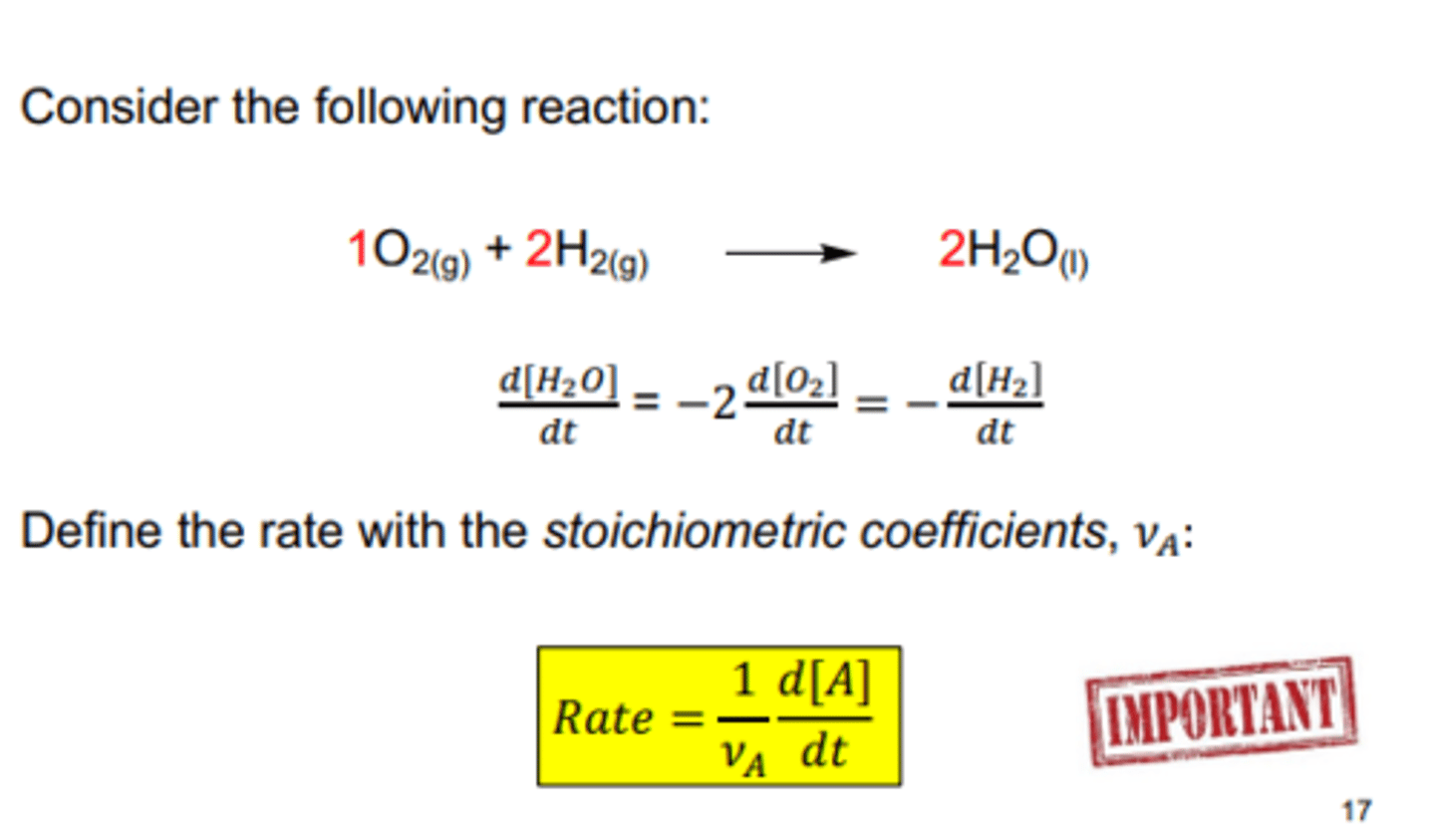
relating rate to the stoichiometric coefficient
Define the rate with the stoichiometric coefficients, 𝜈𝐴:
Always define the rate of a reaction with a stoichiometric coefficient (number in front of the molecule), if its on the products side its positive if its on the reactant side its negative
what is rate law
Rate laws express the relationship between rates and concentrations in reactions.
rate law calculation
k is the rate constant. It depends on the temperature and other factors.
n + m is the order of the reaction. m is the order with respect to A, n is the order with respect to B.
Units of k depend on the order of the reaction
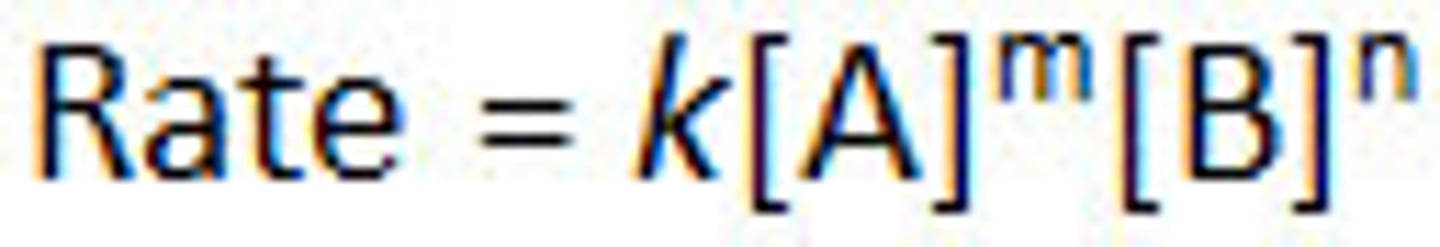
can the rate law be predicted from equations
The rate law cannot be predicted from the stoichiometric equations - it must be determined experimentally, or predicted theoretically
importance of rate laws
Rate laws help predict reaction rates and understand reaction mechanisms.
what types of order reactions have distinct rate laws
Zeroth, first, and second order reactions have distinct rate laws.
what is first order with respect to a reactnat
if we double concentration it will double the rate
what is second order with respect to a reactant
if we double the reaction it will x4 the rate
what is zeroth order with respect to a reactant
doubling the concentration will have no effect on the rate
what is a first, second and zeroth order reaction
sum of the order with respects to the reacants are 1, 2 or 0
simple rate laws
0th order: d[A]/dt = -K0[A]^0
1st order: d[A]/dt = -K1[A]^1
2nd order: d[A]/dt = -K2[A]^2
what is really important in simple rate laws
Units are really important
what are the units of the rate constant depend on
The units of the rate constant depend on the order of the reaction
0th conc. time-1
2st time-1
conc.-1 time-1
what are rate laws
Rate laws are differential equations that describe the rate of change of a reactant or productconcentration with time.
We can solve these equations to find out how the concentration of the reactant or product changes as a function of time using integration
what does integration allow
Integration allows the determination of concentration changes over time in reactions.
straight line gtaph
y = mx + c
Where m is the gradient, and c is the y-intercept
If we can rearrange an equation into this form, it's easy to find the values of m and c graphically
In the case of simple rate laws, if given [A] vs. t, do the following
1. Plot [A] vs. t If linear: 0th order, gradient = -k0th
2. Plot ln[A] vs. t If linear: 1st order, gradient = -k1st
3. Plot 1/[A] vs. t If linear: 2nd order, gradient = k2nd
what happens as rate laws becomes more complex
As rate laws become more complex, it becomes difficult to integrate them by hand.
Can involve multiple concentrations, and so sometimes not even possible
more complex second order reaction
[A] = [B] at time zero tso that is the case at all times to rate law becomes:
d[A]/dt = -k2nd[A]^2
how can we solve complex rate laws
Isolation Method and Differential Method
what is the isolation method
The isolation method involves varying reactant concentrations to determine rate constants.
what is the differential method
Differential methods analyze the rate of change of reactants and products over time.
what is half lives
Another way of determining the orders.
The half life, t1/2, is the time it takes for the concentration of a specific reagent to fall to half of its originalvalue.
how can half lives be used
he half lives can be used to determine the rate constants
or simple reactions, they can be used to predict the order:
Zeroth order: life gets shorter as the reaction proceeds.
First order: Half-life is constant.
second order: successive half lives double
what happens in a chemical reaction
All chemical reactions involve the breaking/making of bonds
This is the result of electrons moving from one molecular orbital to another
how can we calculate the energy of all the electrons
Using molecular orbital (MO) theory
what is MO theory
Quantum theory can be utilized to calculate the energy of a system and how it varies with different nuclear arrangements
When atoms A and B are distant, the energy is the sum of the energies of A and B.
The energy changes as the molecules approach each other, depending on the arrangement of all atoms in the system.
what does a potential energy surface do
The energy changes as the molecules approach each other, depending on the arrangement of all atoms in the system.
what is the transition state
The transition state (≠) exists at the potential energy maximum.
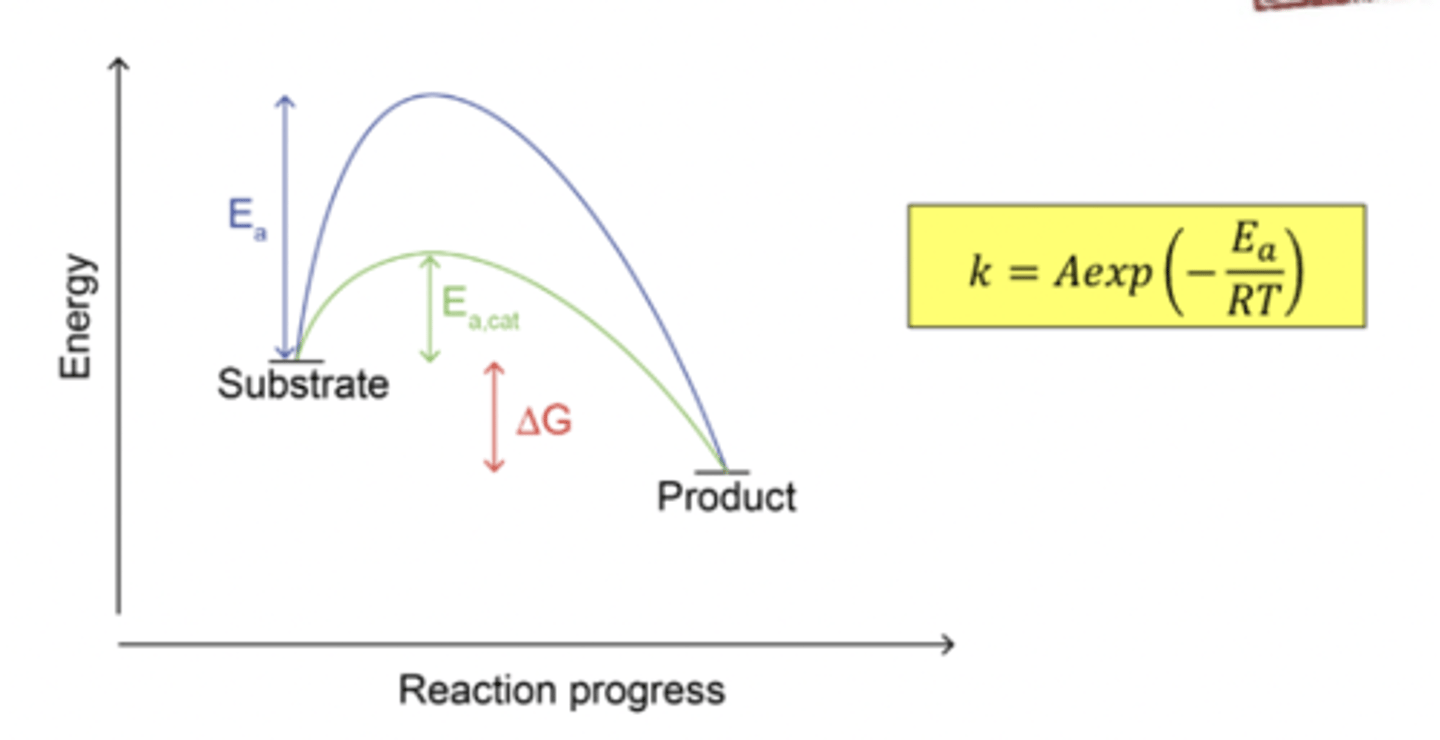
what is the activation energy
The activation energy (𝐸𝑎) is the difference in energy between the transition state and the reactants. (Transition state is the highest energy point on the lowest energy pathway)
It is the energy required for the reaction to occur- needed to break bonds
collision theory
One way of understanding reactions is to assume that they take place when molecules collide
what is wrong with collision rate
he reaction rate is found to be lower than the collision rate.
This is because of the activation energy.
Not only must the molecules collide, but they must collide with enough energy to overcome the energybarrier for the reaction
what is the Maxwell-Boltzmann distribution
Maxwell-Boltzmann energy distribution curves give the distribution of kinetic energy of the particles at agiven temperature.
The highest point represents the most probable energy of the particles.
It only applies to gases, but the idea can also be applied to liquids
The fraction of molecules having enough energy to overcome the energy barrier and reach the transitionstate can be calculated:
probability = exp(-Ea/RT)
Where R is the gas constant, and T is the temperature in Kelvin
how do we calculate overall rate using collision rate and energy requirement
Rate =Zexp(-Ea/RT)
This is the successful number of collisions in terms of molecules per unit time per unit volume
Therefore, per mole:
v= Z/Na exp(-Ea/RT)
Temperature dependence of the rate constant
Experimentally, it has been found that many rate constants are found to vary according to the Arrheniusequati
k(T) = Aexp(-Ea/RT)
Where A is the pre-exponential factor, which is often found to be independent of temperature, or at least only weakly dependent on temperature
Comparison between experiment and theiry
p = Aexperiment/A collision theory
what are elementary reactions
Elementary reactions are basic building blocks of a reaction involving a small number of molecules
eg. H + Br2 --> HBr + Br
These reactions cannot be broken down further and are the fundamental steps in a reaction.
what is molecularity in elementary reactions
Molecularity refers to the number of molecules involved in an elementary reaction.
It can be unimolecular (involving 1 molecule) or bimolecular (involving 2 molecules).
is it different from the overall order of a reaction
Molecularity is different from the overall order of the reaction.
Molecularity refers to the number of molecules or atoms that must collide to produce a reaction, while the overall order of the reaction is the sum of the powers to which the concentrations of the reactants are raised in the rate law equation
rate laws for elementary
Rate laws can be defined for elementary reactions
Unimolecular reactions are first order with respect to the reactant
Bimolecular reactions are second order with respect to the reactants.

how can the rate law be written/determined
Rate laws for elementary reactions can be directly determined by inspecting the stoichiometric equation.
Rate Equations for Consecutive Reactions
In consecutive reactions with an intermediate, rate equations can be written for each species.
(remember + if being formed and - if we are losing it)

sequential first order reactins
In sequential first order reactions, the rate determining step is the one with the smallest rate constant.
The overall rate of production is dependent on the rate of the slowest step
pre-equilibrium hypothesis
In many reactions, species involves in rate determining step are in equilibrium with the reagents:
The rate of the backward process is much faster than the forward process, leading to an equilibrium state
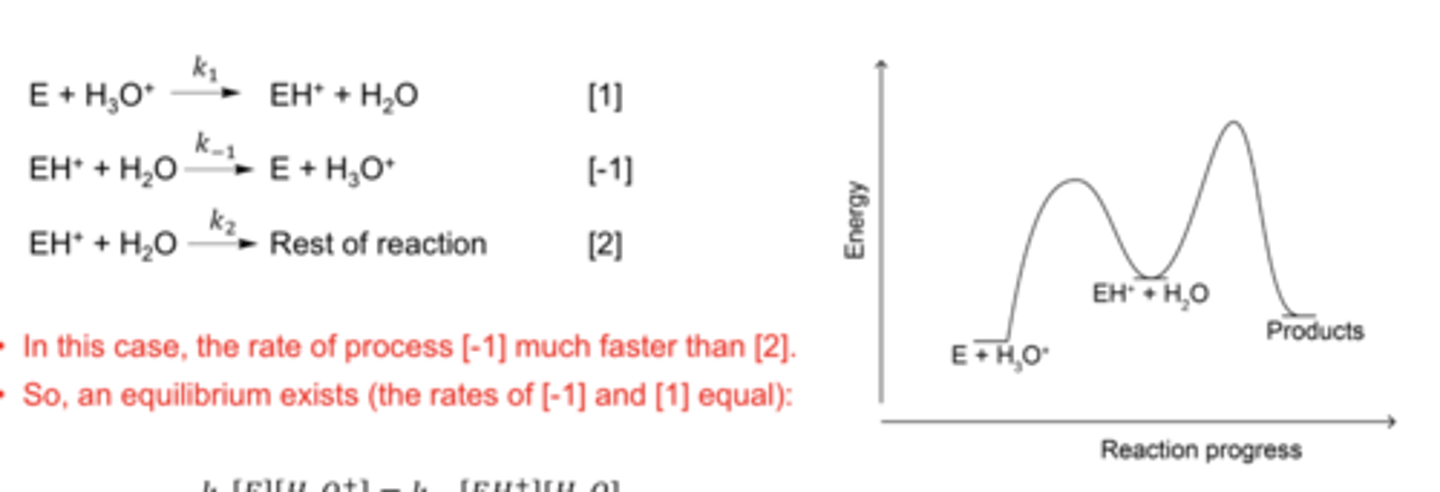
rate reaction for pre-equilibrium

how to calculate a pre-quilibrium
1. Write out equilibrium constant
2. Write out how the reaction occurs to form the product
3. Substitute your intermediate into that reaction
what does the steady state approximation do
Steady State Approximation (SSA) simplifies solving differential equations by assuming constant concentration of intermediates
Used when the reaction involves intermediates at a constant concentration, termed 'in the steady state'
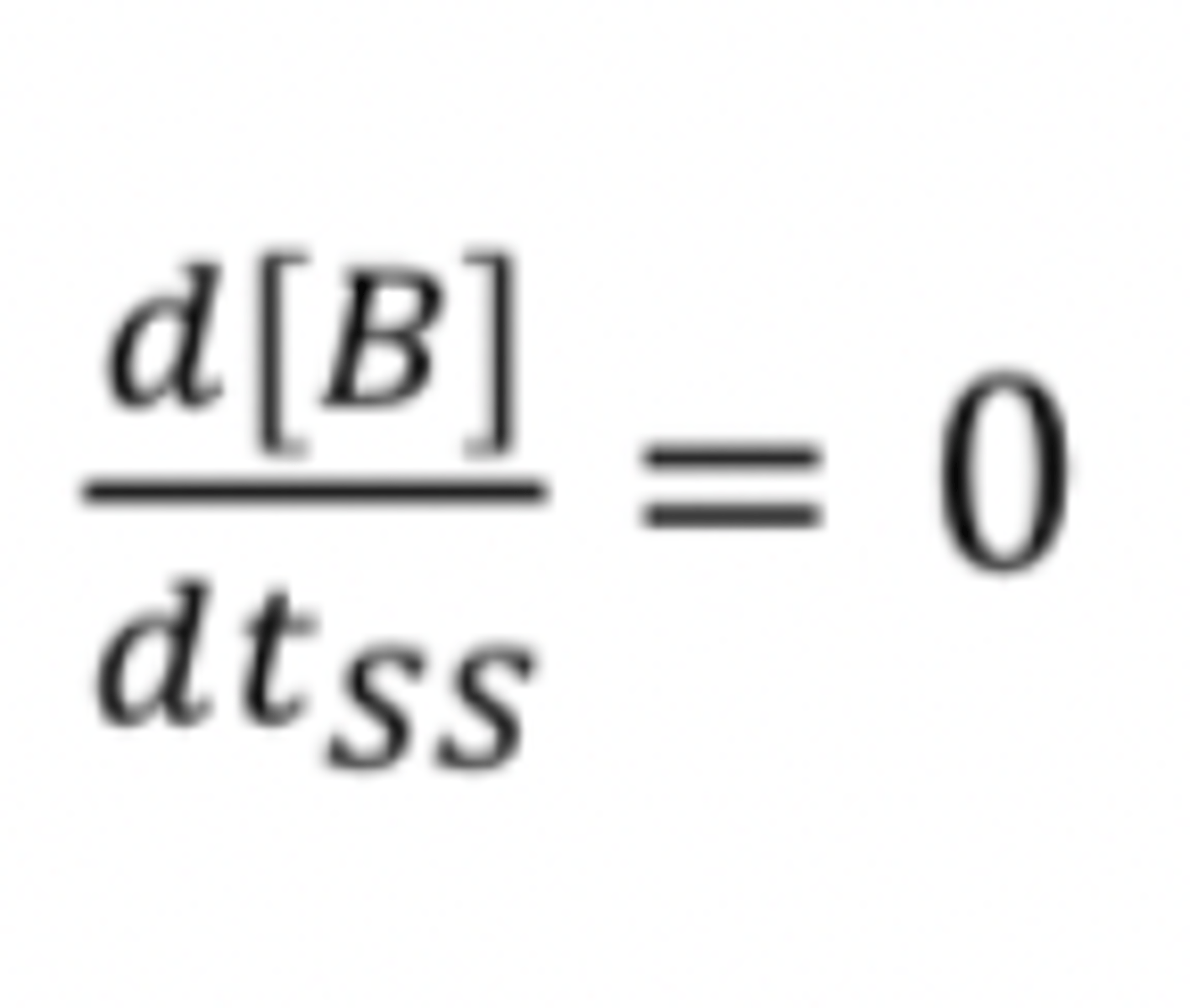
what does the SSA simplify
The steady state approximation simplifies solving the differential equations (it removes the timedependence for the intermediates)
solving SSA

limitations of SSA
SSA simplifies solving rate equations but must be applied carefully
Not suitable for intermediates that decay or accumulate during a reaction
Primarily used for high energy reactive intermediates
Rate laws derived using SSA are valid only after the reaction reaches steady state, not at the reaction's start
how to solve SSA recipe
1. Write down SSA for reactive intermediates.
2. Solve steady state equations to give concentrations of intermediates in terms of reactantsand products
3. Write equation for overall rate using solved SSA equations
what are enzymes
Enzymes are biological catalysts that speed up chemical reactions.
features of enzymes
Highly efficient and selective in catalyzing reactions
enzyme specificity
Enzymes are highly specific in terms of reactions they catalyse and substrates they work with
how do enzymes work
Enzymes work by lowering the energy of the transition state of the reaction. I.e. they stabilise the transition state (if anything is stabilised they lower the energy).
what do enzymes form
Enzymes form an 'Enzyme Substrate' complex where the substrate binds to the enzyme's active site
This has catalytic groups which form non-covalent interactions to stabilise the transition state (lowerits energy)
what is the active sites shape
The active site is Complimentary shape to the substrate
lock and key hypothesis
The active site is a "cleft" in the enzyme. It usually only makes up a small fraction of the protein- the rest of the structure acts as scaffolding.
It has a very precise three-dimensional structure that gives it the high specificity for reactions and substrates.
what other type of fit can an enzyme have
an induced fit
SSA for enzyme reactions

overall rate equatin for enzyme reactions
𝑉 = 𝑘2[𝐸𝑆]
![<p>𝑉 = 𝑘2[𝐸𝑆]</p>](https://knowt-user-attachments.s3.amazonaws.com/f9d1fc20-bc57-40a6-9d6f-206d6fe2d7aa.png)
what is the fastest rate equation
Vmax = k2[E]T
![<p>Vmax = k2[E]T</p>](https://knowt-user-attachments.s3.amazonaws.com/dcd374fc-3096-49fa-96c6-f6a1308abccb.png)
lineweaver-burke plot
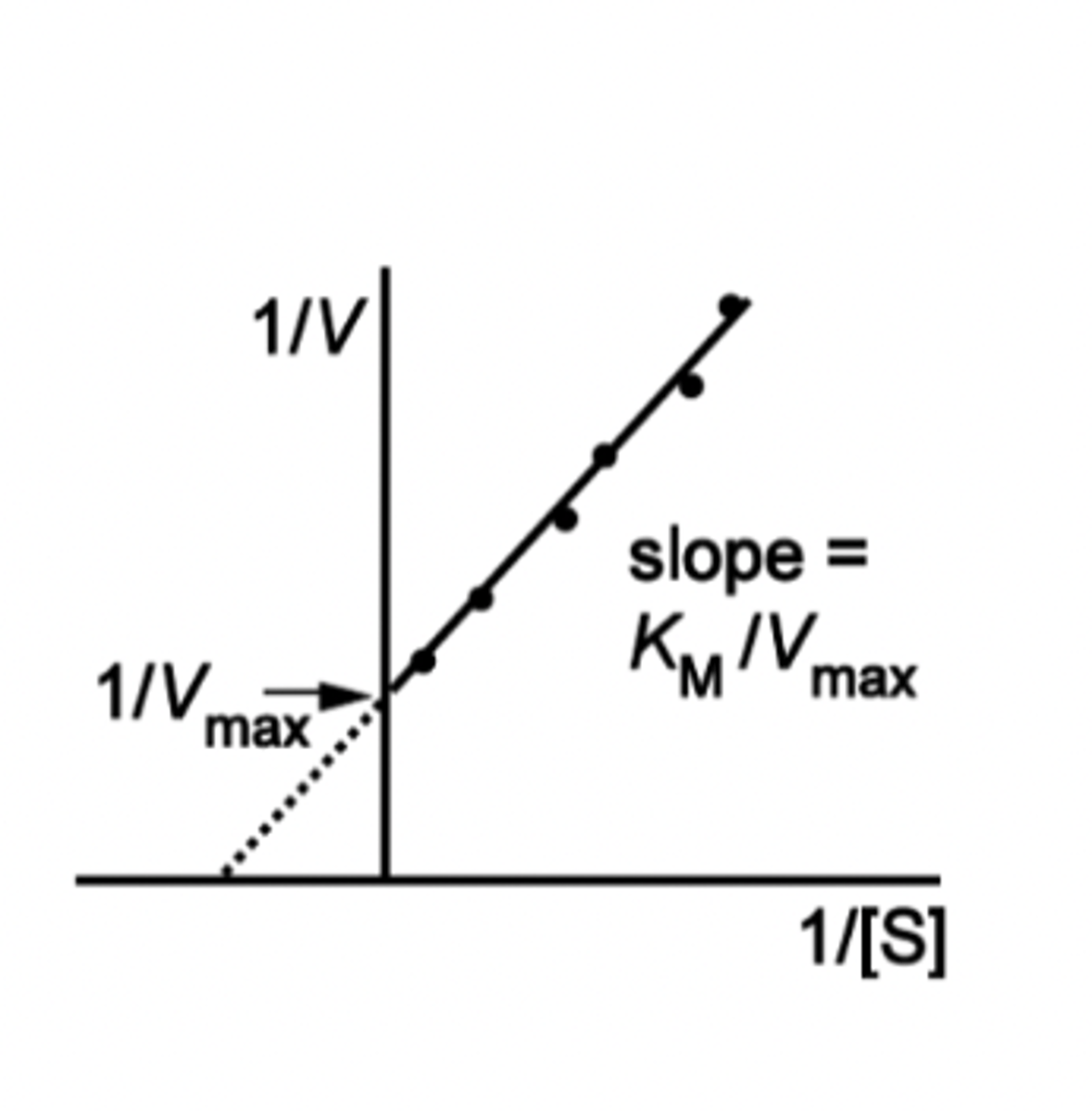
equation if given V vs substrate concentration
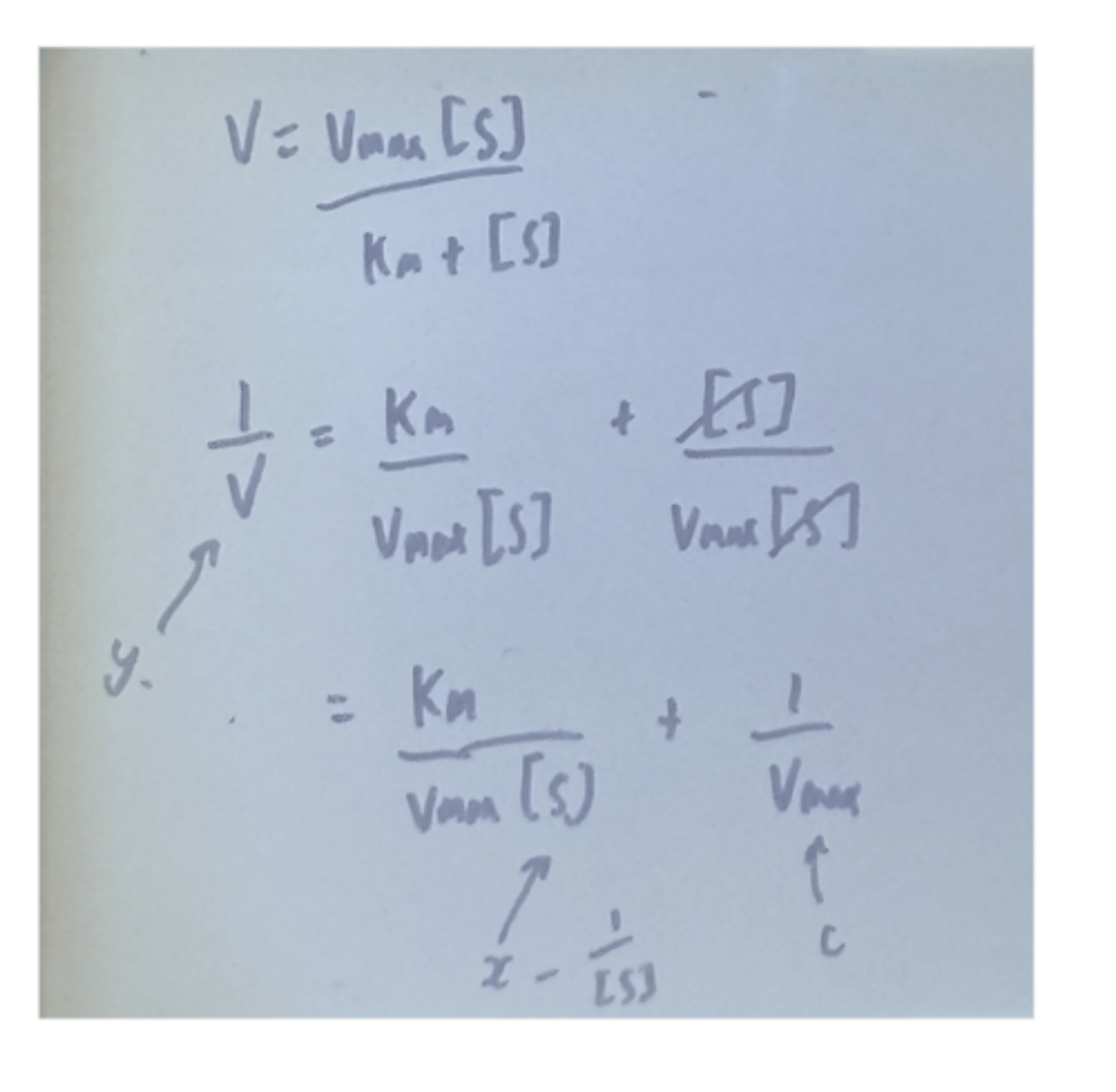
intepretation of values KM, Vmax
KM is the concentration at which half the active sites are filled- i.e. half the rate!
A high KM value represents a weak binding- need higher [S] to occupy half the active sites. A low KM means strong binding.
Vmax is the maximum rate for the reaction- this is the "turnover" number of the enzyme