A2 Unit 3.3 & 3.4 Chemistry of the p and d block elements
1/40
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
41 Terms
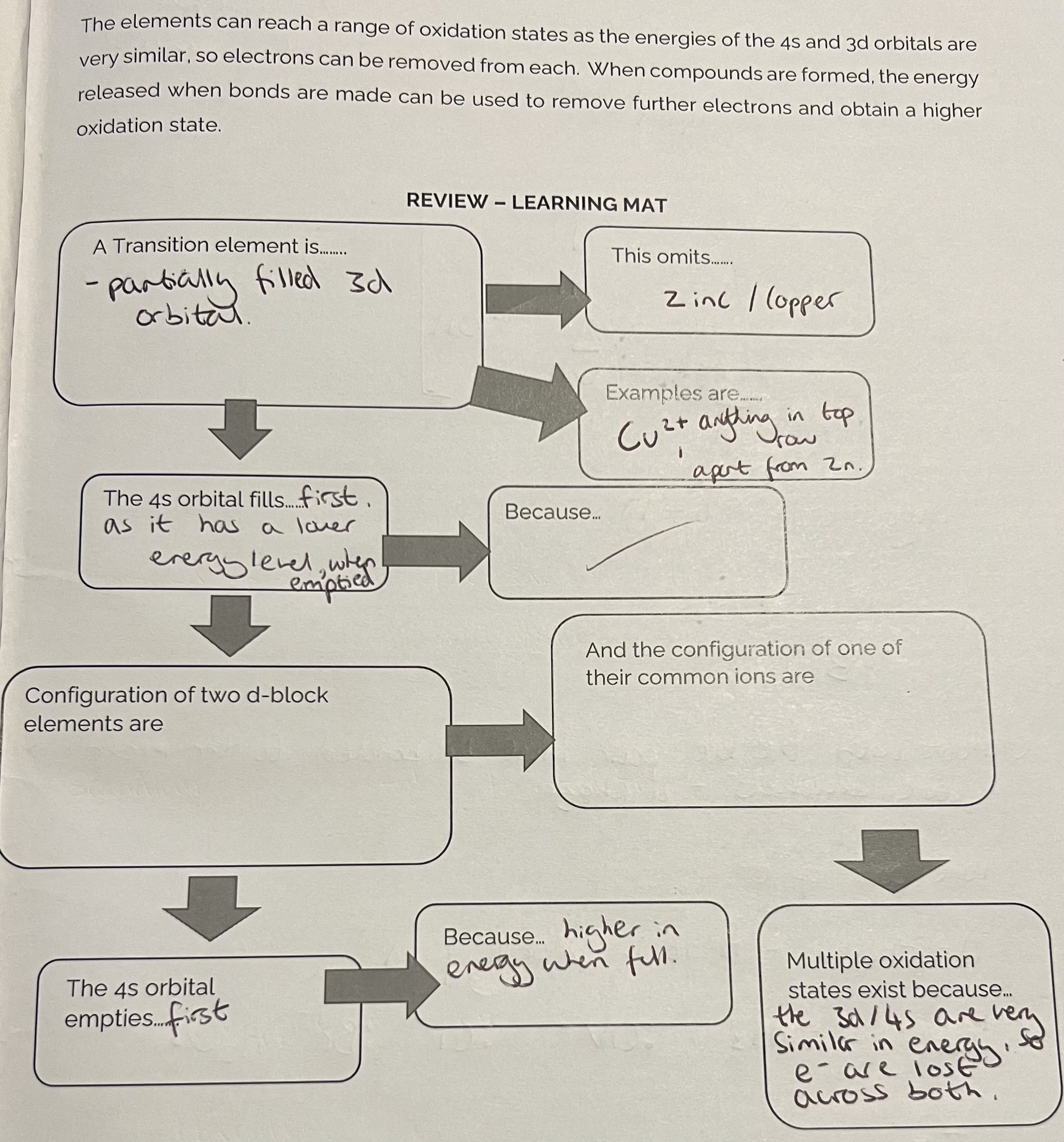
The d-block transition elements (metals)

Electronic configuration for ions

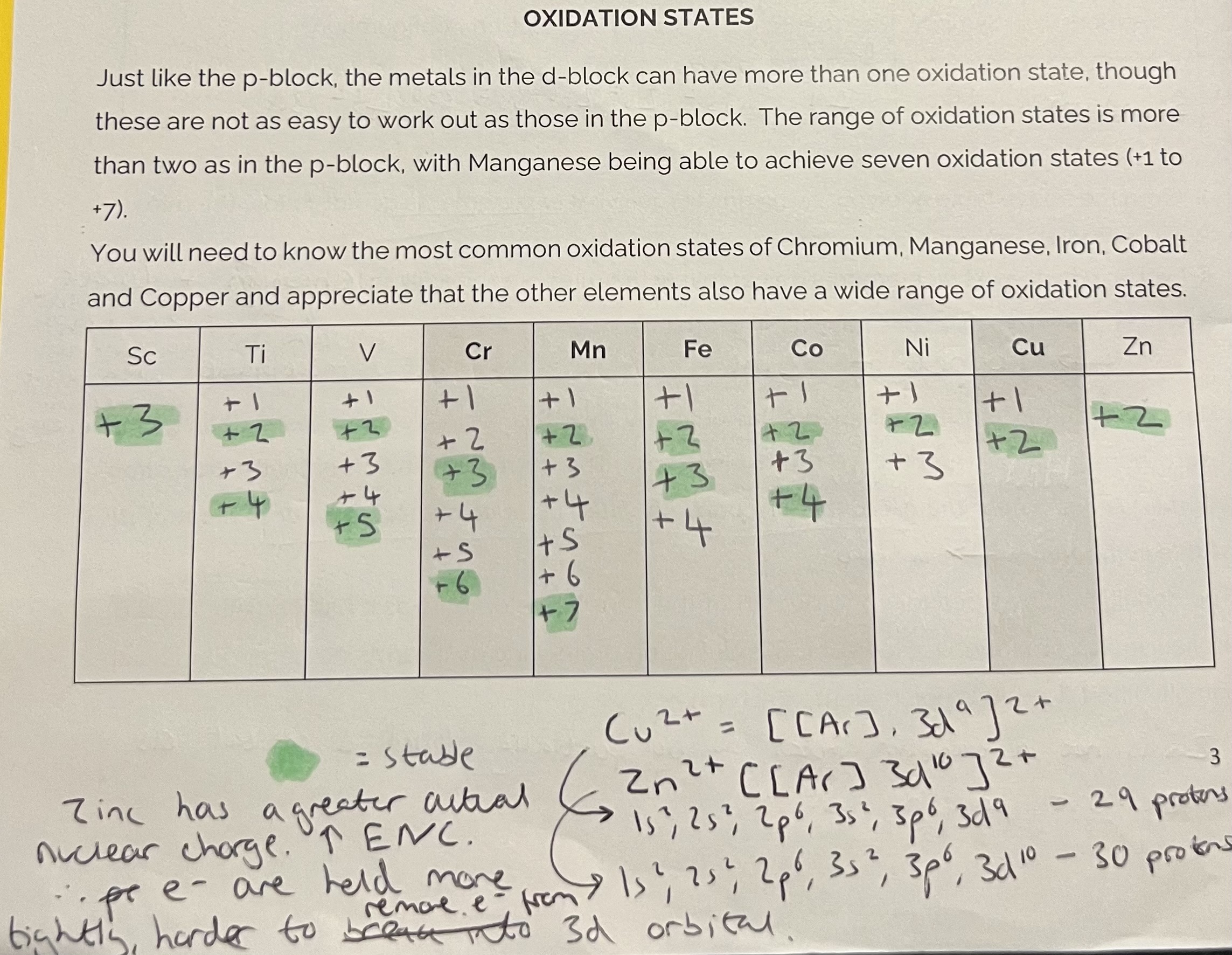
Oxidation States
The elements can reach a range of oxidation states as the energies of the 4s and 3d orbitals are very similar, so electrons can be removed from each.
When compounds are formed, the energy released when bonds are made can be used to remove further electrons and obtain a higher oxidation state.
Transition elements summary
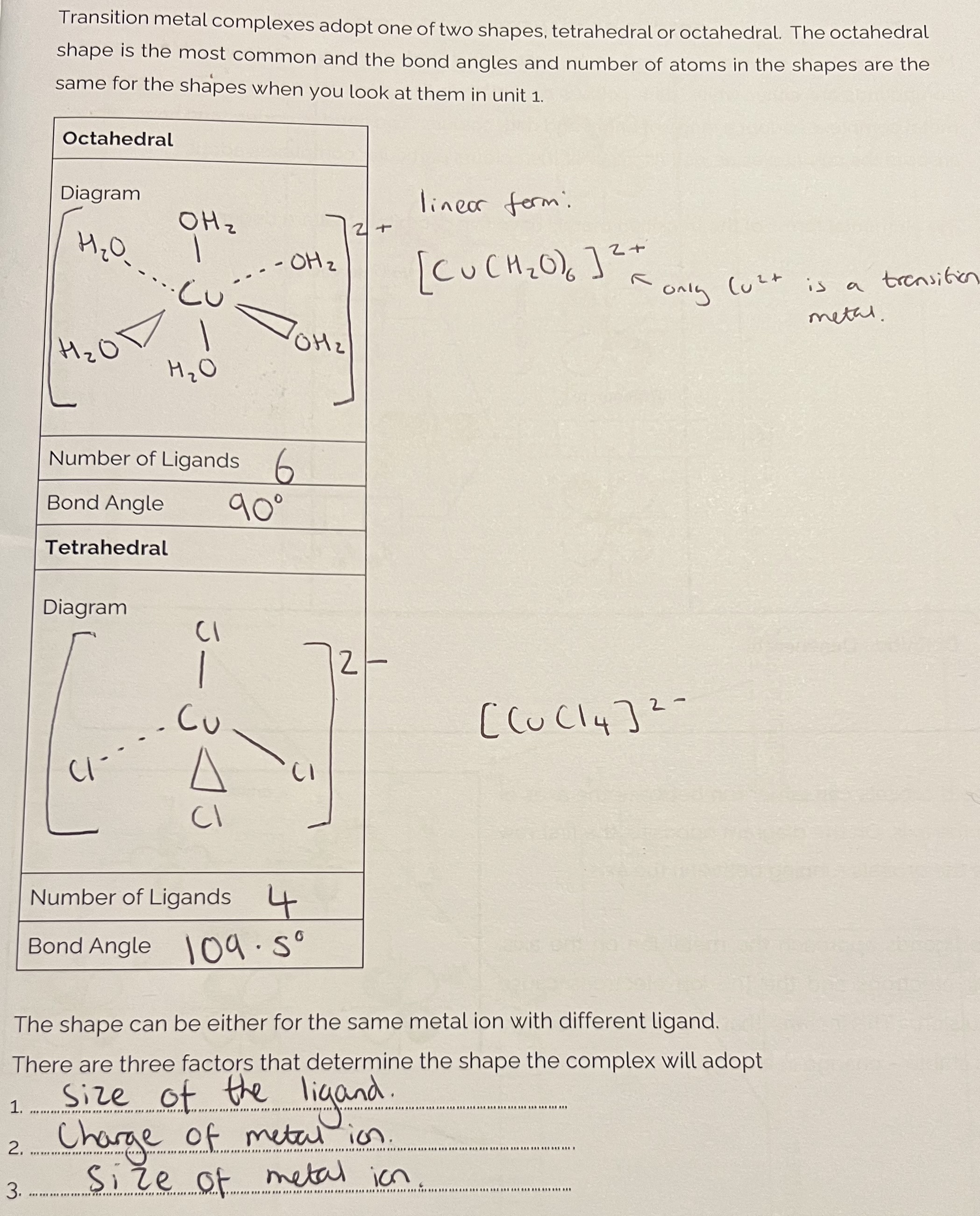
Transition Metal Complexes

Shapes of Transition Metal Complexes
Colour of transition metal complexes
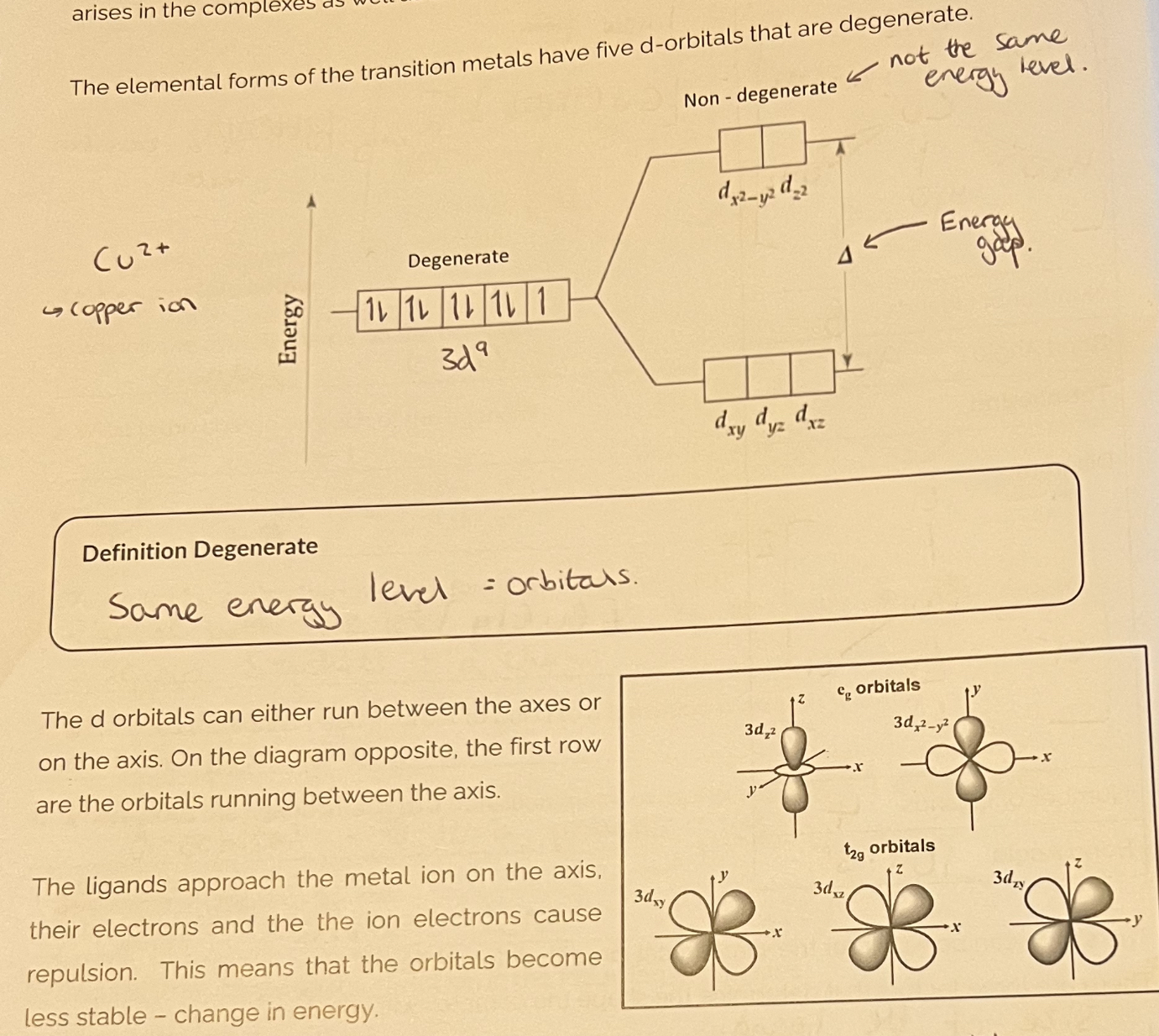
How do transition metal complexes exhibit colour?
1) 3d orbitals are degenerate
2) Ligands (e.g H2O) approach, forming coordinate bonds in the partially filled d- orbital
3) Lots of e- —> repulsion which causes splitting into 2 higher & 3 lower energies
4) e- are excited from lower —> higher by absorbing a wavelength of white light (energy gap corresponds to the wavelength of light absorbed, gap is dependent on ligand & shape)
5) The colour observed is the wavelength that is not absorbed

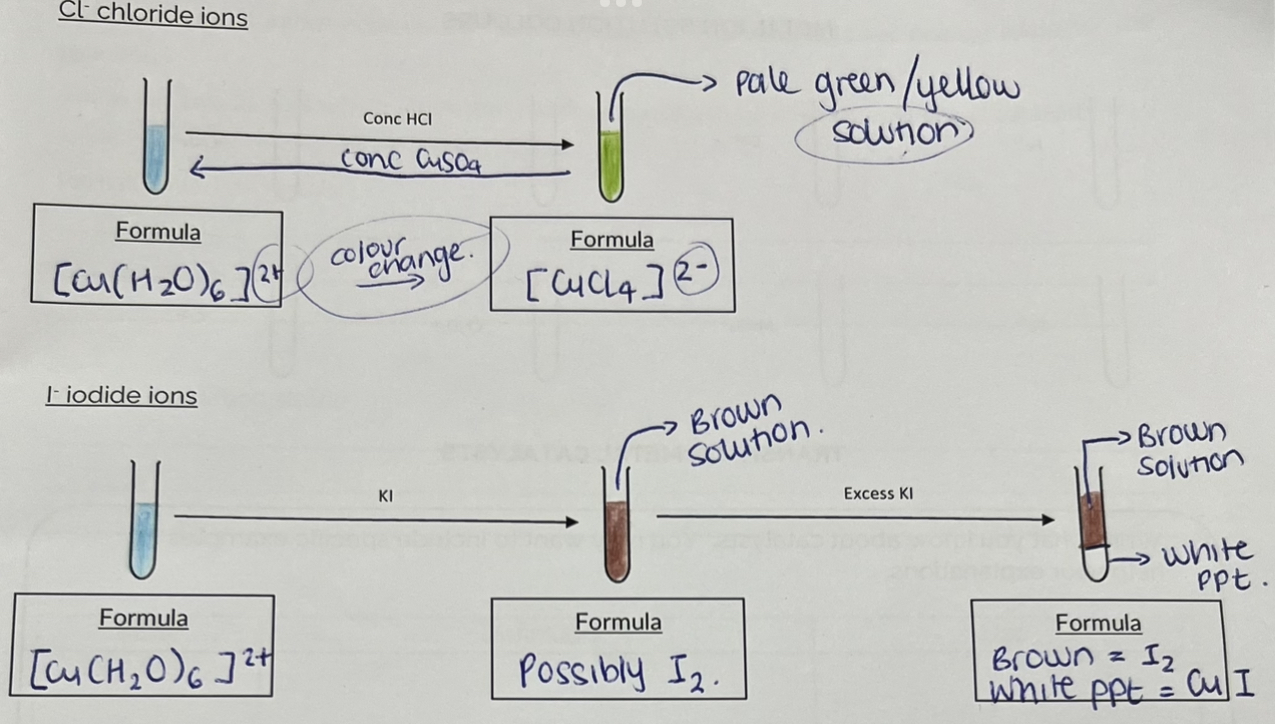
Experiment - Ligand exchange in copper complexes
Should be a 2+ charge on royal blue solution
Solution = charge
Precipitate formed = no charge

Ligand exchange in copper complexes
Pale green —> Pale blue
Add H20 not CuSo4
Ligand exchange in cobalt complexes

Metal ion solution colours

Transition Metal Catalysts
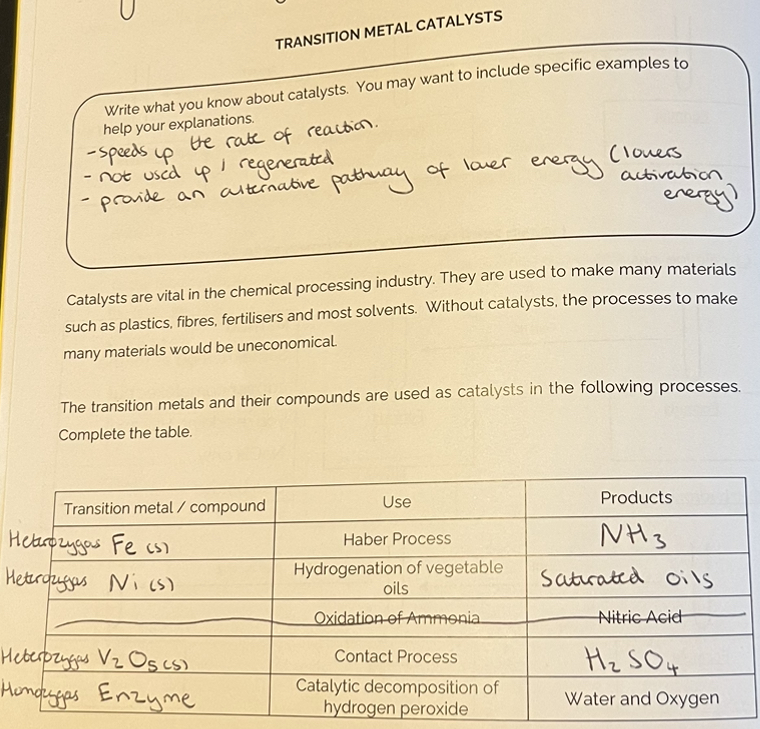
Factors which make transition metals good catalysts

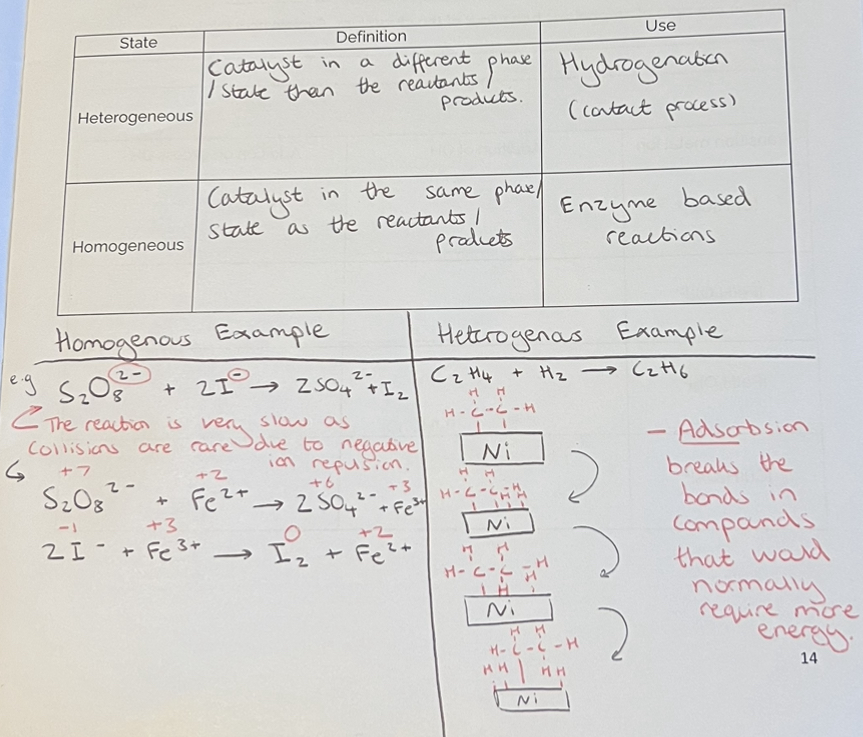
Heterogenous & Homogenous Catalysts
REACTIONS OF TRANSITION METAL IONS WITH HYDROXIDE IONS - Test for amphoteric behaviour

Colour changes observed - Transition metal ions + Addition of OH-

Observations of Chromium

General ideas of the P-block elements

Oxidation states of the p-block

Typical oxidation states of the p-block

Inert pair effect
As you go down group 4, the Ionisation Energy decreases, however there is a slight increase from Sn —> Pb.
In tin and lead (which prefer to bond ionically), the p2 e- do take part in bonding, the s2 e- do not, due to the inert pair effect.

Inert pair effect + Covalent bonding
Elements further down the group cannot reach the highest oxidation states in a covalent bond as the energy required to excite the e- is greater than the energy released by bond formation - due to the inert pair effect.

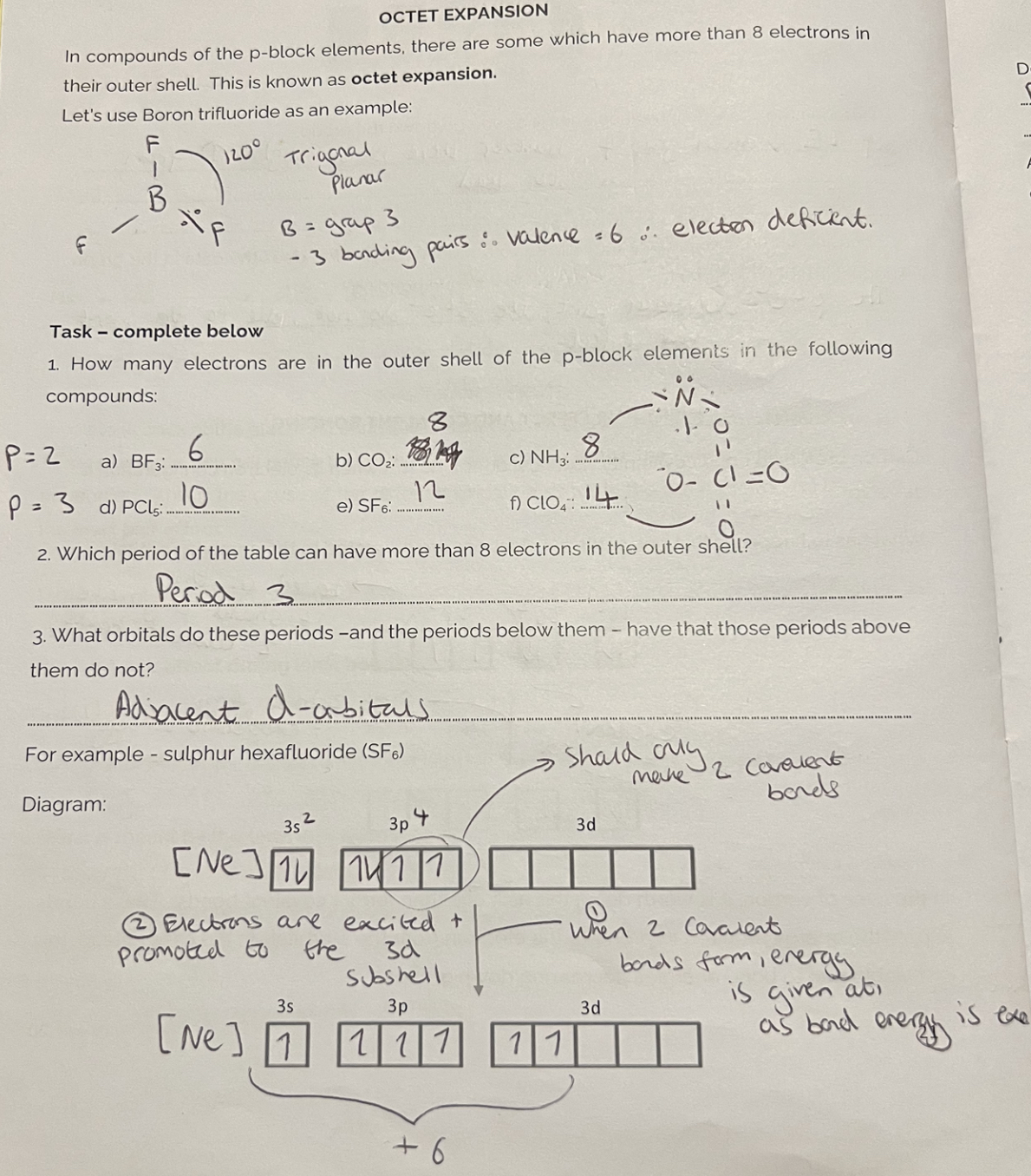
Octet expansion
Definition: NH3 cannot expand its octet as it has no adjacent 3d orbitals to promote e- easily.
PCl5 can expand its octet as it has an adjacent 3d orbital to promote e- easily.
Group 3 Chemistry - e- deficiency

Dimerisation

Boron Nitride

Cubic boron nitride v Diamond

Hexagonal Boron Nitride v Graphite

Reactions of Group 3 & 4 - Aluminium
Spectator ions do not get involved in reactions (their oxidation state doesn’t change).
Acids donate H+ and Bases accept H+.

Reactions of Group 3 & 4 - Lead

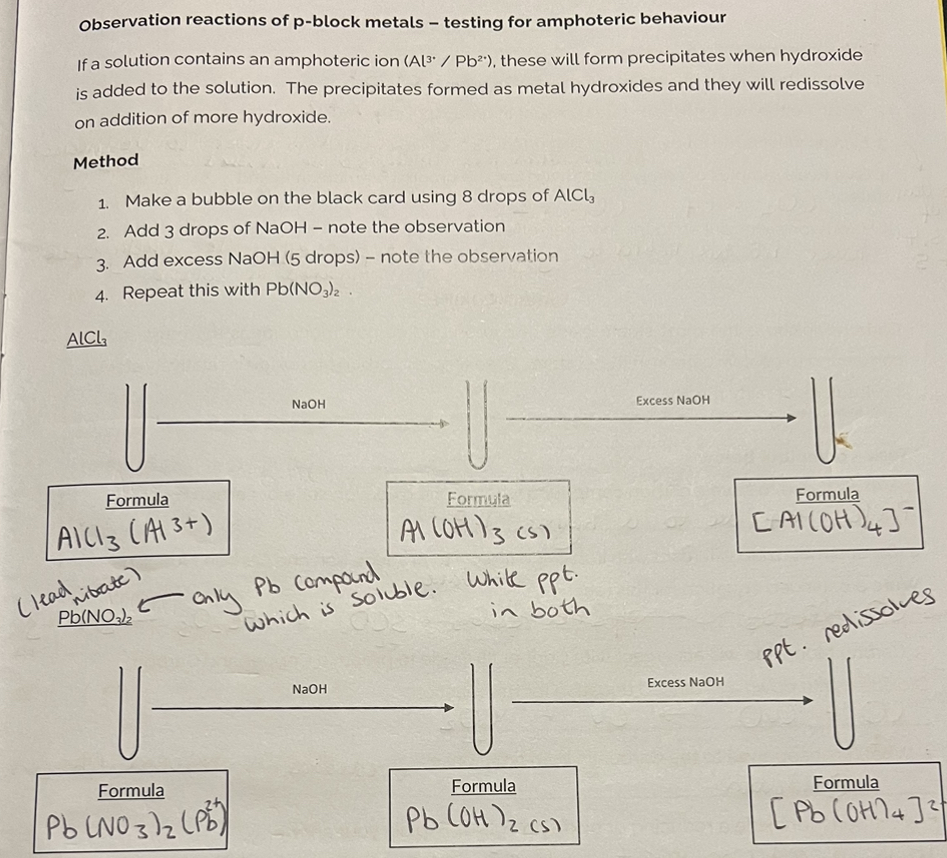
Observation reactions of p-block metals - testing for amphoteric behaviour
Group 4 Chemistry + Carbon Oxides

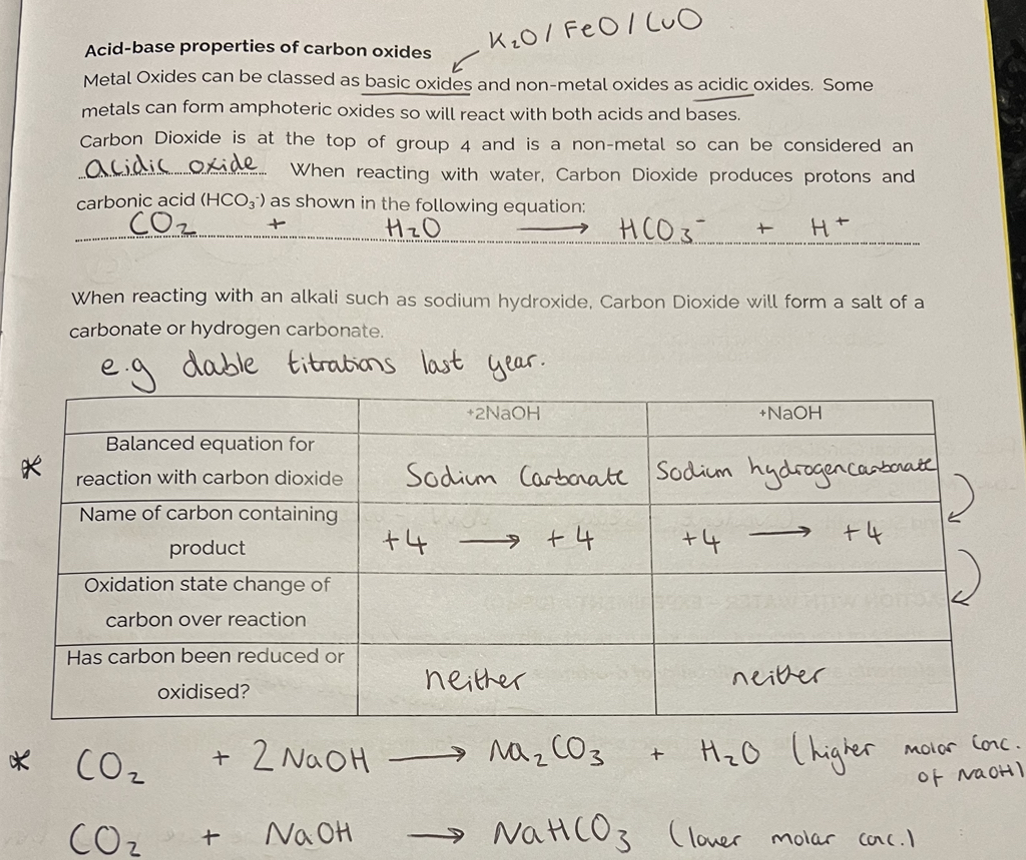
Acid-base properties of Carbon oxides
The chlorides of Group 4

Reaction of G4 Chlorides with water

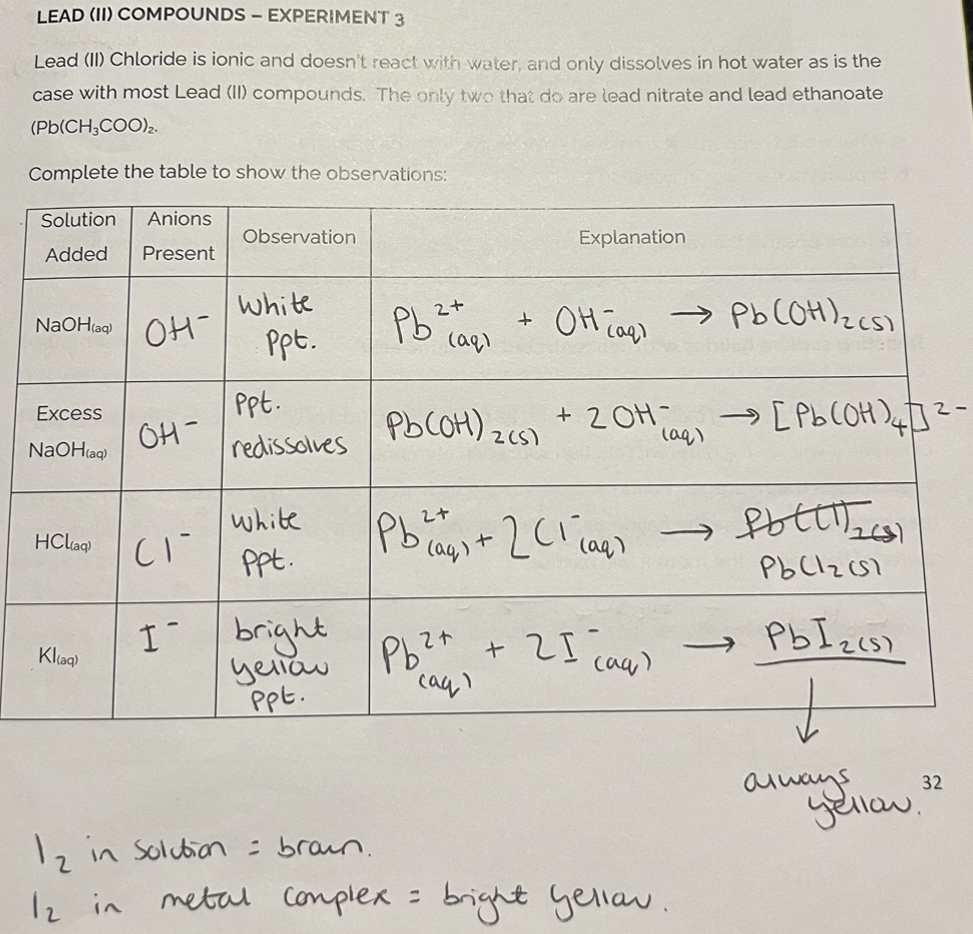
Lead (ll) Compounds
Group 7: The Halogens

NaCl, NaBr, HBr + NaI reactions
Hydrogen sulphide gives rotten egg smell

HI reaction

Reaction of Cl2 & NaOH
