5. Periodicity // R3.2, S2.2-3
1/40
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
41 Terms

Alkali metals
Group 1
Metals
Very reactive — oxidation — Losing their outer electron to form the metal ion.
Good reducing agents
Reactivity increases down the group — outer electron is in higher energy levels and requires less energy to remove it
Stored under liquid paraffin to prevent them reacting with air
What do Alkali Metals react with
All react with H2O → Metal hydroxide alkaline solution + H2 (g)
React readily with Cl, Br, and I to form ionic salts
Li — floats and reacts quietly
Na — melts into a ball which darts around on the surface
K — heat generated from the reaction ignites hydrogen

Halogens
Group 17
Non metals
Very reactive (reduction) and replacement reactions
Good oxidizing agents
Create diatomic molecules
Halogens from F to I
Radius increases:
Reactivity decreases (outer shell is at increasingly higher energy levels + decreases the attraction for an extra electron)
Dispersion forces increase (bigger atom = more likely to create bigger dipole = stronger intermolecular forces)
Hydrides of the elements become more acidic (HF, HCl, …) as covalent bond becomes weaker
How do Halogens react
By gaining one more electron to form halide ions
Chlorine is a stronger oxidizing agent than bromine, but both are stronger than Iodine:
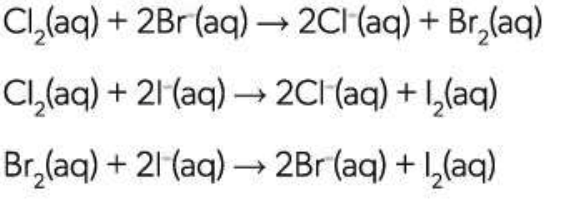
How to test for Halide Ions
Add silver nitrate solution
Ag+ (aq) + X- (aq) → AgX (s) —(Light)→ Ag (s) + (1/2)X2
X = Cl, Br, I
Silver ions react with the halide ions to form a precipitate of the silver halide.
The silver halides can be distinguished by their colour.
Basis of old-fashioned film photography.
AgCl = White
AgBr = Cream
AgI = Yellow
Nobel Gasses
Group 18; non metals, not acids nor bases, low reactivity, monoatomic, weak intermolecular forces (LDP)
Diatomic
Molecules composed of only two atoms, of the same or different chemical elements. E.g. H2 or O2 or CO
Group
Column in the PT. Elements in the same group have same number of valence electrons
Period
Row in the PT. Periodic number explains the number of energy levels
Block
s,p,d,f. Indicates the last occupied electron orbital
Atomic Radius
Half the distance between the nuclei of two bonded atoms or the distance from the nucleus to the outermost electron.
Atomic Radius trend
Becomes bigge
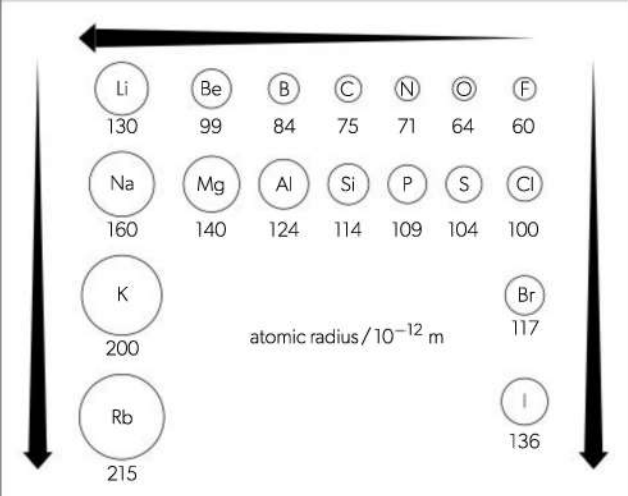
r down a group and from right to left on a period
Factors affecting Atomic Radius
Nuclear charge
– Attractive force that pulls all the electrons closer to the nucleus. With an increase in nuclear charge, the atomic radius becomes smaller.Shielding effect by the electrons of the inner shell(s)
– this makes the atomic radius larger. The shielding effect is the result of repulsion between the electrons in the inner shell and those in the outer or valence shell.
Atomic Radius Trend
Bigger down a group and from right to left on a period
Ionic Radius trend
Bigger down a group and from right to left on a period
Except for Non-metals (Bigger) and Noble Gasses (Smaller)
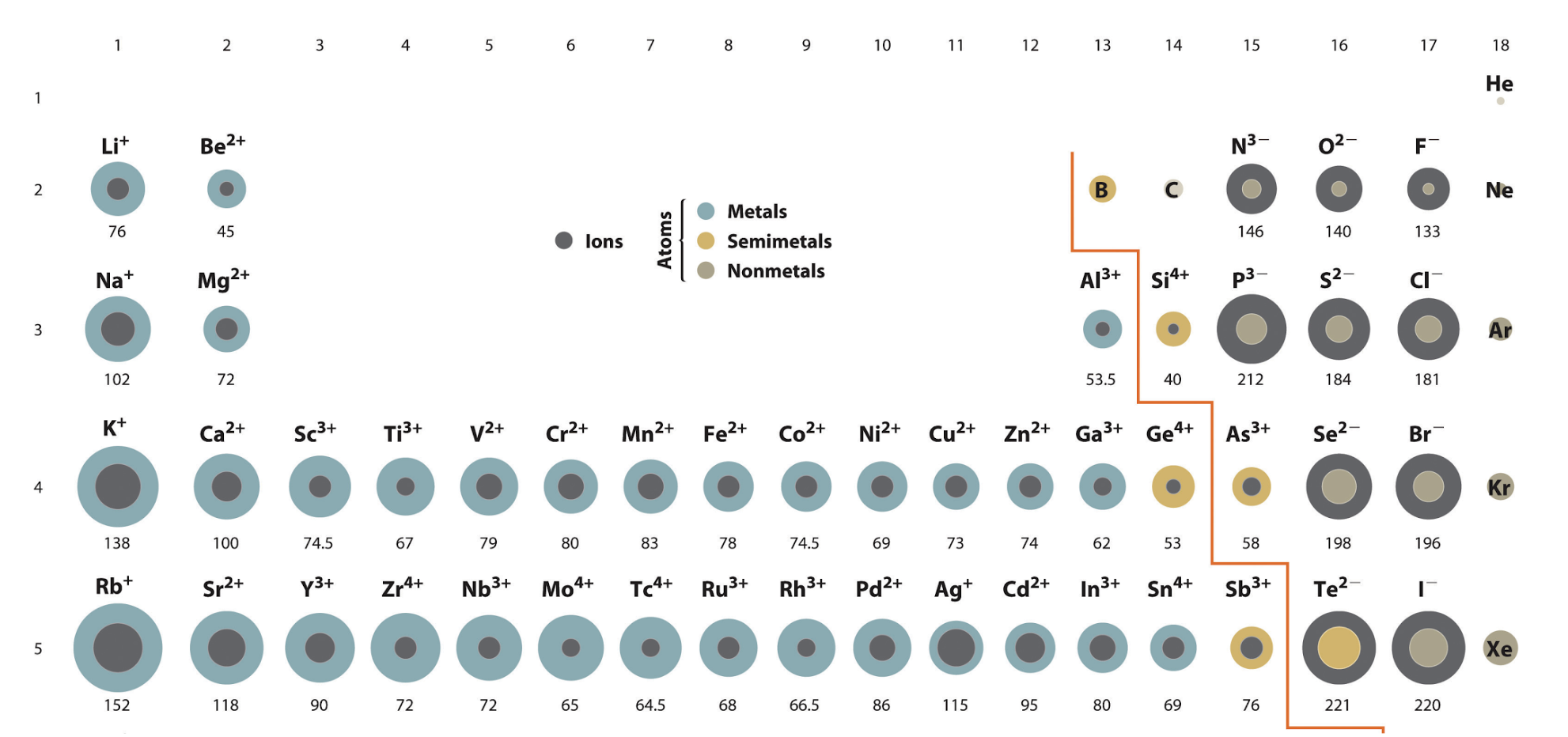
Are Cations bigger or smaller than the parent atom?
Smaller, because they have less electrons, less shells, higher attraction between nucleus and outermost electron
Are Anions bigger or smaller than the parent atom?
Larger, because they have more electrons, maybe more shells, lower attraction between nucleus and outermost electron
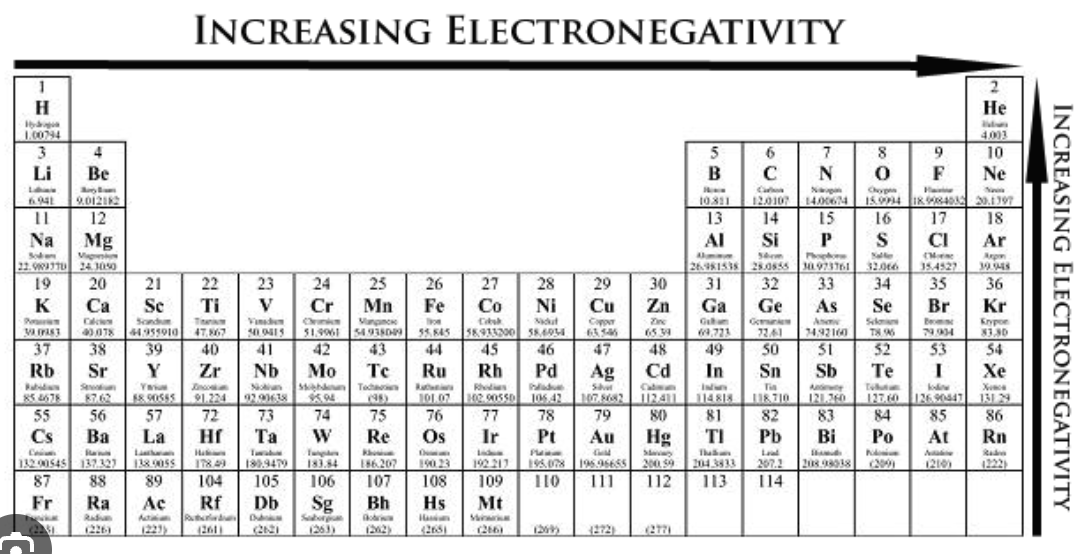
Electronegativity
The tendency for an atom to attract electrons toward itself
The three most important electronegative elements are Fl, O, and N.
Electronegativity trend
Increases up a group and from left to right on a period.
Effective nuclear charge
Distance of the electrons.
(=no of protons and electron shells)
Electronegativity difference (define and remember symbol)
The difference in electronegativity between two bonding atoms. It can tell us a rough measure of polarity and thus the bond type.
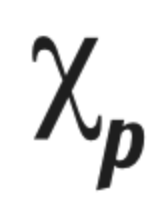
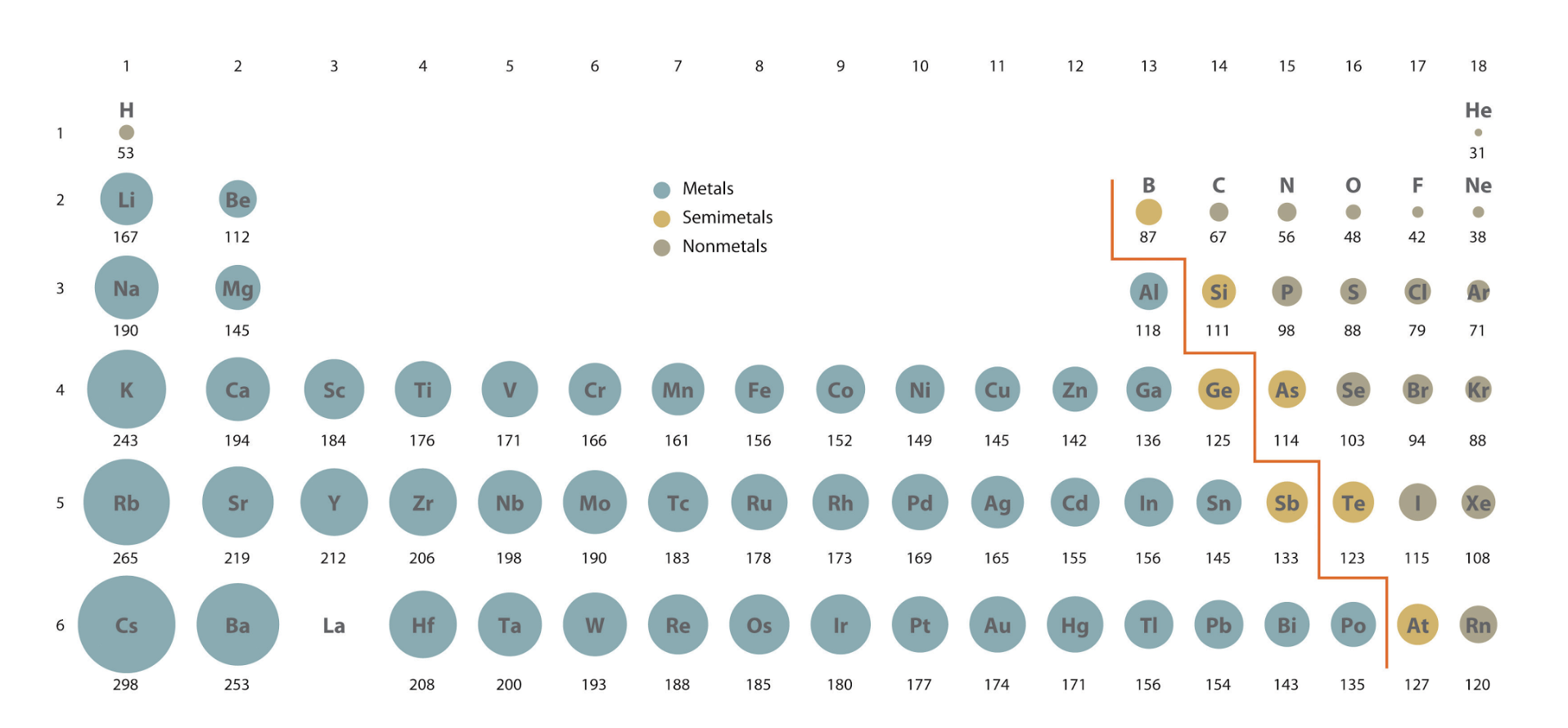
Classify the elements into metals, nonmetals and metalloids
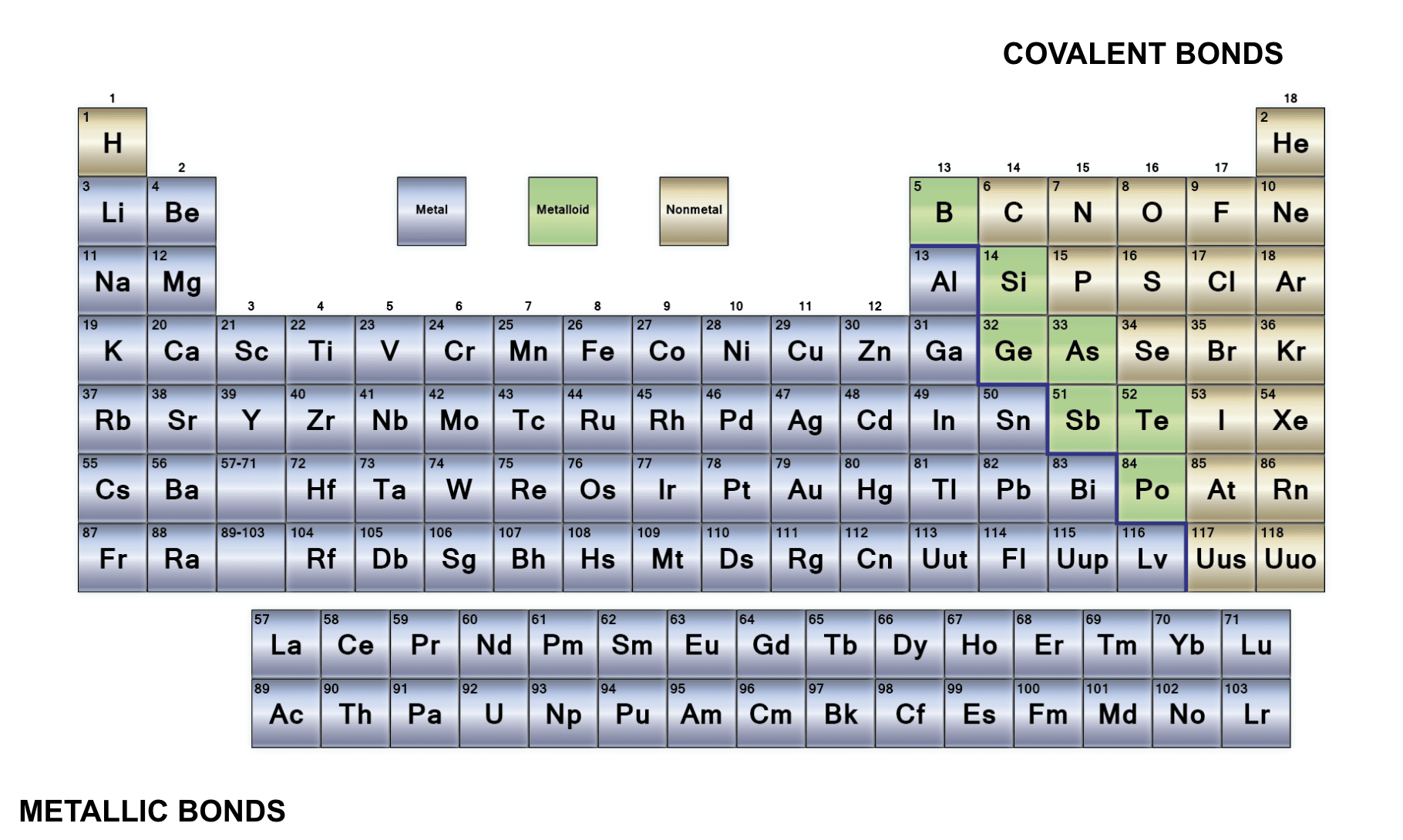
Metals Properties
Shiny
Good conductors of heat
good conductor of electricity
Metal oxides tend to be basic
Metallic properties increase down a group
Non-metals properties
Non-metal oxides tend to be acidic
Important Non-metal oxides
Sulfur:
Sulfur trioxide, SO3, reacts with water to form sulfuric acid
Sulfur dioxide, S02, and oxides of nitrogen, such as N02, are the main gases responsible for the formation of acid rain
Carbon
Carbon dioxide, CO2, is acidic. Increasing atmospheric levels are responsible for ocean acidification, which affects marine life.
Ionization energy
Minimum energy needed to remove one mol of electrons from one mol of gaseous atoms.
Usually an endothermic process
Measured in kJ mol-1
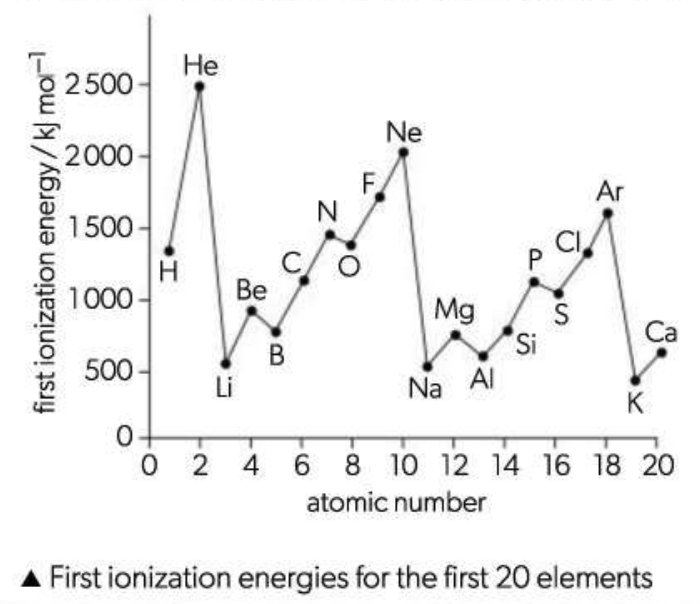
Ionization Energy Trend
Generally, the values increase across a period because the extra electrons fill the same energy level and the extra protons in the nucleus attract this energy level more closely, making it harder to remove an electron. The values do not increase regularly across a period because new sub-levels are being filled . The p sub-level is higher in energy than the s sub-level. This explains why the value for B (ls22s22p1) is slightly lower than the value for Be (l s22s2) and the value for Al ([Ne]3s23p1) is slightly lower than Mg ([Ne]3s2). There is also a drop in value between N (ls22s22p3) and O (ls22s22p4) and between P ([Ne]3s23p3) and S ([Ne]3s23p4). This is because when electrons pair up in an orbital there is increased repulsion, so the paired electron is easier to remove compared with when the three electrons are all unpaired, one each in the three separate p orbitals.
![<p>Generally, the values increase across a period because the extra electrons fill the same energy level and the extra protons in the nucleus attract this energy level more closely, making it harder to remove an electron. The values do not increase regularly across a period because new sub-levels are being filled . The p sub-level is higher in energy than the s sub-level. This explains why the value for B (ls22s22p1) is slightly lower than the value for Be (l s22s2) and the value for Al ([Ne]3s23p1) is slightly lower than Mg ([Ne]3s2). There is also a drop in value between N (ls22s22p3) and O (ls22s22p4) and between P ([Ne]3s23p3) and S ([Ne]3s23p4). This is because when electrons pair up in an orbital there is increased repulsion, so the paired electron is easier to remove compared with when the three electrons are all unpaired, one each in the three separate p orbitals.</p>](https://assets.knowt.com/user-attachments/05f9c48b-aab5-44b1-a8d3-102dabe6d49b.png)
Electron affinity
Minimum energy released when one mole electrons is added to one mole of gaseous atoms.
Usually an exothermic process
When the numerical value increases, electron affinity decreases
Electron Affinity trend

Aluminium Special case
Al is a metal but its oxide is amphoteric, that is, it can be either basic or acidic depending on whether it is reacting with an acid or a base
Oxidation state
The number assigned to that atom to show the number of electrons transferred in forming a bond
Tool to identify which species has been oxidized and which reduced in a redox reaction
Help determine the correct formula and name for ionic compounds.
Oxidation state rules:
Ionic compound: OS of each element is equal to the charge carried by the ion
Covalent compounds: Assume that the compound is ionic, with the more electronegative element forming the negative ion
∑OS’s = Zero
∑OS’s ion = Charge of Ion
Elements not combined with other elements have an oxidation state of zero
Group 1 metals Oxidation state
Always +1
Group 2 metals Oxidation State
Always +2
Hydrogen Oxidation state
Usually +1, Metal Hydrides -1
Oxygen Oxidation state
Usually -2, Peroxides (-1) and F2O
Fluorine Oxidation State
Always -1
Chlorine Oxidation State
Usually -1, Compounds with O or F
Naming of ionic compounds
Metals in groups l, 2, and 13 form cations with oxidation states of +1, +2, and +3, respectively
Transition metals (and other metals, like tin and lead) form more than one ion as they have variable oxidation states. Roman numerals are used for oxidation numbers.
Oxyanions
Formed when oxygen combines with another element to form a complex ion
Should nowadays always have suffix “-ate” and then roman numeral
Complex Ion
Charged species consisting of a central metal atom or ion bonded to one or more molecules or anions, known as ligands. These ligands can be neutral molecules or anions that donate pairs of electrons to the metal, facilitating coordination bonds.