6.2.3 Polyesters and polyamides
1/16
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
17 Terms
condensation polymerisation
formed from reactions between monomers that contain at least 2 functional groups
the functional group on each each monomer react forming links between repeat units
a small molecule is eliminated each time a link forms
tyes of condensation olymers
olyster
olyamides
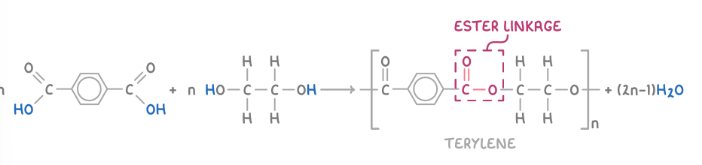
2 methods to form olyesters
dicarboxylic acid and a diol
one monomer with one end carboxyl other end alcohol
polyester formation examle from benzene-1,4-dicarboxylic acid and ethane-1,2-diol
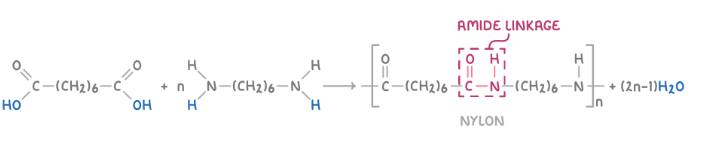
2 methods to make olymadies
dicarboxylic acid ad diamine
one end cooh other end NH2
formation of olymer examle from hexanedioic acid and 1,6-diaminohexane
acid hydrolysis of olyesters
alcohol and carboxylic acid
base hydrolysis of olyesters
alcohol and carboxylate salt
acid hydrolysis of olyamides
ammonium salt and carboxylic acid
base hydrolysis of olyamides
amine,carboxylic acid
how to deduce the monomers that form condensation olymers
Identify the amide (HN-CO) or ester (CO-O) linkage between repeat units.
Cut the link in the middle to separate the polymer chain.
Add an H atom or -OH group to both ends of the molecules. H atoms are added to N or O atoms, and -OH groups to C atoms
how to draw reeat units from condensation monomers
Place the structures of the two monomers side by side.
Remove an H atom from one monomer (e.g., diol or diamine) and an OH group from the other (e.g., dicarboxylic acid), forming a H2O molecule as a by-product. For diacyl chlorides, remove Cl instead of OH, forming HCl.
Connect the monomers through either an amide or ester linkage, depending on their functional groups.
To indicate continuity in the polymer chain, remove another H atom and OH/Cl group from the opposite ends.
monomers used addition vs condensation olymers
addition alkenes
condensation Carboxylic acids, acyl chlorides, amines, alcohols, amino acids
olymer chain used addition vs condensation olymers
addition Continuous chain of carbon atoms
condensation Contains ester or amide bonds
roducts formed addition vs condensation olymers
addition olymer only
condensation olymer and small molecule
what are olyesters and olyamdies used fro
fabrics
why are polyesters biodegradable
ester bond can be hydrolysed