bio kahoots and practice questions exam 1
1/96
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
97 Terms
which statement best describes isotopes?
atoms with the same number of protons, but different neutrons
true or false:
an element consists of one kind of atom
true
which is an example of emergent properties?
sodium chloride (NaCl)
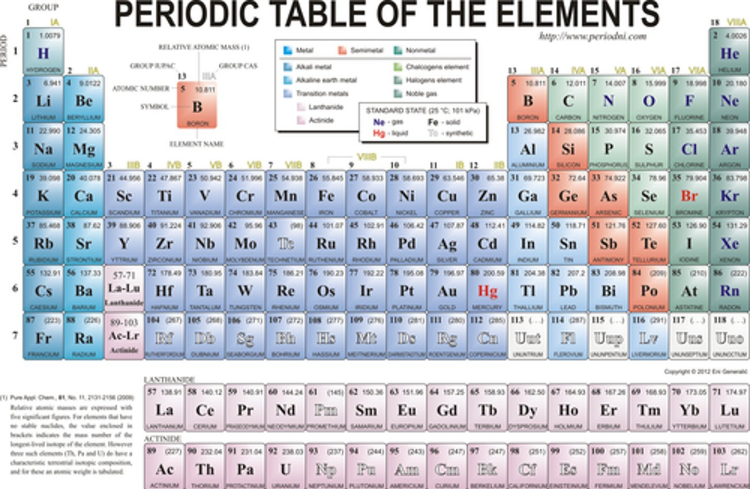
which element has 26 protons?
iron (fe)
which particle determines the identity of an element?
proton
true or false:
atomic number changes when an atom becomes an isotope.
false
Magnesium has an atomic number of 12. How many electrons does a neutral atom of magnesium have?
12
two atoms with the same number of protons but different neutrons are called ________.
isotopes
the sum of protons and neutrons in an atom is called:
mass number
Which statement is FALSE about isotopes?
they are different elements
about how many elements are essential for human life?
25
what are the 4 elements that make up 96% of the weight of most living organisms?
carbon, oxygen, hydrogen, nitrogen
true or false:
same trace elements are required to prevent disease.
true
true or false:
there is only one electron shell.
false
what is something radioactive isotopes are used for?
PET scans
what is the difference between ionic and covalent bonds?
ionic: transfer electrons; covalent: share electrons
true or false:
ionic bonds can be polar or non-polar
false
what makes a covalent bond non-polar?
electrons are shared equally
what makes a covalent bond polar?
electrons are shared unequally
true or false:
an ion is not an atom
false
which property explains water "beading up" on a leaf or on your face after a workout?
cohesion
what does surface tension measure?
how difficult it is to break the suface of a liquid
what does adhesion describe?
the clinging of one substance to another
true or false:
thermal energy is the energy associated with the random movement of atoms and molecules.
true
what does temperature measure?
the intensity of heat
true or false:
water is more dense as a solid than as a liquid
false
When a substance evaporates, the surface of the liquid that remains behind cools down. What is this process called?
evaporative cooling
what allows ice to float on liquid water?
ice molecules are less densely packed than in liquid water
water can exist in what 3 forms?
solid, liquid, gas
what happens when polar or charged solutes are placed in water?
they dissolve as water molecules surround them
true or false:
water's versatility as a solvent results from the polarity of its molecules.
true
what is an aqueous solution?
a solution where water is the solvent
what does a buffer do in a solution?
minimizes changes in pH
true or false:
The pH scale is used to describe how acidic or basic a solution is.
true
true or false:
As a solutions pH increases, it's pH number increases also
false
What is a neutral pH?
7
What is an acidic pH?
1
true or false:
As pH increases, the concentration of hydrogen ions (H⁺) in a solution decreases.
true
true or false:
As pH decreases, the concentration of hydrogen ions (H⁺) in a solution increases.
true
How does the acid formed from CO₂ affect coral reefs?
it disrupts the formation of coral skeletons
true or false:
When carbon dioxide (CO₂) dissolves in water, it reacts with water to form a base.
false
how might a chemical compound in the air harm coral reefs?
through chemistry-- CO2 forms an acid in water
why are carbon atoms considered the "lead players" in the chemistry of life?
they can form four covalent bonds, allowing complex molecules
true or false:
the shape of a molecule usually determines its function in the body.
true
true or false:
Cyclohexane (C6H12) and benzene (C6H6) are isomers.
false
Methamphetamine occurs as two isomers: one is an illegal drug, and the other is a sinus medication. What does this show?
the shape of an isomer can change its function
Which of the following is NOT one of the four main ways carbon skeletons can vary?
ability to dissolve in water
true or false:
The carboxyl group can act as an acid by donating an H+ to a solution.
true
which chemical groups do not contain carbon?
hydroxyl, amino, phosphate
which group contains a carbon atom double-bonded to an oxygen atom?
carbonyl
Which group is often involved in energy transfers, such as in ATP?
phosphate
what is lactose?
the main sugar found in milk
why do some adults lose the ability to digest lactose?
their body stops producing lactase after infancy
________ is the enzyme responsible for breaking down lactose.
lactase
which of the following correctly describes the carboxyl group?
-COOH
true or false: the amino group (-NH₂) contains a nitrogen atom bonded to two hydrogen atoms and can act as a base.
true
____________ is the process of breaking a polymer into monomers by adding water.
hydrolysis
which of the following is essentially the reverse of a dehydration reaction?
hydrolysis
true or false:
both hydrolysis and dehydration reactions require enzymes to help make and break bonds.
true
after digestion, new proteins are built in your body cells from amino acids using:
dehydration reaction
what are the building block of polymers?
monomers
does hydrolysis break or create bonds?
breaks
in dehydration reactions, bonds are _________.
created
the four most common elements in living organisms are:
carbon, nitrogen, hydrogen, oxygen
which of the following is commonly added to table salt to prevent the formation of goiters?
iodine
a radioactive isotope has a nucleus that
decays spontaneously
a chemical bond that results in one or more molecules stripping electrons from the outer shell of another molecule is called a(n) ______________ bond
ionic
an uncharged atom of argon has an atomic number of 18 and an atomic mass of 40. this atom has ___ protons, ___ neutrons, and ___ electrons. (same number of protons and electrons)
ps- you get the atomic mass number by adding protons and neutrons
18; 22; 18
which of the following is an example of a polysaccharide?
starch
lactose intolerance is the inability to properly __________.
digest lactose
what are saturated fats saturated with?
hydrogen
what name is given to the following reaction?
(glucose + glucose → maltose + water)
dehydration reaction
the shape of a protein is determined by:
all of the above (the interaction between R-groups pf a chain of amino acids; hydrogen bonds along the backbone of the polypeptide; the sequence of amino acids)
a ______ is an example of a unicellular organism
paramecium
bacteria cells are prokaryotic; unlike a typical eukaryotic cell, they _______.
lack a nucleus
____ are the major lipids of plasma membranes
phospholipids
which of these pairs of organelles are responsible for energy conversion
mitochondrion and chloroplast
information is transfered from the cell nucleus to ribosomes via the molecule __________.
RNA
explain why fats and steroids, which are structurally very different, are both classified as lipids
they are hydrophobic; made almost entirely out of carbon and hydrogens
why does a denatured protein no longer function normally?
if you change the shape of a protein, it ceases to function the same way and may not function at all
why are R groups important?
they decide the shape of a chain
true or false:
RNA does not form strong enough partnerships to form a double helix
true
molecules that have hydroxyl groups (OH) are called _______
alcohols
why are fats hydrophobic
they are nonpolar, so there is no way for water to interact with it
A protein’s _______ structure is the sequence of amino acids in its polypeptide chain, which can consist of alpha helixes or beta pleated sheets
primary
A protein's ________ structure is the overall three-dimensional shape of a polypeptide, resulting from interactions among R groups
tertiary
A protein's _______ structure is the coiling or folding of the chain, stabilized by hydrogen bonds.
secondary
Proteins made of more than one polypeptide have ________ structure
quaternary
the 4 most common nucleotides found in DNA are:
adenine, thymine, cystocine, guanine
what holds nucleotides to the same strand?
bond between the sugar and the phosphate (sugar phosphate backbone)
true or false:
DNA can copy itself
true
DNA and RNA are both _______ acids
nucleic
what are the domains of living things?
eukarya, bacteria, archaea
_____ and ____ consist of prokaryotic cells; all other forms of life are placed in domain ________ and have eukaryotic cells
bacteria and archaea; eukarya
true or false:
the cytoskeleton gives the cell shape and structural support
true
________ endoplasmic reticulum has no ribosomes attached while the ________ endoplasmic reticulum has ribosomes attached
smooth; rough
smooth ER ___________________, while the rough ER ____________, and ribosomes on its surface make membrane and secretory proteins
synthesizes lipids and processes toxins; produces membranes