AS organic
1/208
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
209 Terms
name the 6 ways organic compounds can be represented:
empirical formula
molecular formula
general formula
structural formula
displayed formula
skeletal formula
what is the empirical formula of a compound?
formula that gives the simplest ratio of atoms of each element in a compound
what is the molecular formula of a compound?
formula which gives the actual number of atoms of each element in one molecule
what is the general formula of a compound?
formula which shows the number of atoms of each element in a substance which has n carbon atoms
what is the structural formula of a compound?
formula which shows how the atoms are joined together in a molecule
what is the displayed formula of a compound?
formula which shows all bonds and atoms in a molecule
(does not show accurate bond angles)
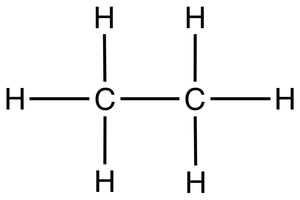
what is the skeletal formula of a compound?
simplified displayed formula:
H atoms not shown (unless part of a functional group)
C atoms represented by corners
double bonds shown
other atoms shown
what is a homologous series?
a series of compounds containing the same functional group
what are the characteristics of a homologous series?
same general formulae
same functional group
each subsequent compound differs by a CH2 unit, forming next homologue in series
give the IUPAC rules for nomenclature:
find and number longest C chain to determine stem name (e.g. but-)
identify key functional group (e.g. -oic acid)
add side chains in alphabetical order and w/ correct numbers (indicating which C atom side chains are on), adding multipliers if necessary (e.g. 3-chloro, 2,2-dimethyl)
→ 3-chloro-2,2-dimethylbutanoicacid
(molecules that contain rings have the prefix cyclo-)
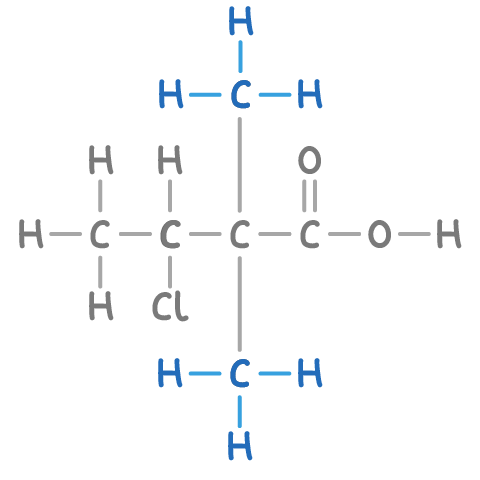
what is the functional group of a molecule?
the part of a molecule that determines how it reacts and is the same across the homologous series
what is a structural isomer?
a molecule w/ the same molecular formula but different structural formulae
what are the 3 types of structural isomer?
functional group isomers
chain isomers
position isomers
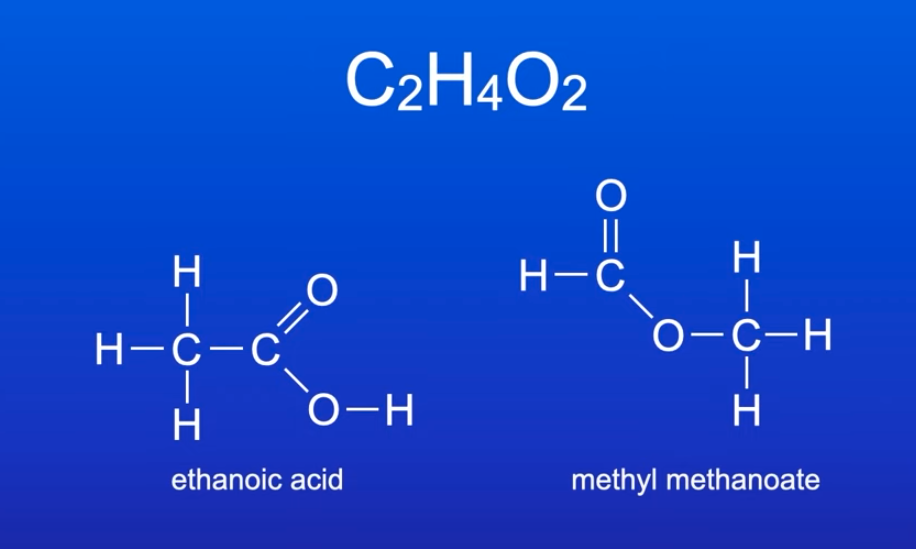
what is a functional group isomer? give an example:
structural isomers w/ same molecular formula but different functional groups
e.g. ethanoic acid and methylmethanoate
what is a chain isomer? give an example:
structural isomers w/ same molecular formula but different carbon chain arrangements
e.g. butane and methylpropane
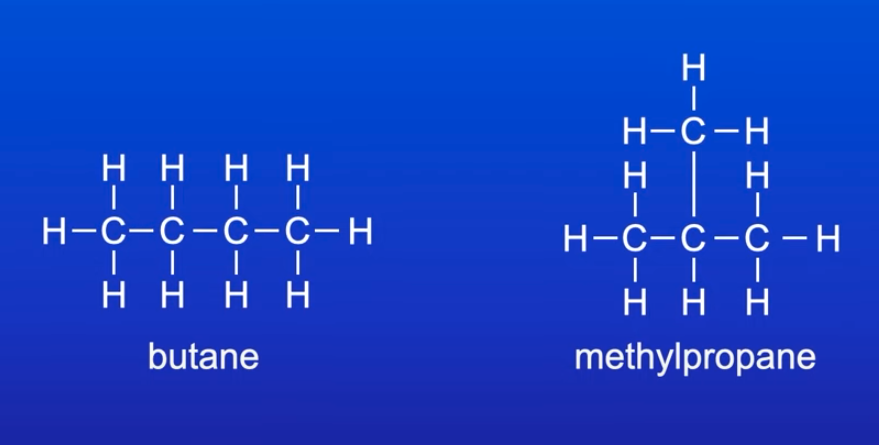
what is a position isomer?
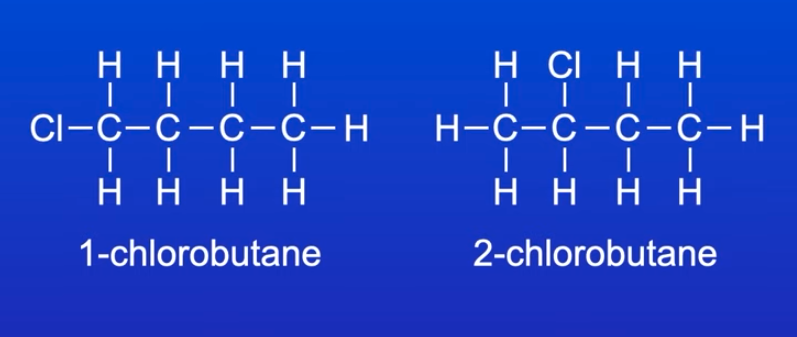
structural isomers w/ same molecular formula but different position of functional group
e.g. 1-chlorobutane and 2-chlorobutane
what is a stereoisomer?
molecules that have the same molecular formula but a different arrangement of the atoms in space
when does E/Z / geometric isomerism occur?
when you have restricted rotation about the planar C=C
(both atoms of C=C must have 2 or more different functional groups)
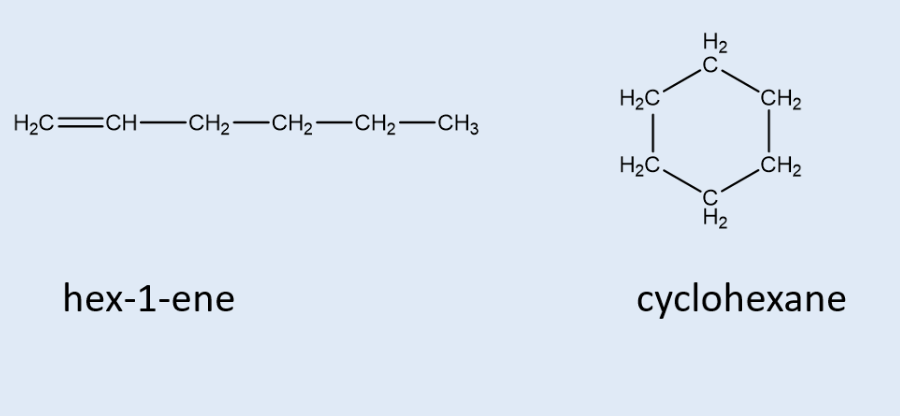
does this but-2-ene display E or Z isomerism? why?
E (entgegen) as the higher priority functional groups are opposite each other
does this but-2-ene display E or Z isomerism? why?
Z (zusammen) as the functional groups are together
explain the Cahn-Ingold Prelog (CIP) rules:
we look at the atomic numbers of the atoms directly connected to the C=C and give them a ‘priority’
highest values opposite = E
highest values together = Z
what is geometric isomerism?
compounds/molecules w/ the same structural formula
but atoms/bonds/groups arranged differently in space
what is stereoisomerism? give 2 examples:
compounds/molecules w/ the same molecular and same structural formula but a different arrangement of atoms in space:
geometric isomerism
optical isomerism
what are structural isomers? give 3 examples:
compounds/molecules with the same molecular formula but a different structural formula:
chain isomerism
position isomerism
functional group isomerism
what are the 3 types of structural isomerism?
chain isomer
position isomer
functional group isomer
whata chain isomer?
compounds/molecules w/ the same functional group and molecular formula but a different arrangement of the carbon chain
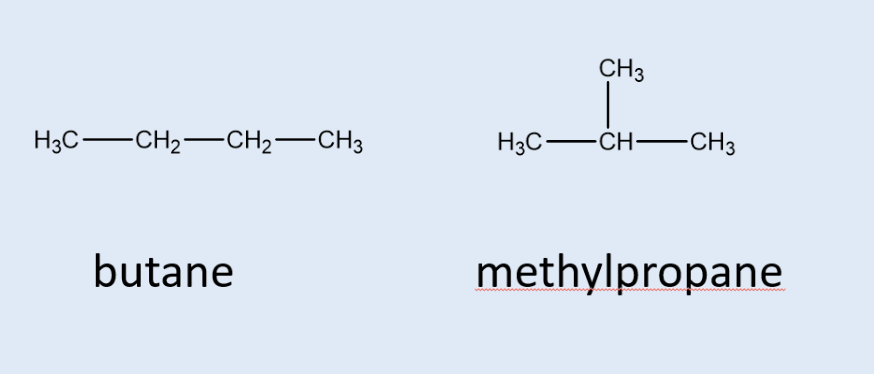
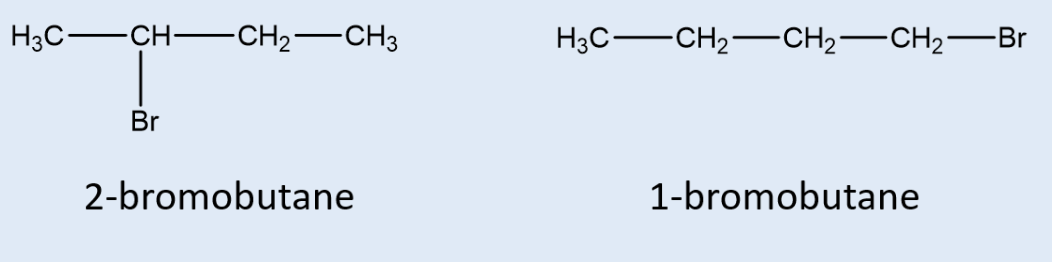
what is a position isomer?
compounds/molecules w/ the same molecular formula and carbon chain but w/ the functional group in a different place
what are functional group isomers?
compounds/molecules w/ the same no. of atoms but arranged to form a diff functional group
why does E-Z isomerism occur?
restricted rotation around the C=C double bond
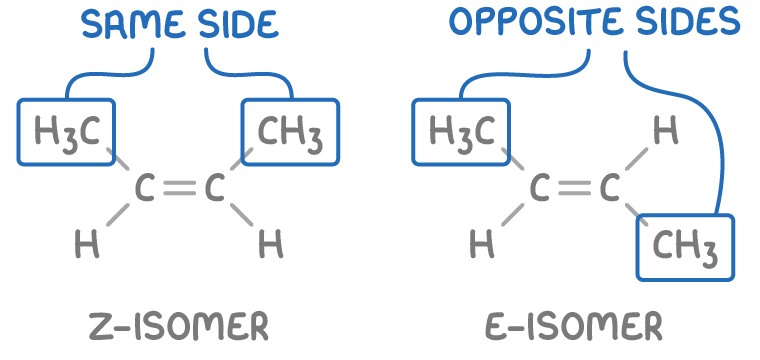
give the 2 types of E/Z isomers:
Z isomer (zusammen) = higher priority groups are on the same side of the C=C double bond
E isomer (entgegen) = higher priority groups are on opposite sides of the C=C double bond
summarise the CIP priority rules:
C w/ the groups w/ the highest atomic number = highest priority

what are alkanes?
homologous series of saturated hydrocarbons w/ the general formula CnH2n+2
how reactive are alkanes?
very unreactive, but do burn and react w/ halogens (to form haloalkanes)
what is a homologous series?
family of compounds:
same general formula
similar chemical properties
each member differs by the addition of a CH2 group and a gradual change in physical properties
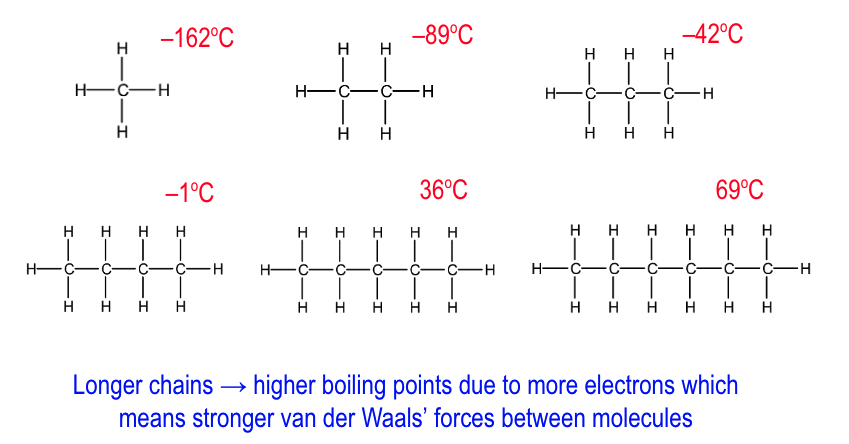
how does bpt change as the C chain gets longer?
longer C chain = higher bpt
as more e-
so stronger VDWs between molecules
which require more energy to break
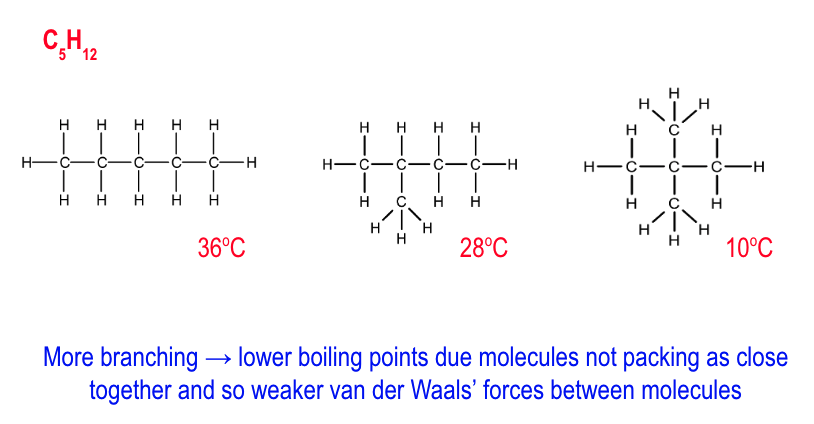
how does bpt change w/ increase in branching?
more branching = lower bpt
as molecules do not pack as close together
so weaker VDWs between molecules
which require less energy to break
how does viscosity change as the C chain gets longer?
viscosity increases (can be found by residue in fractional distillation)
how does flammability change as the C chain gets longer?
flammability decreases
how does the flame change as the C chain gets longer?
flame gets dirtier (clean → smoky flame)
what can the products of combustion be?
complete: CO2 + H2O
incomplete: CO + H2O or C + H2O
which pollutants does an internal combustion engine produce?
NOx, CO, C and unburned hydrocarbons
what is the function of a catalytic converter?
removes toxic gaseous pollutants from internal combustion engines and converts them into less harmful substances (e.g. CO2, water vapour, N)
how is CO2 formed in the atmosphere? what potential problems may this cause?
complete combustion of C in fuel
greenhouse gas causing global warming
how is CO formed in the atmosphere? what potential problems may this cause?
incomplete combustion of C in fuel
toxic
how is C formed in the atmosphere? what potential problems may this cause?
incomplete combustion of C in fuel
blackens buildings, global dimming
how is SO2 formed in the atmosphere? what potential problems may this cause?
combustion of S in fuel (through fuel impurities)
acid rain, air pollution
how are NO and NO2 formed in the atmosphere? what potential problems may this cause?
reaction of N2 in air w/ O2 in air at very high temperatures (e.g. engines/furnaces)
acid rain
what potential problems may unburned fuel cause?
fuel waste, harmful greenhouse gas
how can we reduce SO2 emissions?
flue gas desulfurisation:
powdered CaO or CaCO3 mixed w/ water to make an alkaline slurry
when flue gases mix w/ alkaline slurry, acidic SO2 gas reacts w/ calcium compounds to form CaSO3, which is harmless
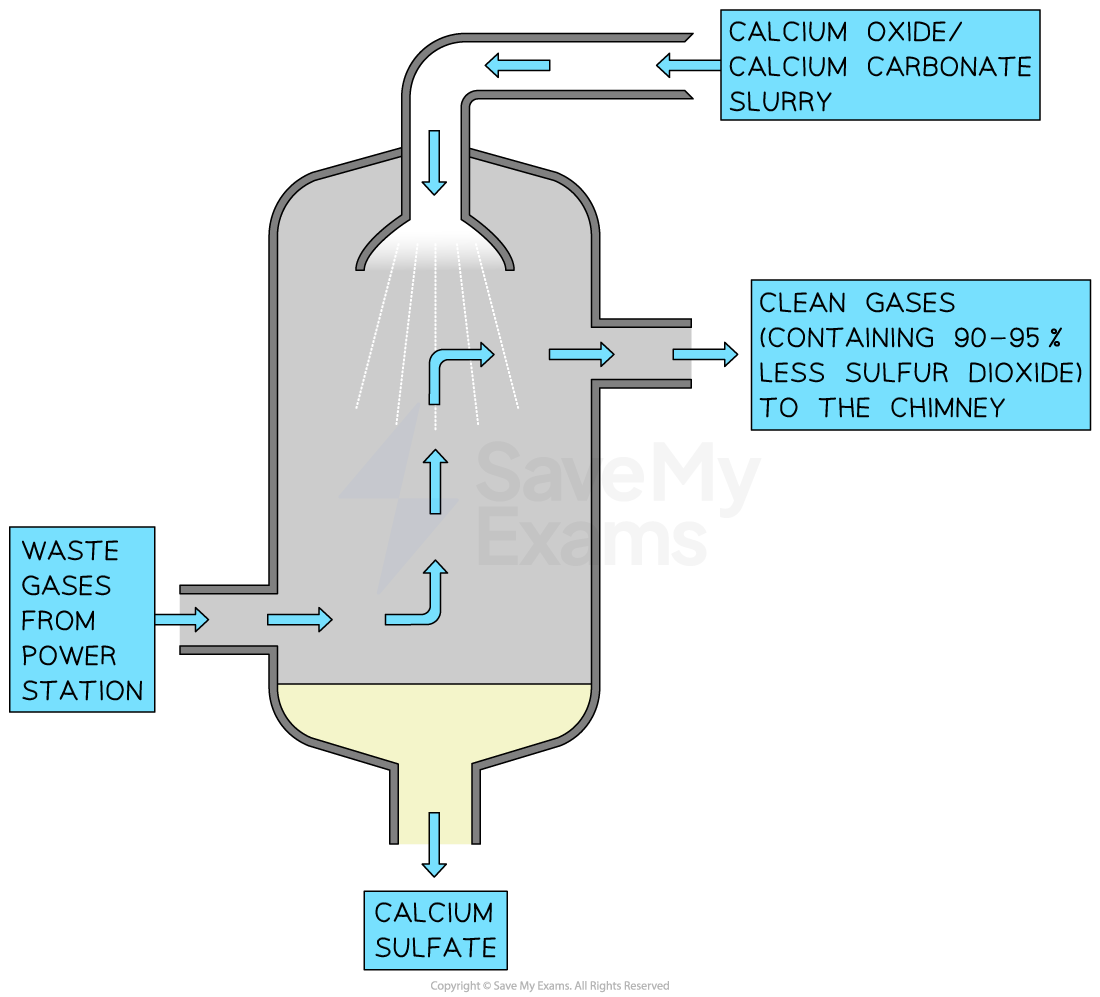
describe the process of fractional distillation:
crude oil vaporised
vapour passed into a column - hot at bottom and cool at top (temp gradient)
as the vapour rises it cools
molecules condense at different fractions as they have different bpts
larger the molecules, the lower down the column they condense
what is thermal decomposition? give an example:
breaking down compounds by heating into simpler compounds - e.g. cracking
what is cracking?
breaking up large, less useful hydrocarbons into smaller, more useful hydrocarbons (thermal decomposition of alkanes)
why do we crack?
petroleum fractions w/ shorter C chains are in higher demand than larger fractions
longer hydrocarbons are cracked to make use of XS larger hydrocarbons and supply demand for shorter ones
products are more valuable than the starting materials
give a use of shorter alkanes:
fuels
give a use of alkenes:
making plastics
describe the conditions and products of thermal cracking:
high temp (900oC)
high pressure (70 atm)
no catalyst
produces alkenes
describe the conditions and products of catalytic cracking:
high temperature (450oC), but lower than thermal cracking
slight pressure (1-2 atm)
zeolite catalyst
produces motor fuels, aromatic hydrocarbons, cyclic hydrocarbons and branched alkanes
what is a haloalkane?
an alkane where at least 1 H atom has been replaced w/ a halogen atom
what is the product of free rad sub?
haloalkanes
what is a mechanism?
process showing the step by step movement of e- as reactants are converted into products
what does a single headed curly arrow represent?
movement of a single e-

what does a double headed curly arrow represent?
movement of an e- pair
what is a free radical?
a molecule w/ an odd no. of e- and so an unpaired e-
how reactive are free radicals?
very reactive!
name the steps of free rad sub:
initiation
propagation
termination
give the overall eqn for the reaction of methane w/ chlorine:
CH4 + Cl2 → CH3Cl + HCl
describe and give the eqn for the initiation step of free rad sub of the reaction of methane w/ chlorine:
UV light breaks Cl molecules into free radicals by providing the E to break the Cl-Cl bond
Cl : Cl → . Cl + . Cl
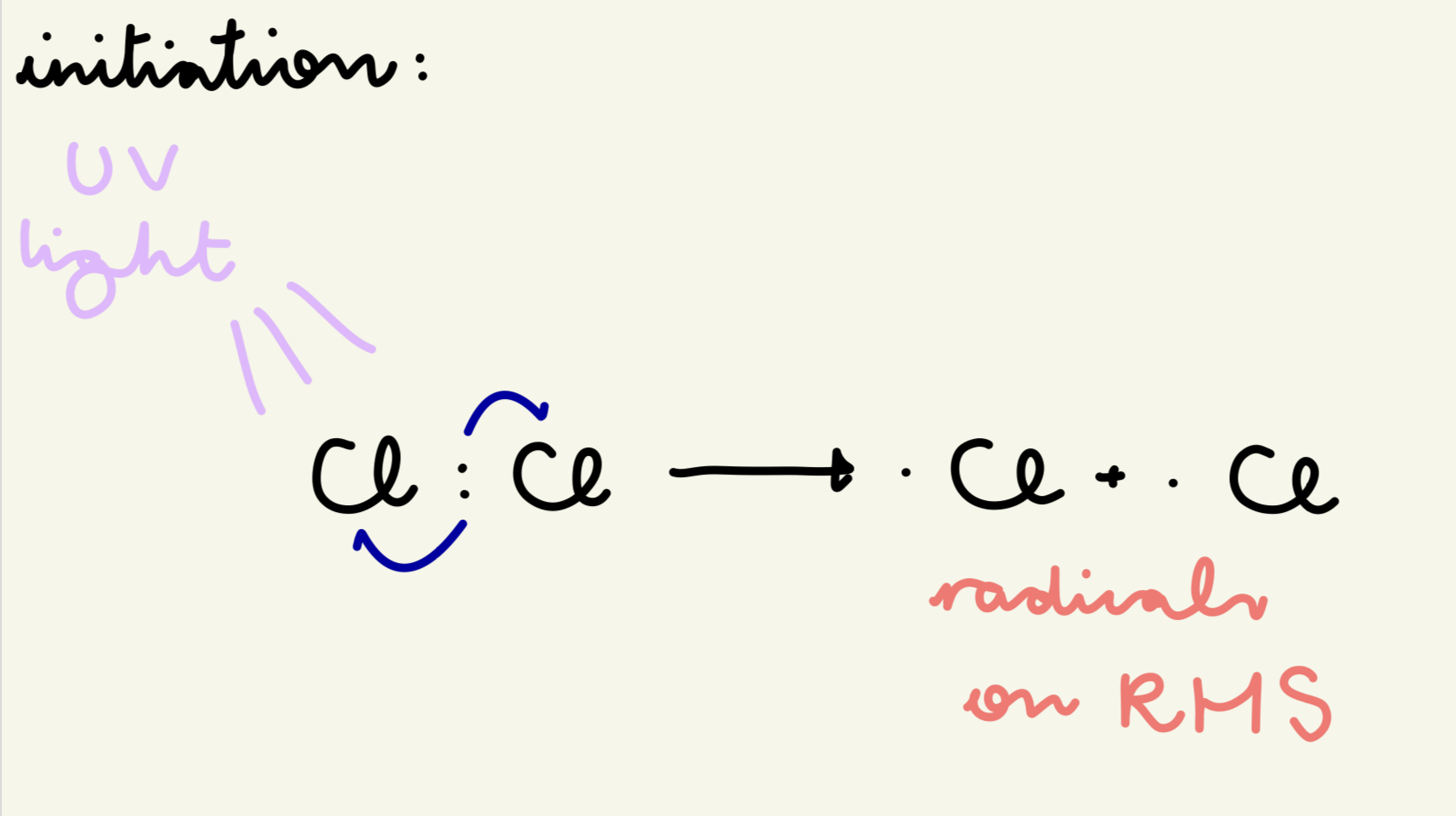
which side are the radicals on in initiation?
RHS
describe and give the eqn for the propagation step of free rad sub of the reaction of methane w/ chlorine:
Cl radicals react w/ alkanes in a chain reaction where the Cl radical acts as a catalyst
for each H replaced, there is a pair of propagation steps
. Cl + CH4 → HCl + . CH3
. CH3 + Cl2 → CH3Cl + . Cl
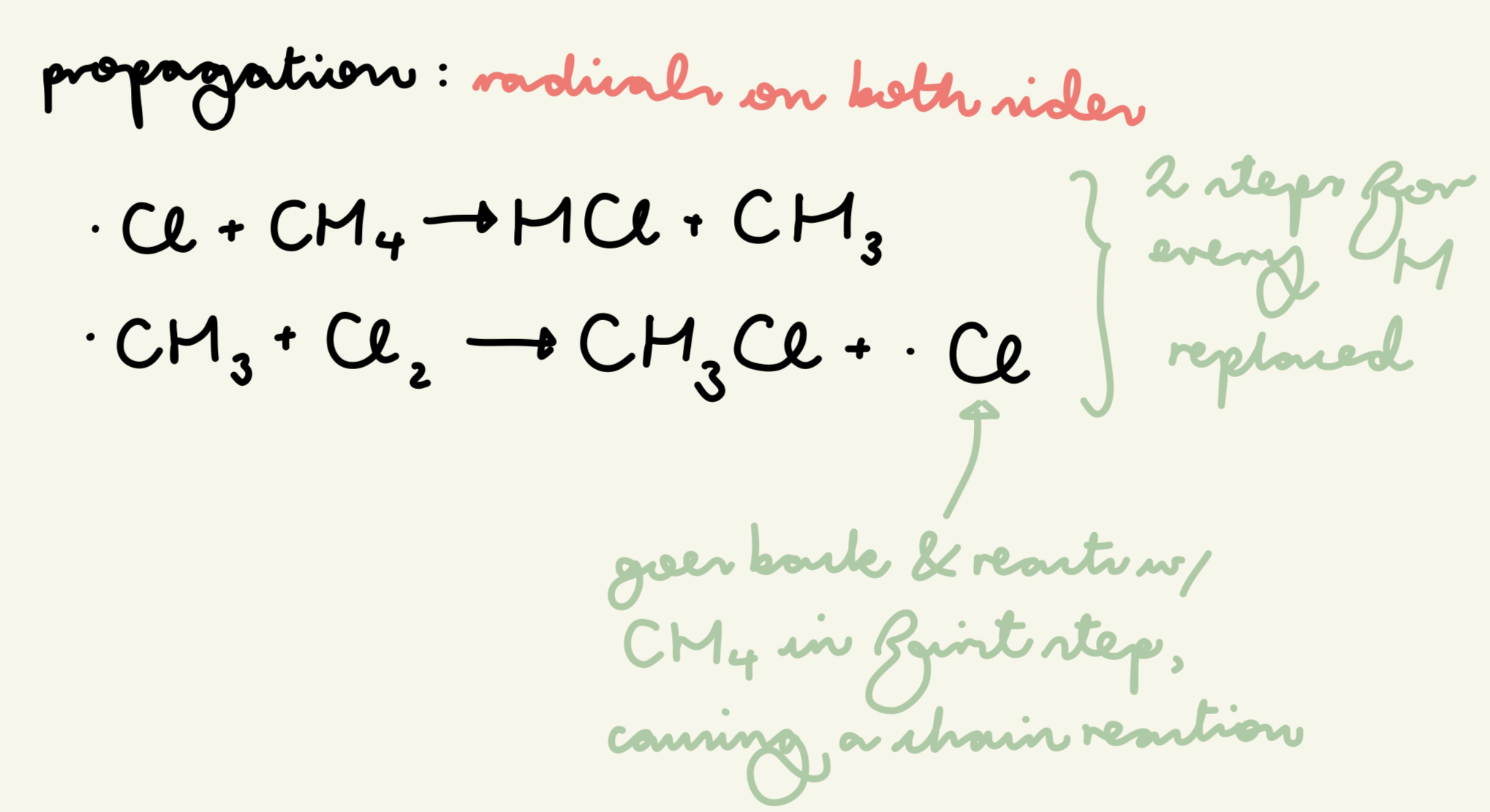
which side are the radicals on in propagation?
both sides
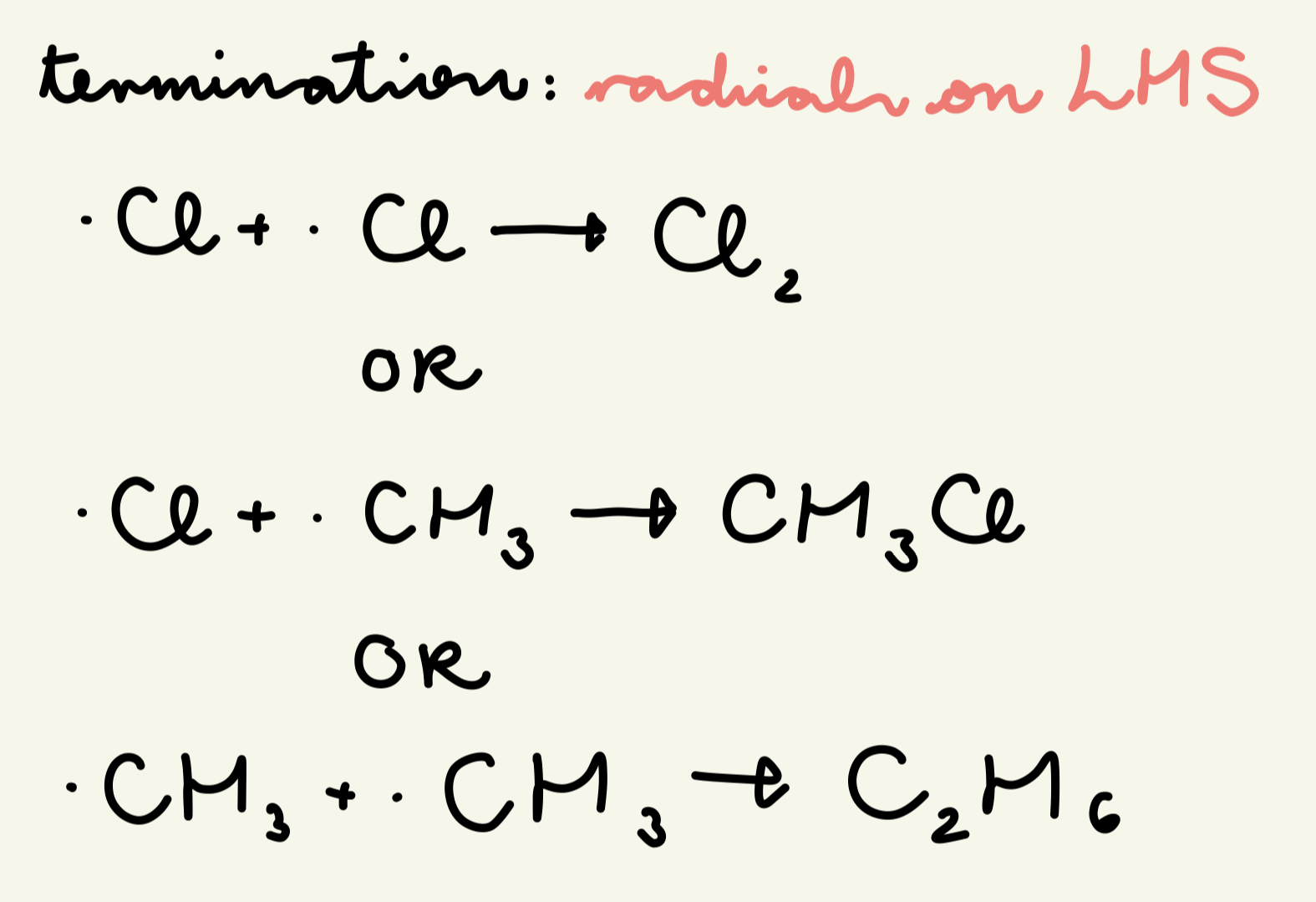
describe and give the eqns for the termination step of free rad sub of the reaction of methane w/ chlorine:
when any 2 free radicals meet, they react to form a molecule w/ no unpaired e-, stopping the chain reaction
. Cl + .Cl → Cl2
OR . Cl + . CH3 → CH3Cl
OR . CH3 + . CH3 → C2H6
what is an alkene?
an unsaturated hydrocarbon - C=C double bond is an area of high e- density
what is a polymer?
long chain molecule made from lots of small molecules (monomers) joined together
what is a monomer?
small molecules that join together to make polymers
what is addition polymerisation?
formation of long chain molecules from lots of small molecules joining together w/ no other products
draw the monomer structure of poly(ethene):

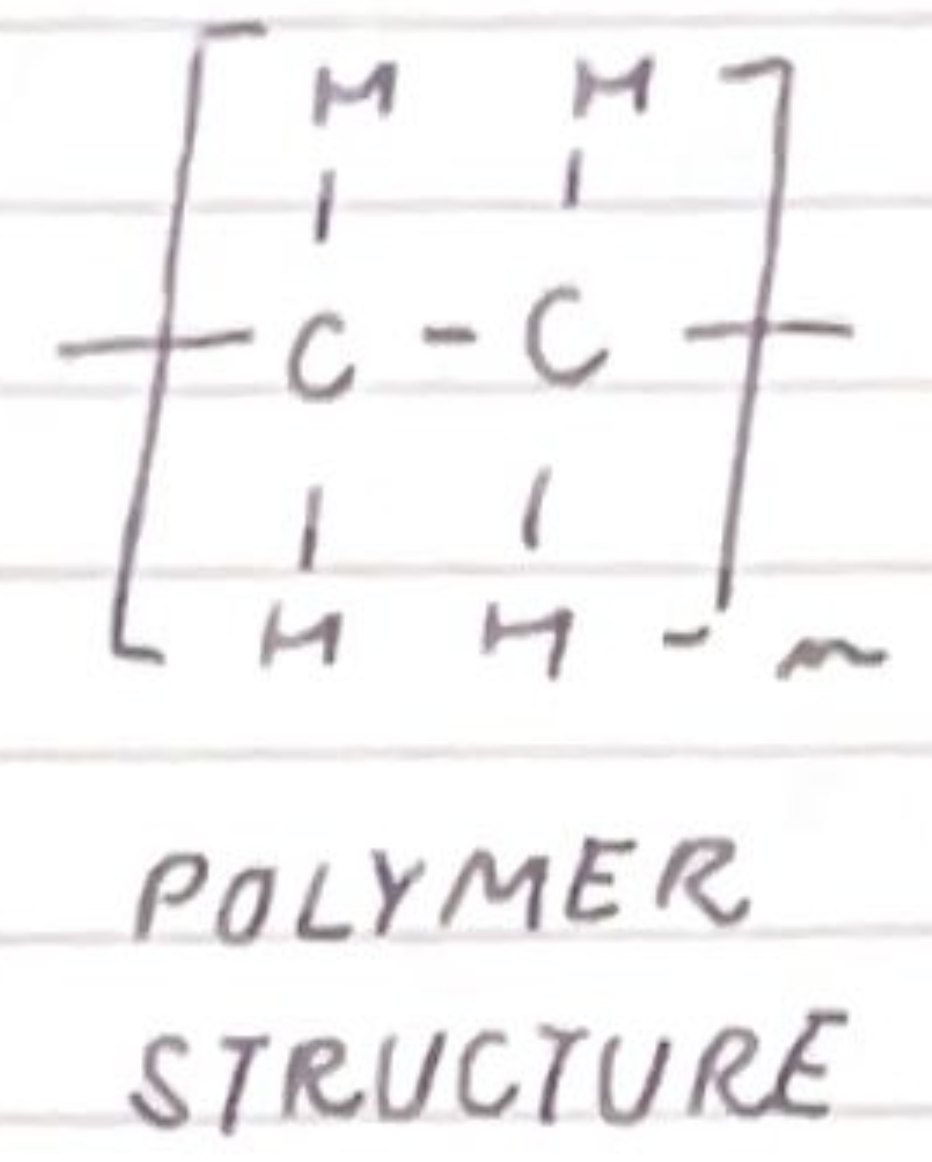
draw the polymer structure of poly(ethene):
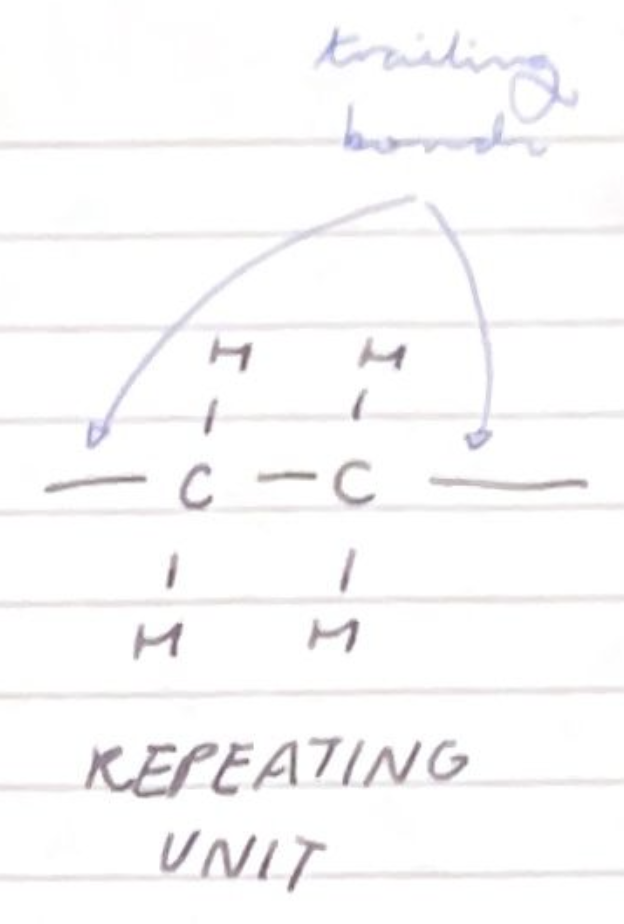
draw the repeating unit of poly(ethene) and name the bonds at either end of the repeating unit:
trailing bonds
how do we name addition polymers?
poly(name of alkene)
what are addition polymers formed from?
alkenes and substituted alkenes
describe the properties of addition polymers:
unreactive
chains usually non-polar so held together by VDWs
the longer the polymer chains are/the closer together the chains can get, the stronger the VDWs between the chains will be
polyalkenes made up of long, straight chains → strong and rigid
polyalkenes made up of short, branched chains → weaker and flexible
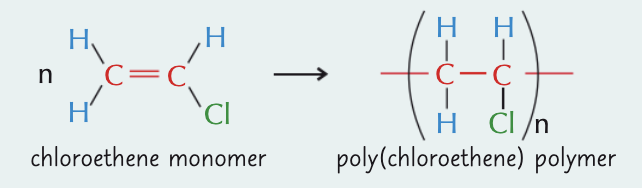
what is poly(chloroethene)/PVC and what can it be used for?
polymer of chloroethene
has long, closely packed polymer chains - hard, but brittle at room temp
rigid PVC - drainpipes and window frames
how can we modify PVC’s properties?
by adding a plasticiser - pushes polymer chains apart, reducing strength of VDWs
this means the polymer can slide around more, making it easier to bend
plasticised PVC - electric cable insulation, flooring tiles, clothes
what is an electrophile?
e- deficient species which will accept a pair of e-
what is a carbocation?
+vely charged C atom w/ only 3 bonds, making it unstable
what does electrophilic addition involve? what does it form?
alkene → dihaloalkane
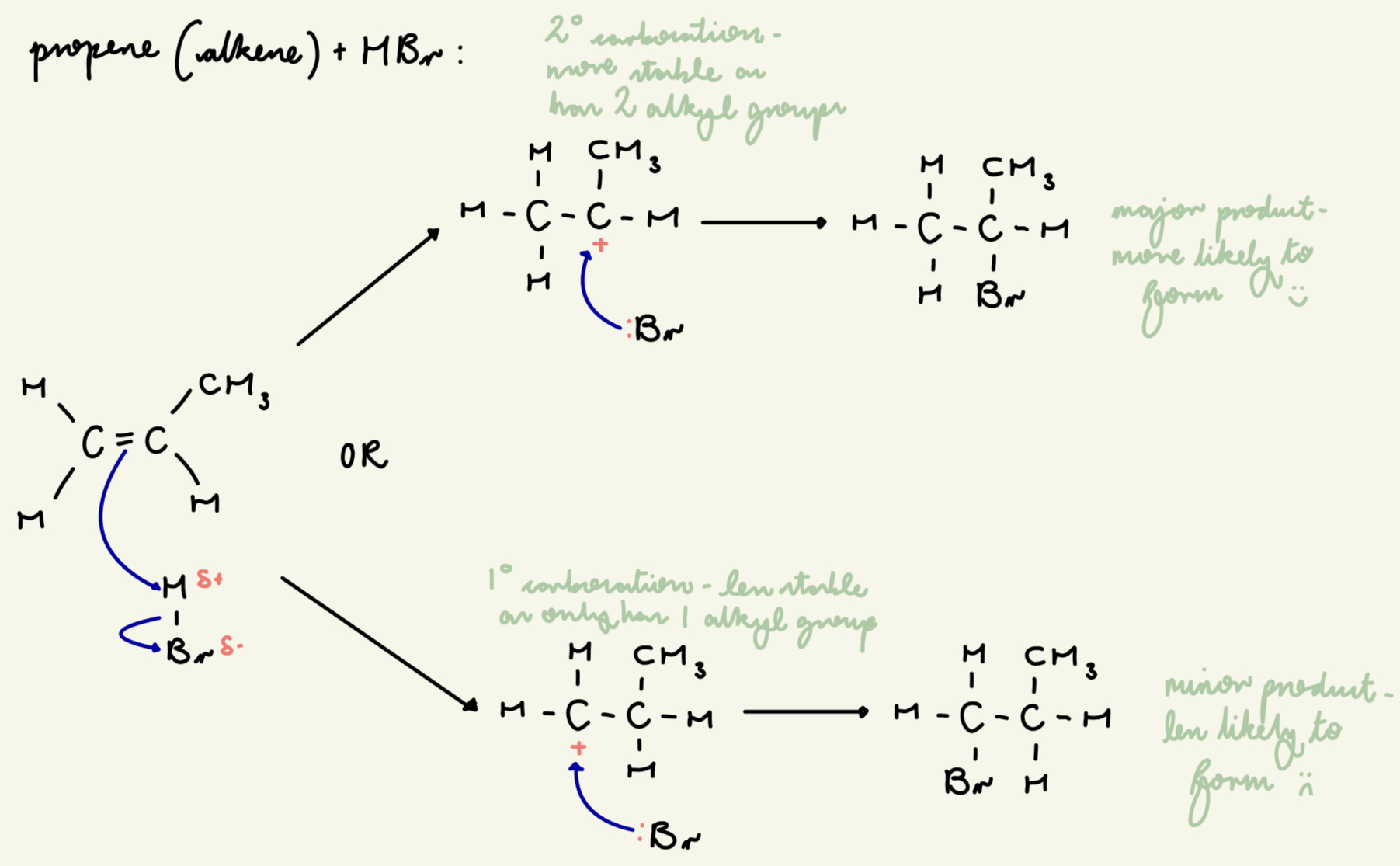
draw out the electrophilic addition mechanism between HBr and propene (for both the major and minor product):
draw out the electrophilic addition mechanism between Br2 and ethene:
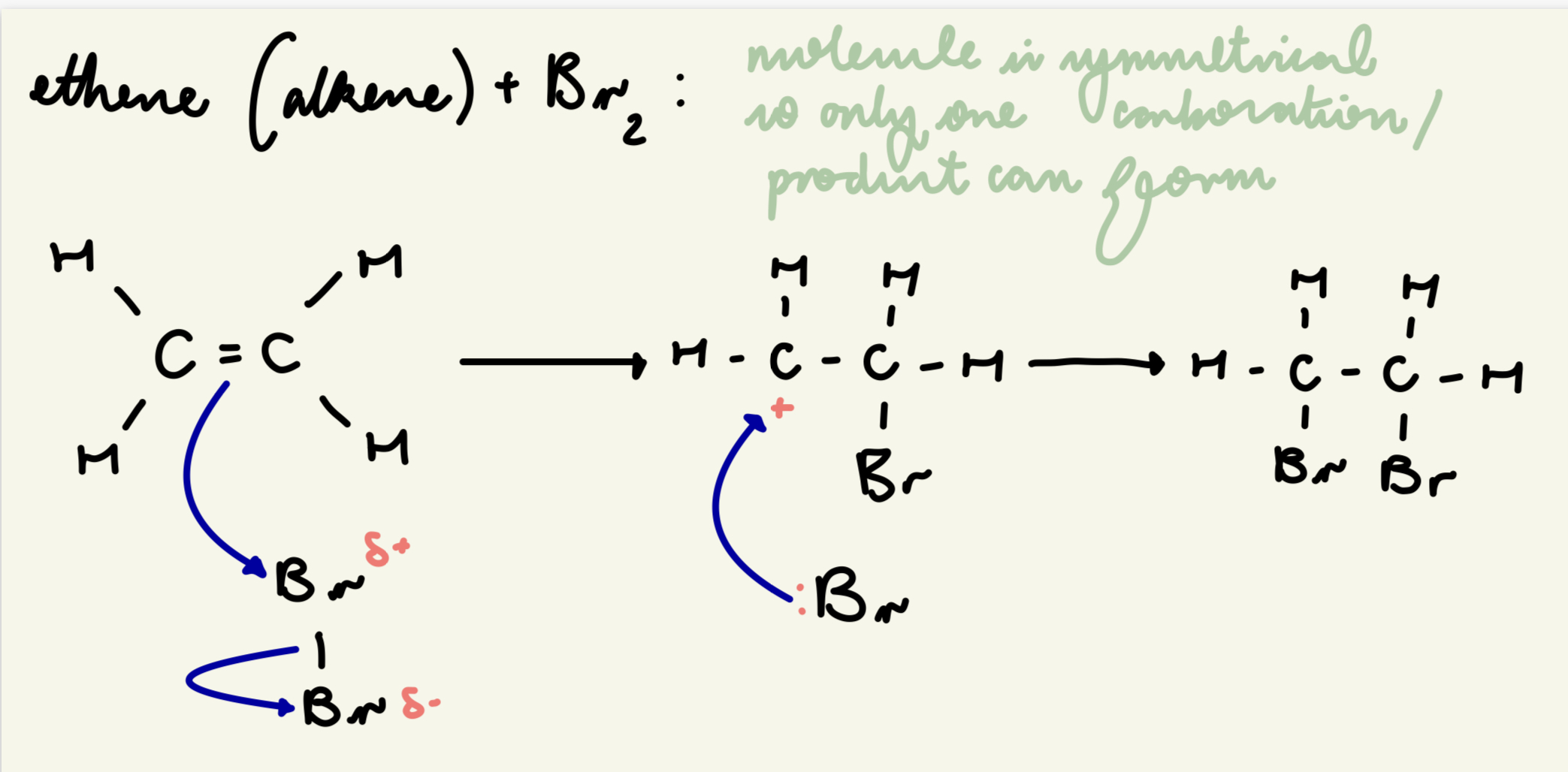
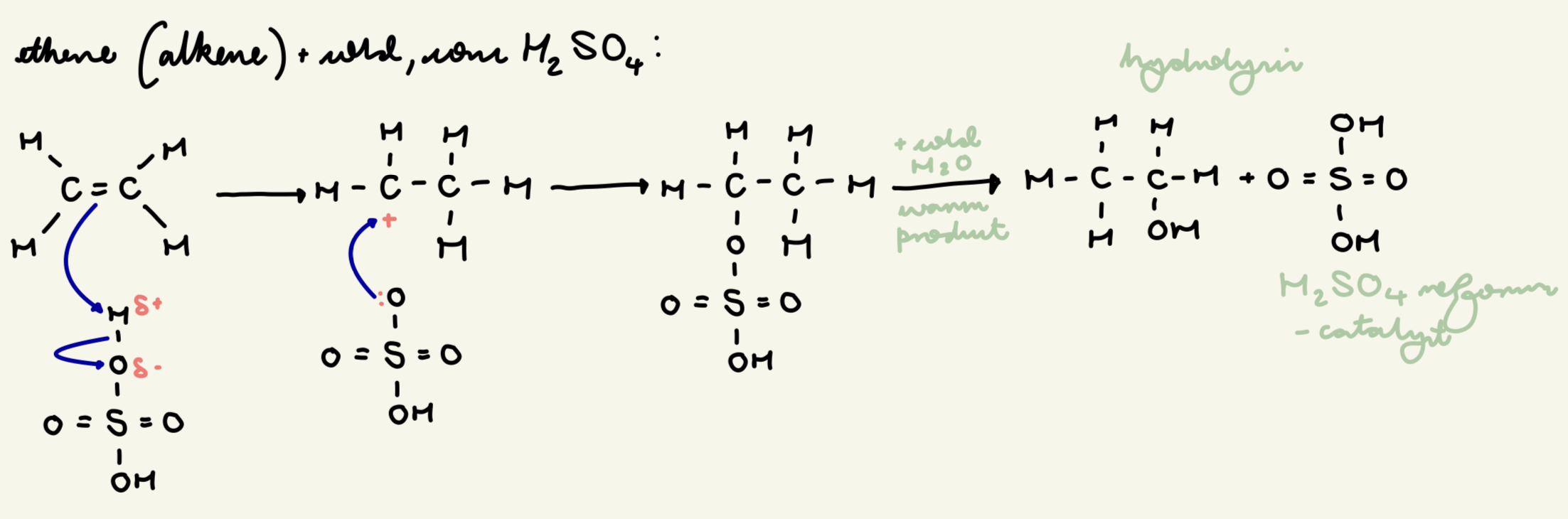
draw out the electrophilic addition mechanism between H2SO4 and ethene and state the conditions:
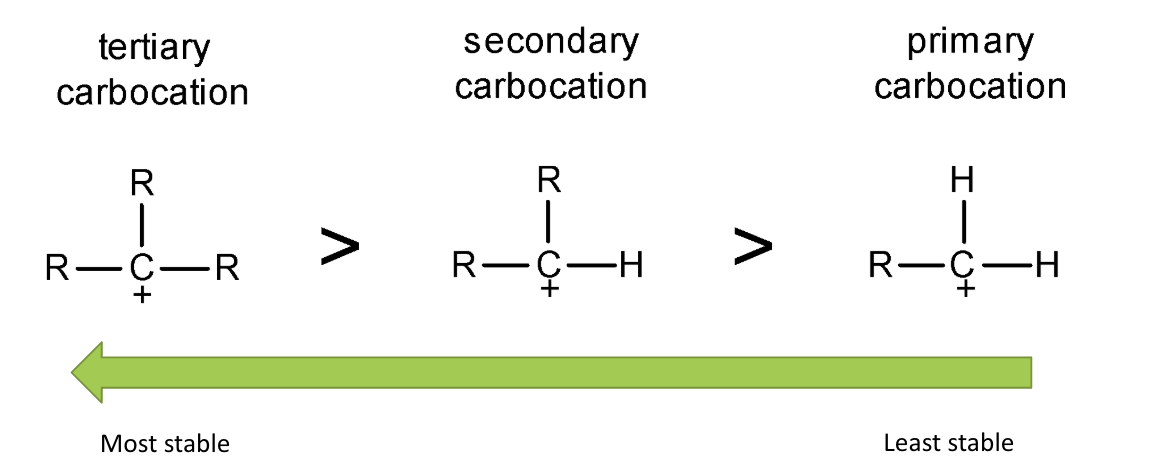
give the order of carbocation stability - why is this the case?
tertiary carbocations have the most stability as they have the greatest +ve inductive effect as they have the most alkyl/C groups
as they push e- density towards the carbocation
the more alkyl groups, the more stable due to +ve inductive effect
what is a primary carbocation?
carbocation w/ only 1 alkyl group
what is a secondary carbocation?
carbocation w/ 2 alkyl groups
what is a tertiary carbocation?
carbocation w/ 3 alkyl groups
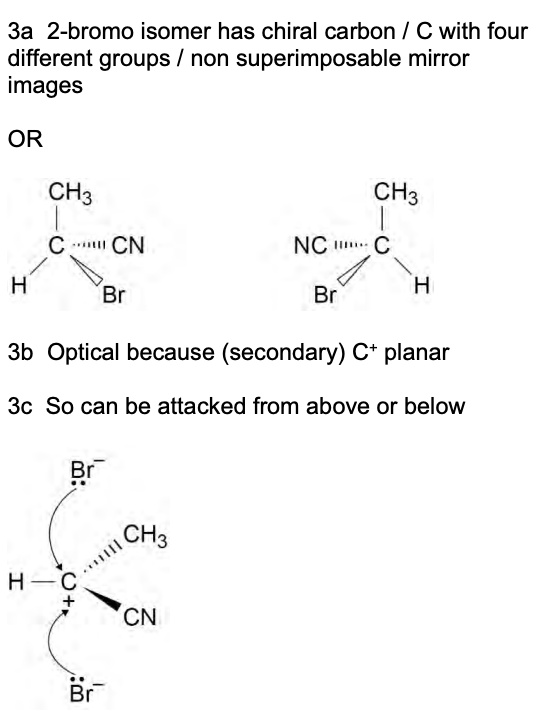
why may a product of electrophilic addition display optical isomerism?
electrophilic addition involving an alkene w/ one of the Hs substituted for a different functional group e.g. CN + HX
product is a haloalkane w/ one of the Hs subbed for the other functional group
one of the carbocations may form a product w/ a chiral C
optical as C+ on carbocation planar so can be attacked from above or below
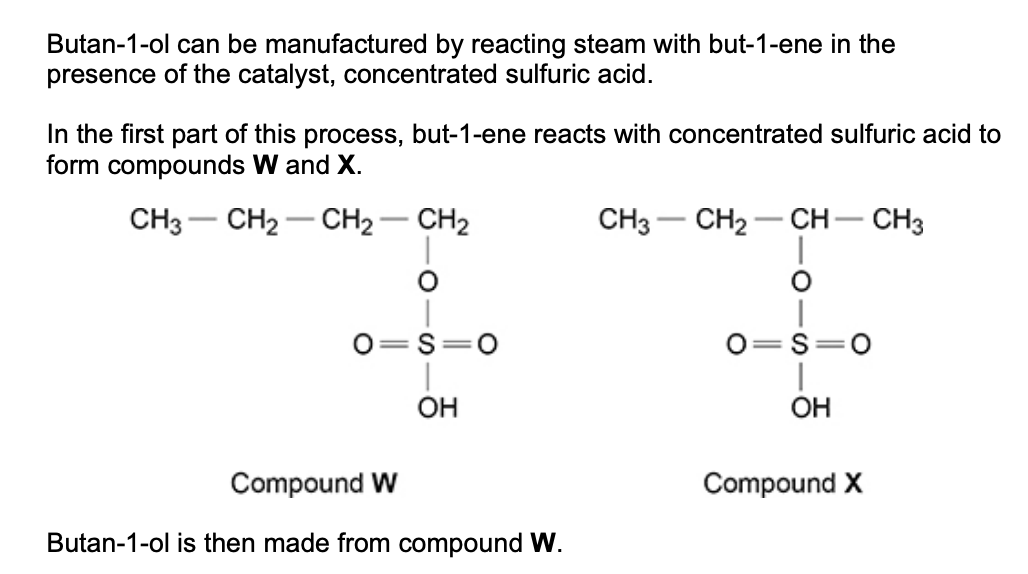
there is a very low yield of butan-1-ol from but-1-ene in this manufacturing process - explain why (2)
formed from less stable carbocation
formed from 1o rather than 2o carbocation
order the C-Br, C-I and C-Cl bonds in order of strength:
(strongest to weakest) C-Cl, C-Br, C-I
what are the 2 factors affecting bond strength? which one is more significant?
polarity
bond enthalpy (more significant)
explain why the C-halogen bond enthalpy influences the RoR:
C-halogen bonds get weaker down the group due to the increased size of the halogen atom
the further apart the atoms are in size, the higher the bond enthalpy
this means the C-F bond is strongest, meaning fluoroalkanes undergo nuc sub reactions more slowly than other alkanes
how is bond enthalpy affecting bond strength more significantly seen in the periodic table?
reactivity decreases down the group
if polarity were more significant, reactivity would increase down the group as the C-F would be predicted to be more reactive as the difference in electronegativity is the greatest
give the molecular formula for ozone:
O3