AP Chemistry: Unit 2 BONDING
1/23
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
24 Terms
Ionic
Electrostatic attraction between ions
Covalent
Sharing of electrons
Metallic
Sea of electrons
Lattice energy
completely separate a mole of solid ionic compound into its gaseous ions
LATTICE ENERGY INCREASES WITH
increased charge of ions
decreased size of ions
Covalent bonding have several interactions…
attraction between electrons and nuclei
repulsions between electrons
repulsions between nuclei
Formation of bonds is
exothermic
Breaking of bonds is
endothermic
Polar Covalent Bonds
Electrons are NOT always shared equally, more electron density on the electron that is pulling HARDER
What determines whether somethin is pulling harder?
electronegativity (increase from left to right, increase from bottom to the top)
When atoms share electrons unequally,
a bond dipole results
A dipole moment
produced by two equal but opposite charges separated by a distance r
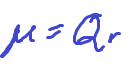
What determines polar covalent strength?
The greater the difference in electronegativity, the stronger the polarity of the bond
Lewis Structures
Representation of molecules showing all electrons, bonding and non bonding
How to write Lewis Structures
fill octet

Central atom in Lewis Structure is the _____ electronegative (that isn’t hydrogen)
LEAST
FORMATION OF MULTIPLE BONDS WITH LEWIS STRUCTURES
ONLY C,N,O,P, and S can form multiple bonds
Resonance
multiple structures can used to describe the same molecule
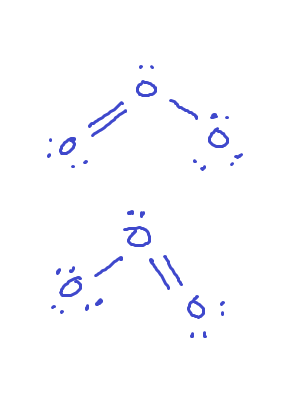
Resonance structure
delocalized, can move to any of the structures at any time
How determine the best lewis structures?
FORMAL CHARGES
How do we determine formula charges?
COUNT ITS ELECTRONS IN LONE PAIRS and HALF OF THE ELECTRONS IT SHARES WITH OTHER ATOMS, subtract that from valence electrons
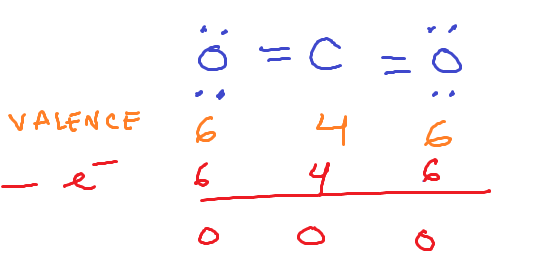
If using formal charge to determine which structure is best and it’s impossible to get all 0 formal charges,
you choose the structure where the most electronegative element has the MOST electronegative element and the MOST negative charge
EXCEPTION TO OCTET RULE
-ions/molecules with an odd number of electrons
-ions or molecules with less than an octet
-ions or molecules with more than eight valence electrons (an expanded octet)