Parasitism final
1/109
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
110 Terms
kinetoplastids
broad range of free-living and parasitic protists
range of cell morphologies and is tailored to the environment parasite occupies
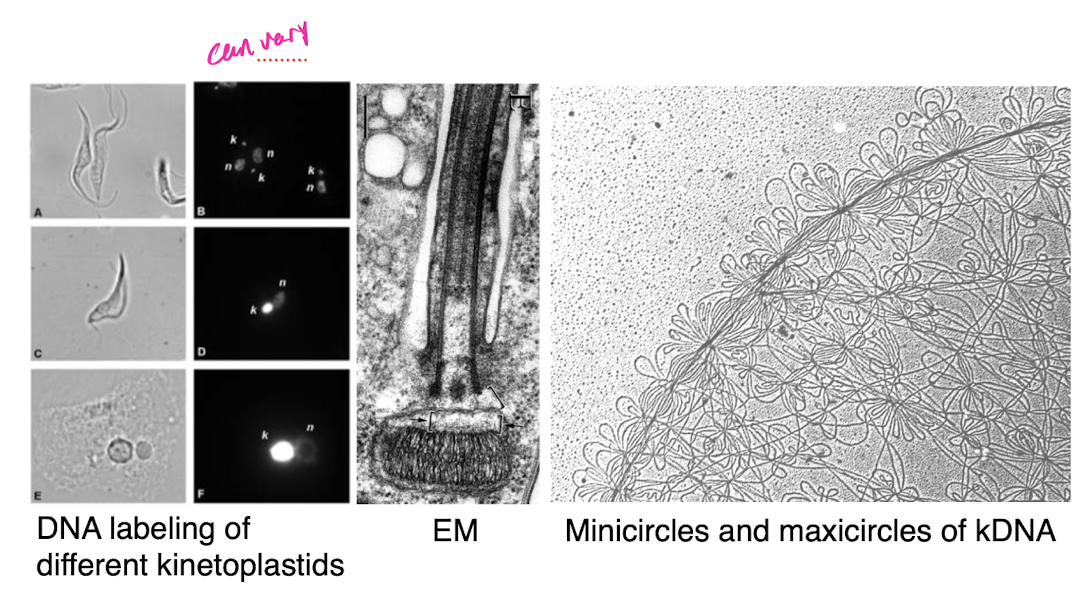
kinetoplast
defining feature of kinetoplastids
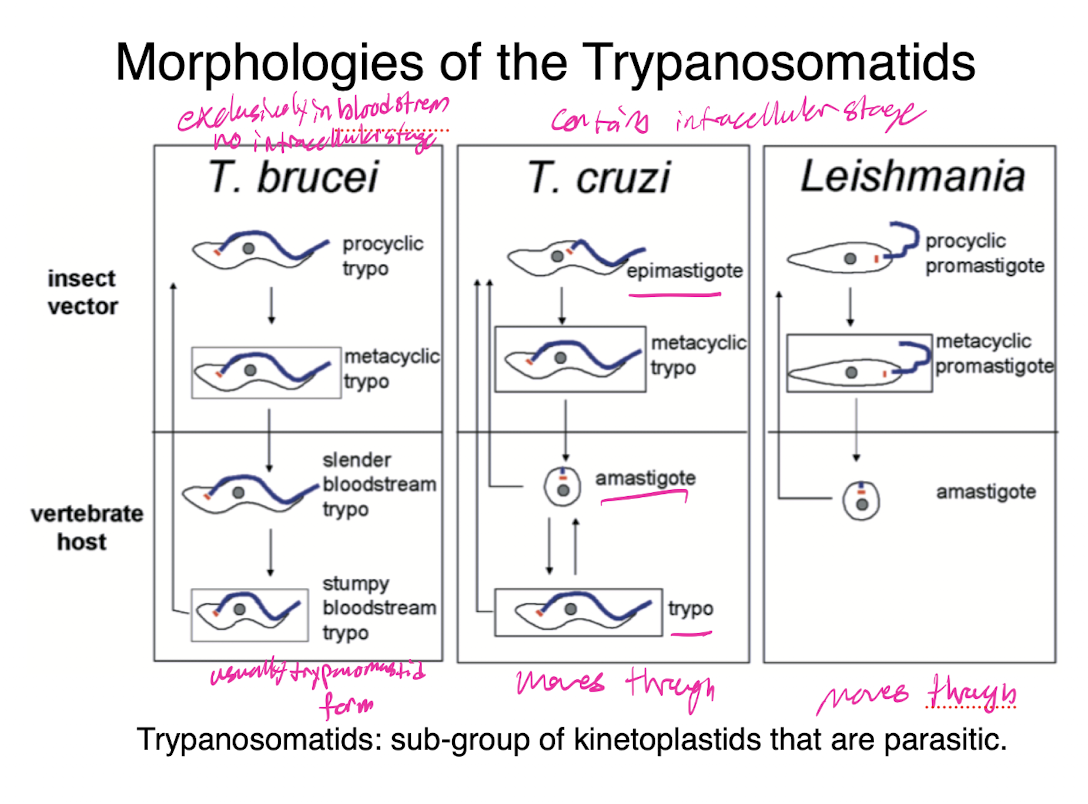
trypanosomatids morphologies
subgroup of kinetoplastids that are parasitic
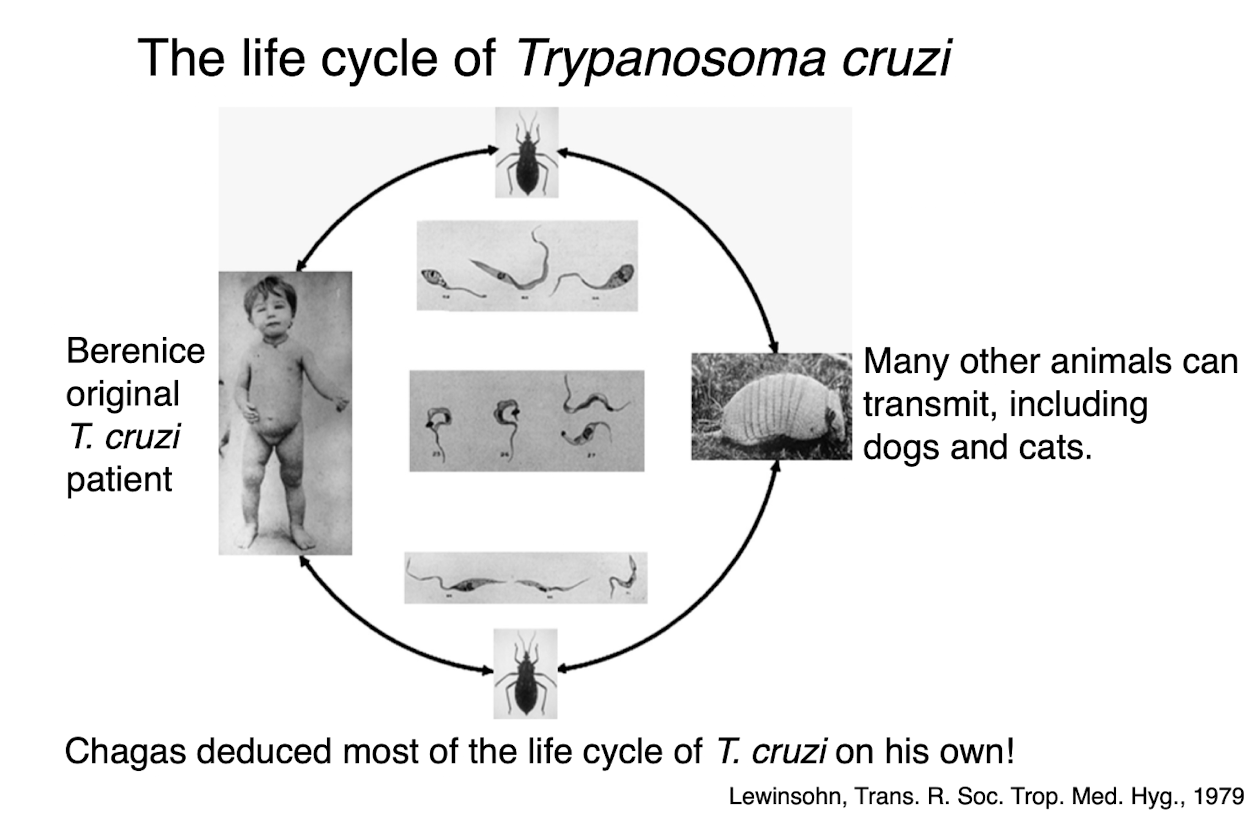
T cruzi discovery
carolos chagas - Brazilian doctor
transfered in triatomine, or kissing bug
triatomine
bites the faces of people when the sleep
vector of T cruzi
take large blood meals and poop when they bite
T cruzi identified in posterior midgut, similar to T brucei
wattle and daub construction is ideal habitat for kissing bugs as they hide in the thatching in roof, come down to feed at night
common across all of Latin America
t cruzi in triatomine
inhabits the digestive tract
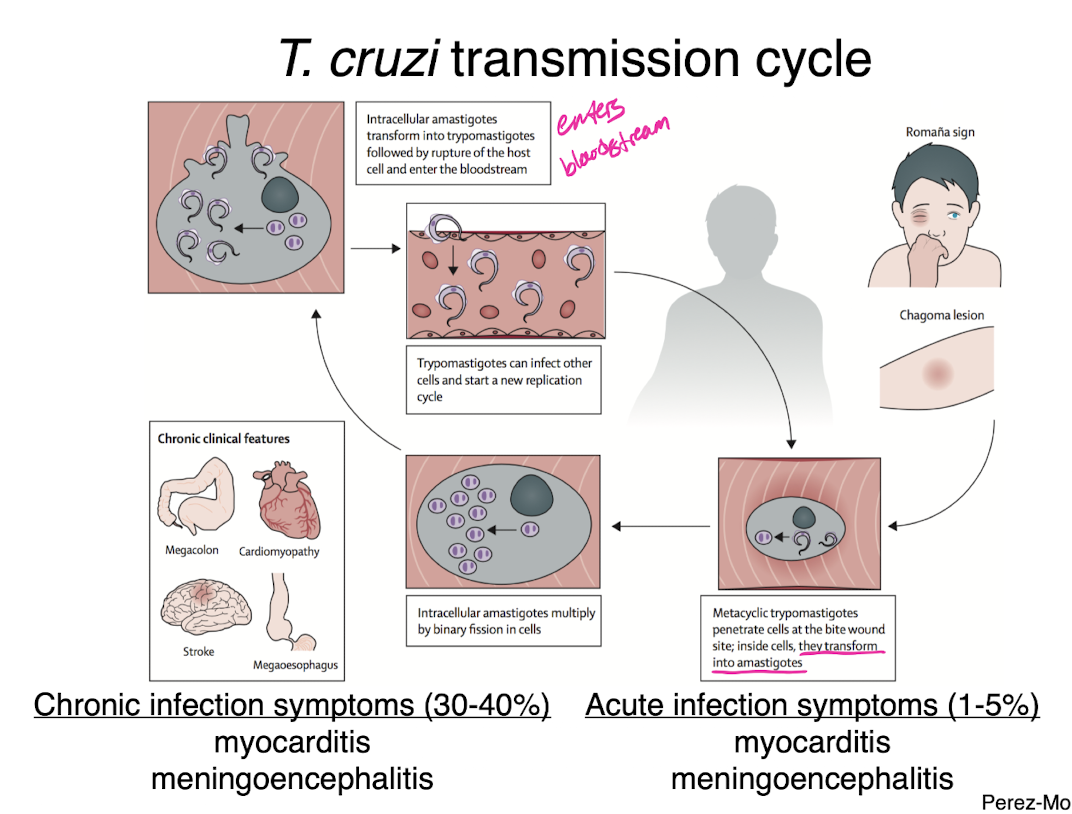
t cruzi life cycle
chronic t cruzi effects
causes range of organ defects
swelling of the lining of the esophagus and colon
peristalsis also stops making it hard to swallow and pass water
t cruzi transmission cycle
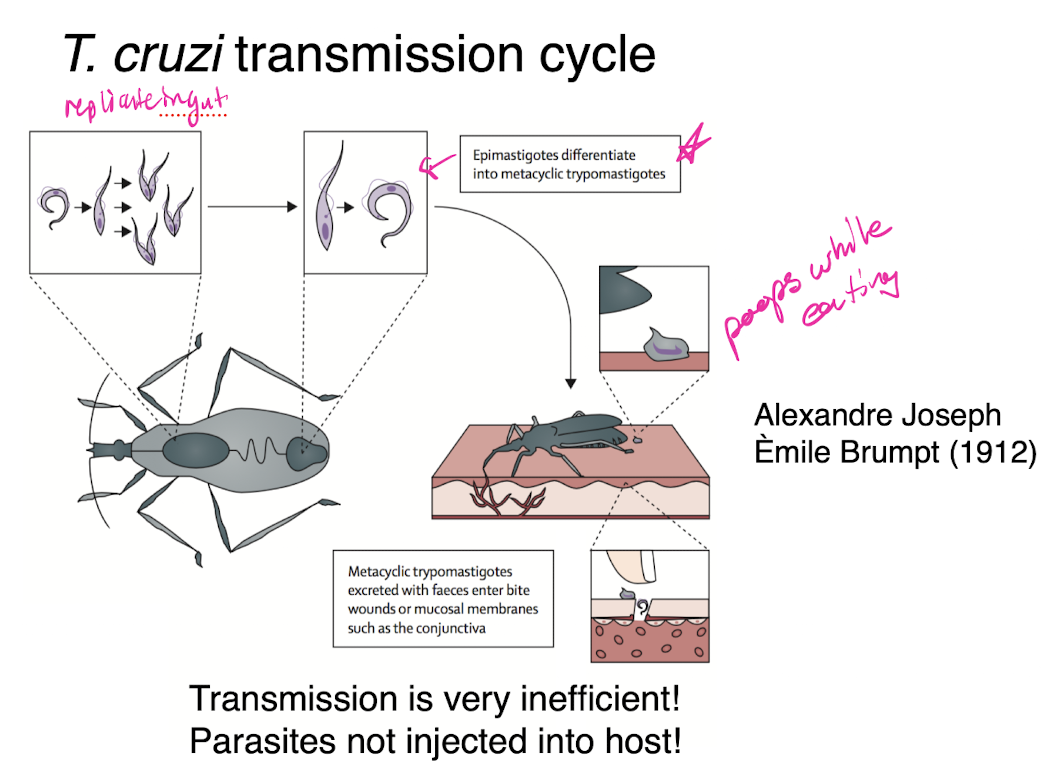
drugs for treating chagas
Nifurtimox and benznidazole are only current drugs
both prodrugs, must be activated by cells before they become toxic
T cruzi contains a bacterial-like nitroreductase, produces radicals from nitro group which is cytotoxic
explains selectivity for parasite over mammalian cells
really only work well if during acute stage of infection
nifurtimox
treats chagas
causes severe nausea and vomiting, leading to weight loss
many people don’t finish course of drug, which can facilitate development of resistance in parasite
benznidazole
treatment for chagas
better tolerated but very poor at clearing chronic infections in adults
why chagas concern in latin America
zoonotic hosts (armadillo, monkeys, cats) means T. cruzi will never be eradicated
afflicted tend to be poorest of poor, don’t seek diagnosis or treatment
deaths occur long after initial infection and are frequently not associated with disease (heart issues especially)
chagas in north america
becoming more of an issue primarily due to immigrants moving from endemic nations
insect vectors moving north
doctors never consider chagas as possible ailment
especially problematic for pregnant women
bigger problem in Europe
clearing t cruzi does not
alter progression of Chagas cardiomyopathy
manifestation of chagas cardiomyopathy
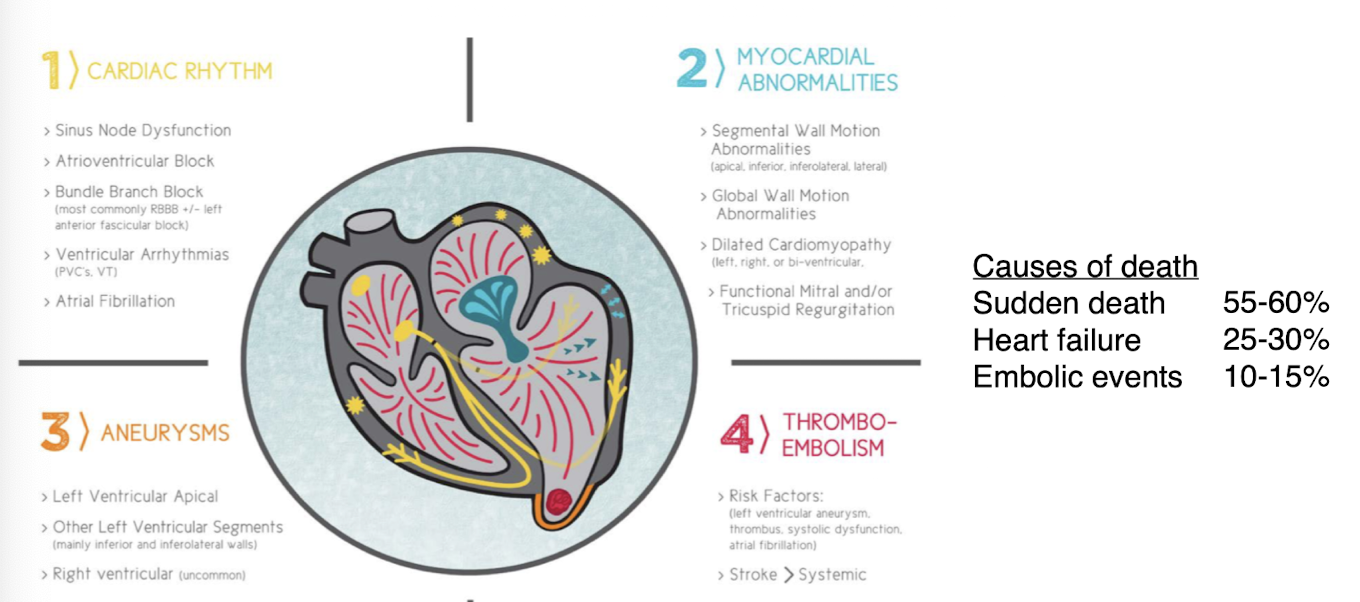
chagas diseases
95% under diagnosis rate
minimal acute infection symptoms
chronic infection can lay dormant for decades
treatment of chronic disease doesn’t undo the damage
30-40% of infected will develop heart problems
sudden death via heart attack is possible
won’t be diagnosed as due to chagas
poor people with poor access to healthcare are most at risk
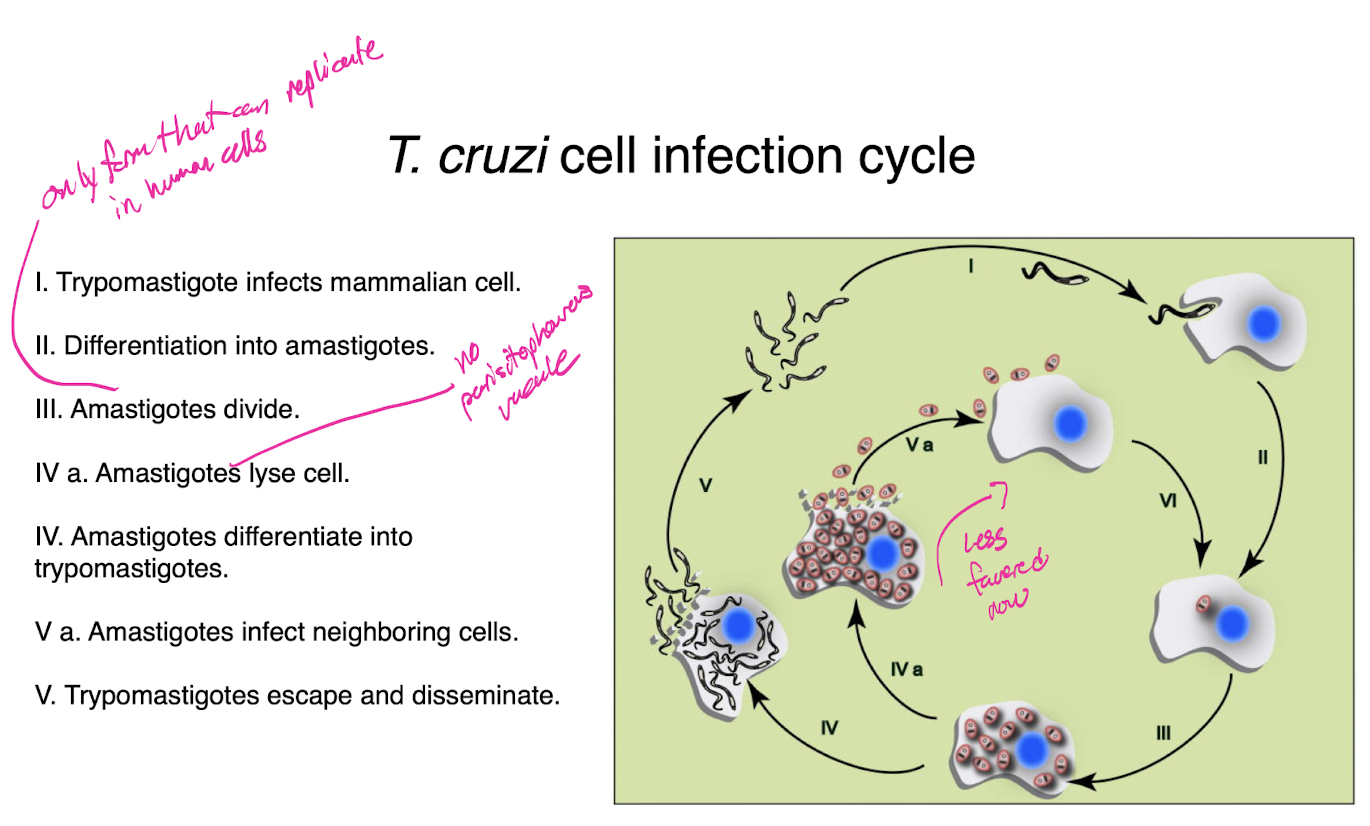
t cruzi cell infection cycle
trypomastigote infects mammalian cell
differentiation into amastigotes
amastigotes divide
amastigotes lyse cell
amastigotes differentiate into trypomastigotes
amastigotes infect neighboring cells
trypomastigotes escape and disseminate
t cruzi cell surface coat
trans-sialidase: ~1400 genes
mucins: heavily glycosylated proteins
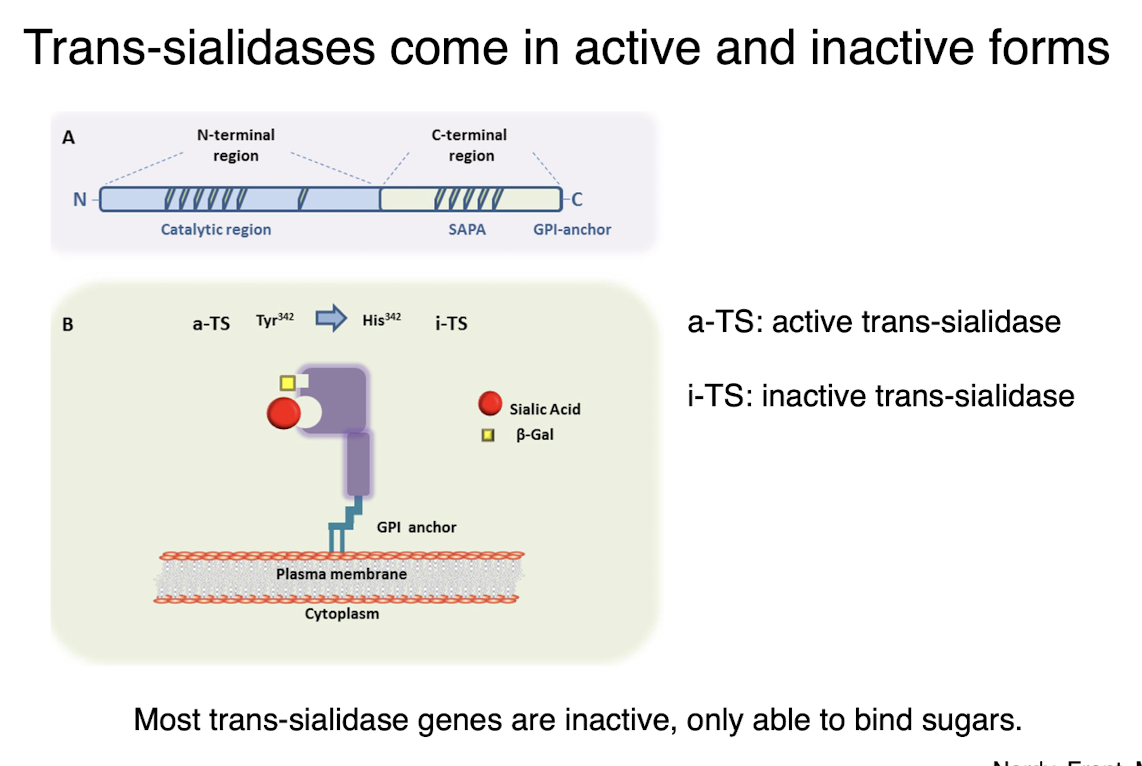
trans-sialidases
come in active and inactive forms
most inactive, only able to bind sugars
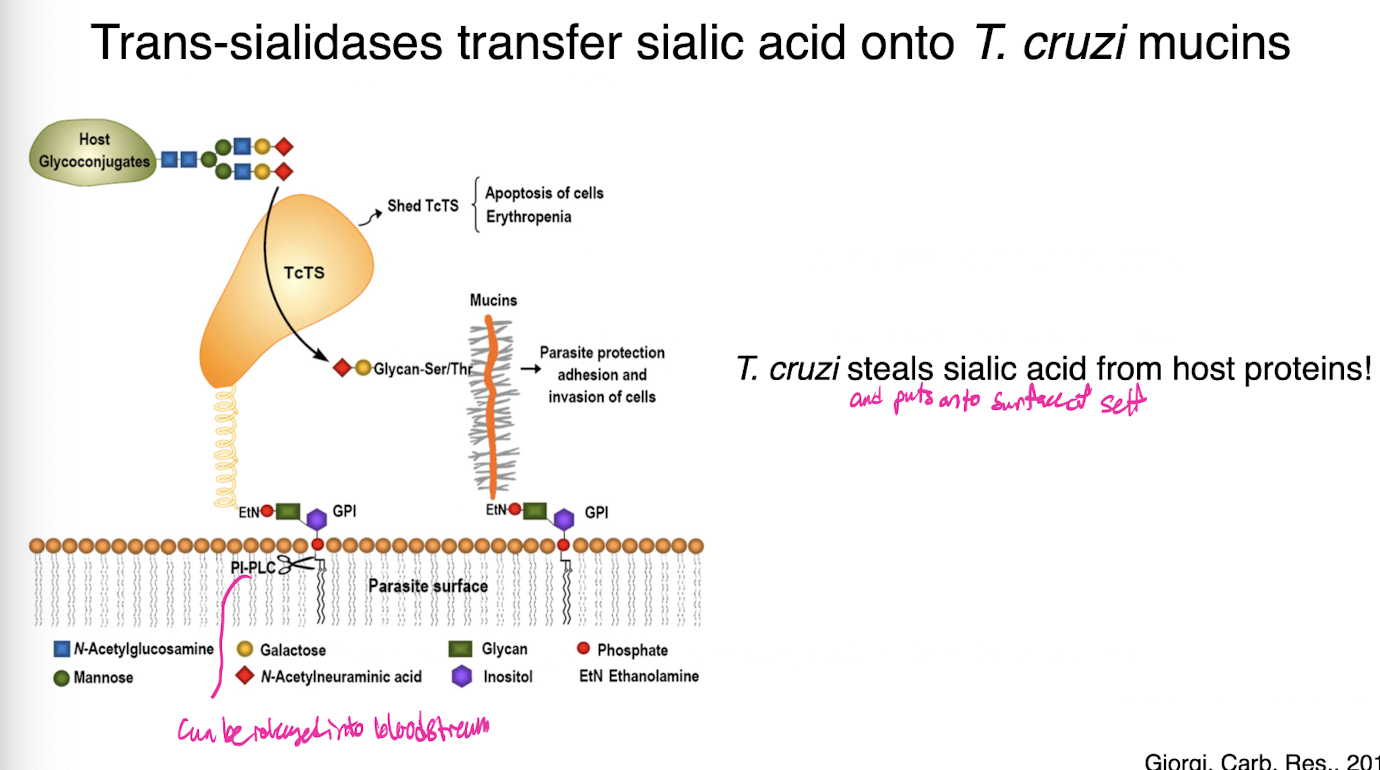
transfer silica acid onto t cruzi mucins
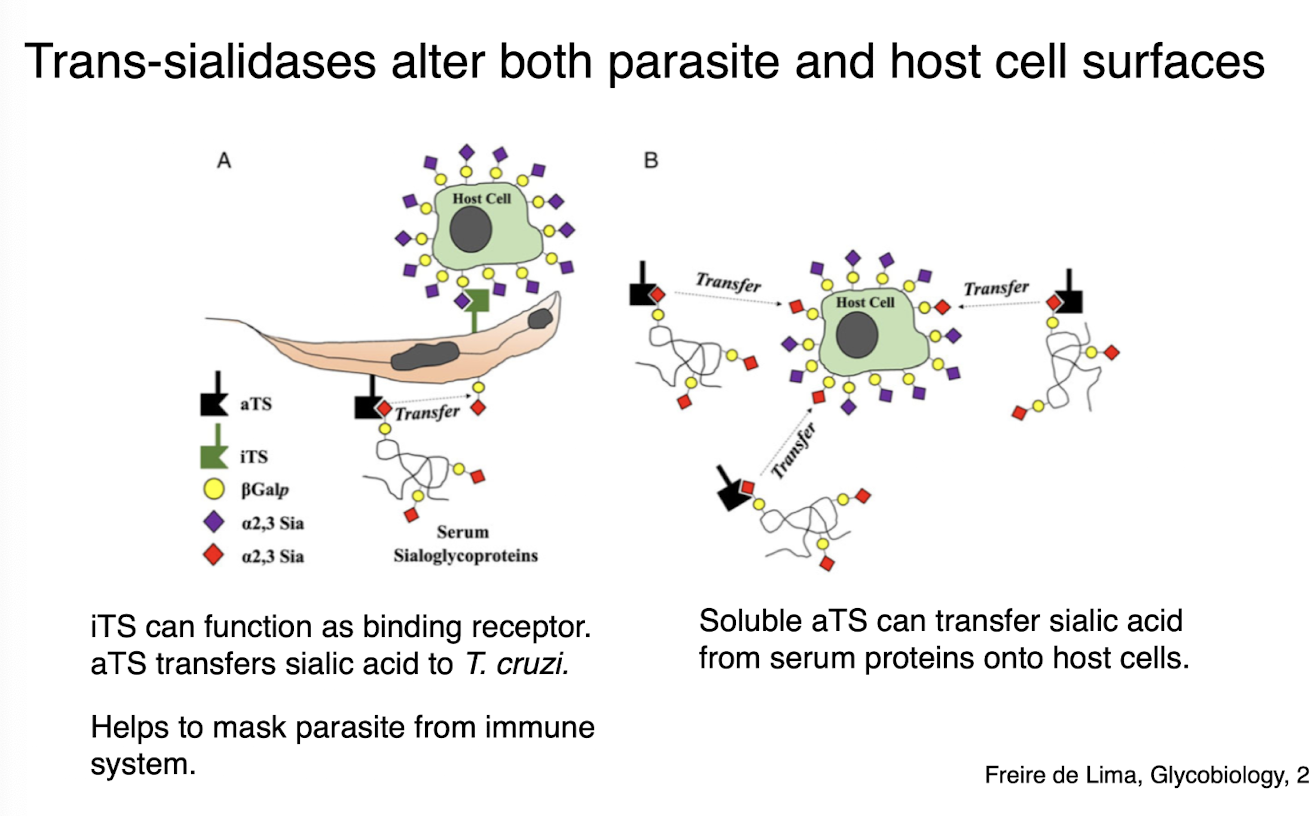
alter both parasite and host cell surfaces
trans-sialidases and immune system
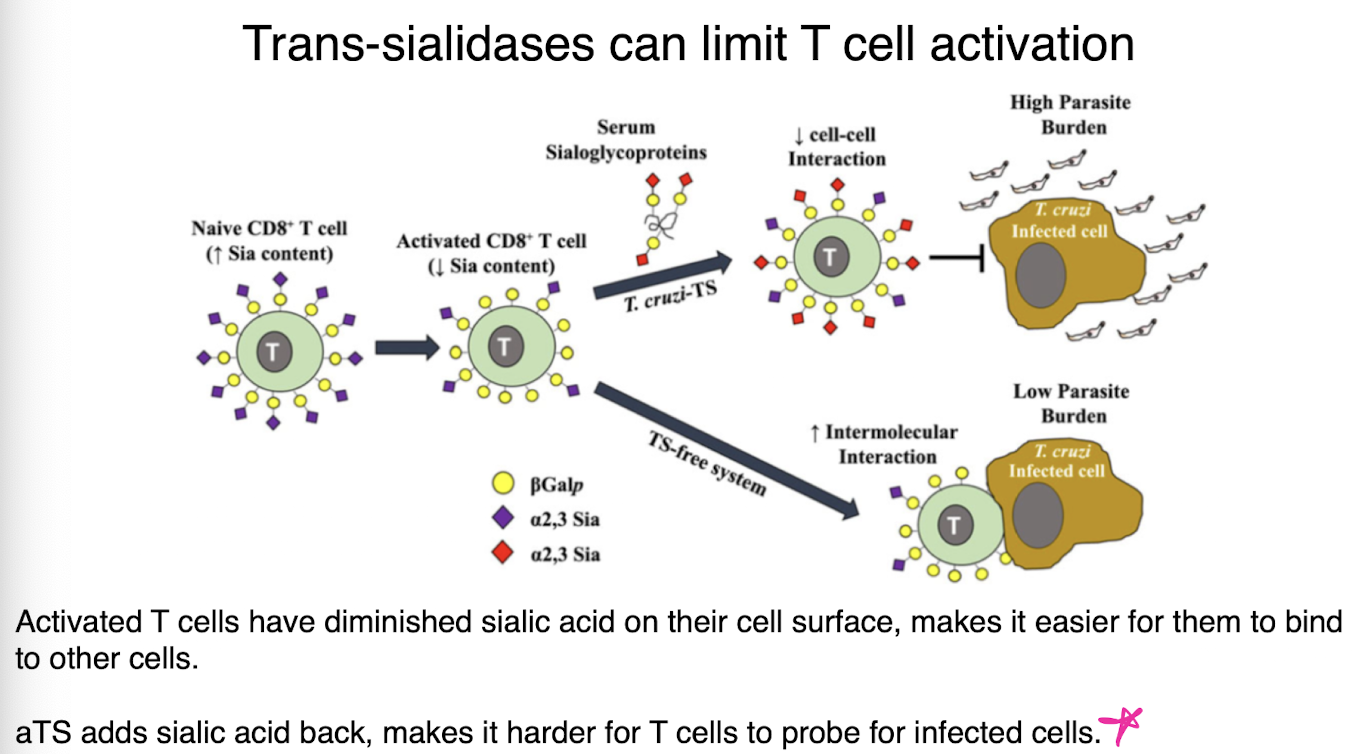
can limit T cell activation
activated T cells have diminished silica acid on their cell surface, makes it easier for them to bind to other cells
aTS adds silica acid back, makes it harder for T cells to probe for infected cells
t cruzi cell infection facilitated by
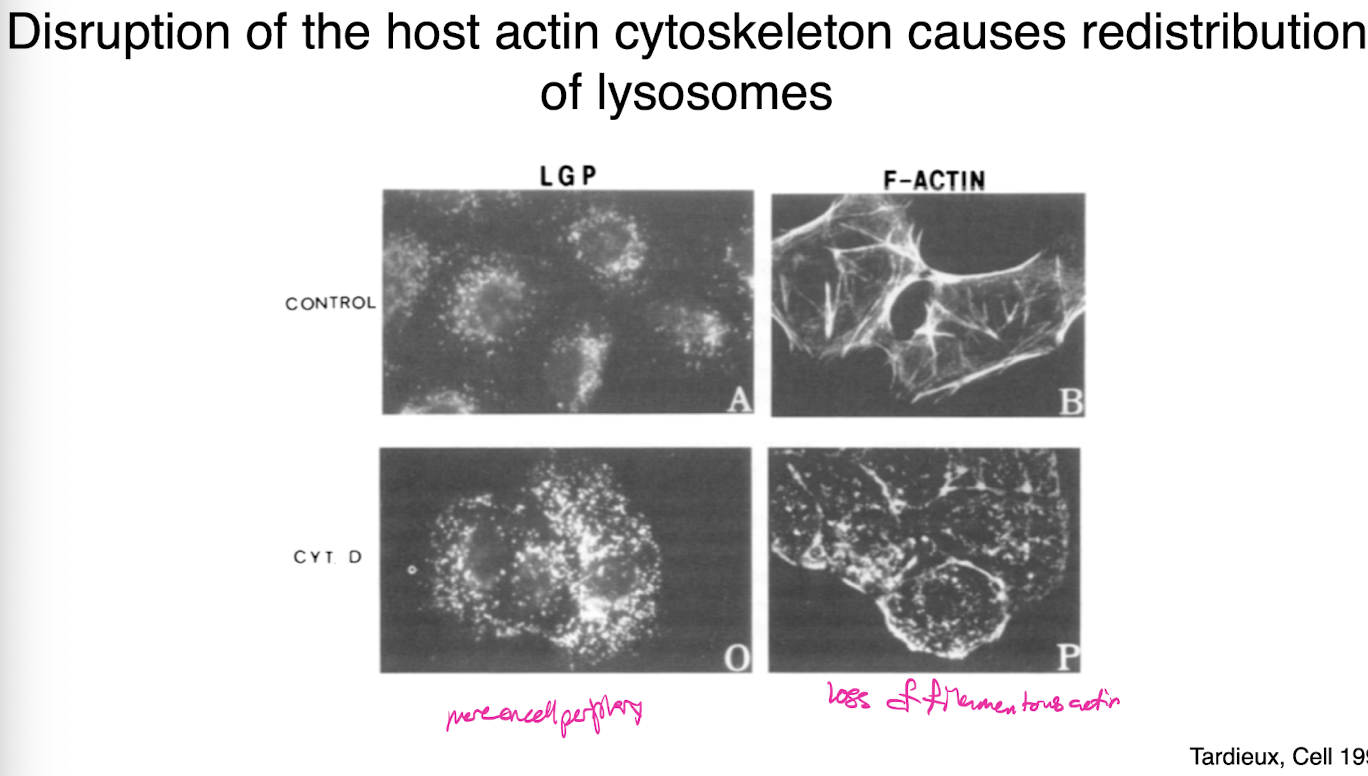
disruption of host actin cytoskeleton
cytochalasin D - blocks formation of actin filments
most pathways for pathogen internalization employ phagocytosis, which requires actin filaments
disruption of host actin cytoskeleton causes redistribution of lysosomes
lysosomes
acidified compartments containing hydrolyzing enzymes
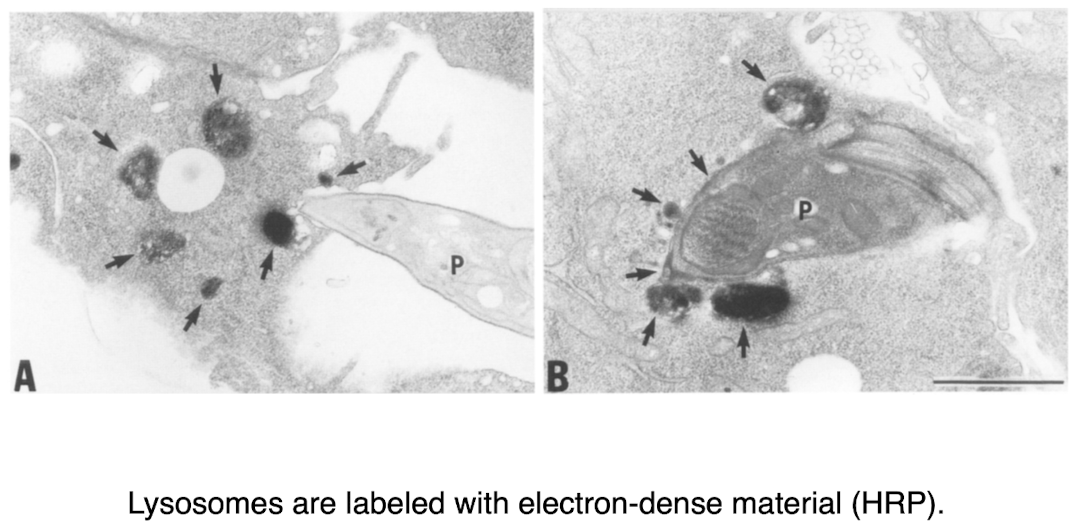
disrupted host lysosome t cruzi
disrupting host lysosomal fusion limits t cruzi infection
filling lysosomes with sucrose makes them swell and limits their ability to fuse to one another
attachment of t cruzi to host cells was not affected
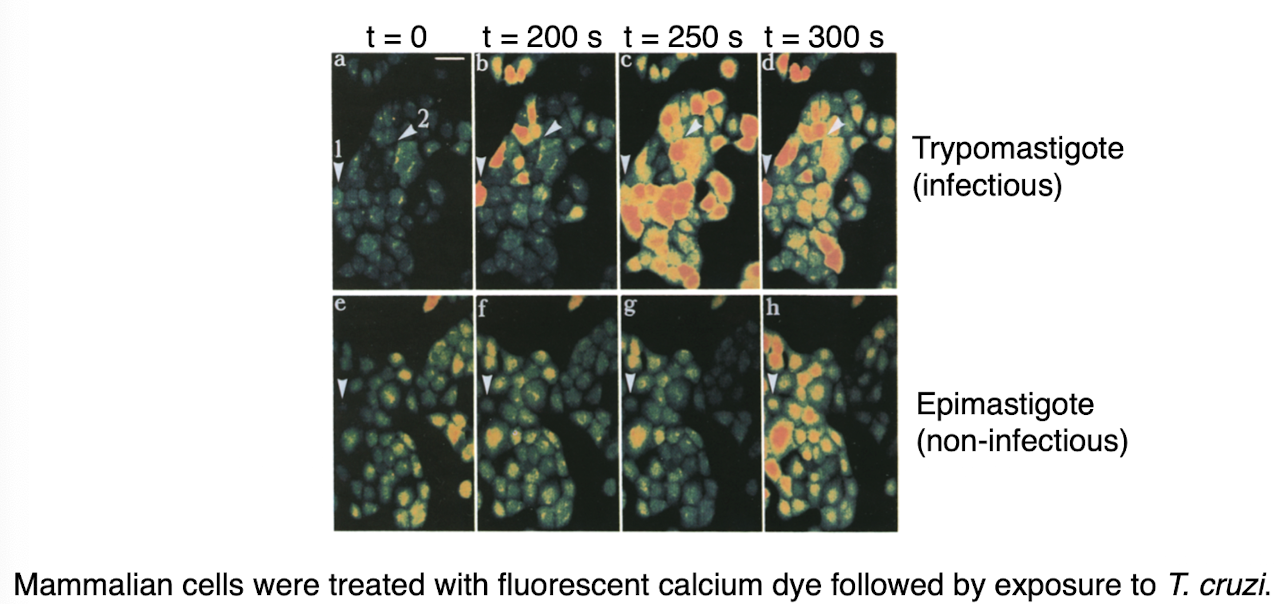
t cruzi early infection
associate with lysosomes very early in invasion process
t cruzi trypomastigotes
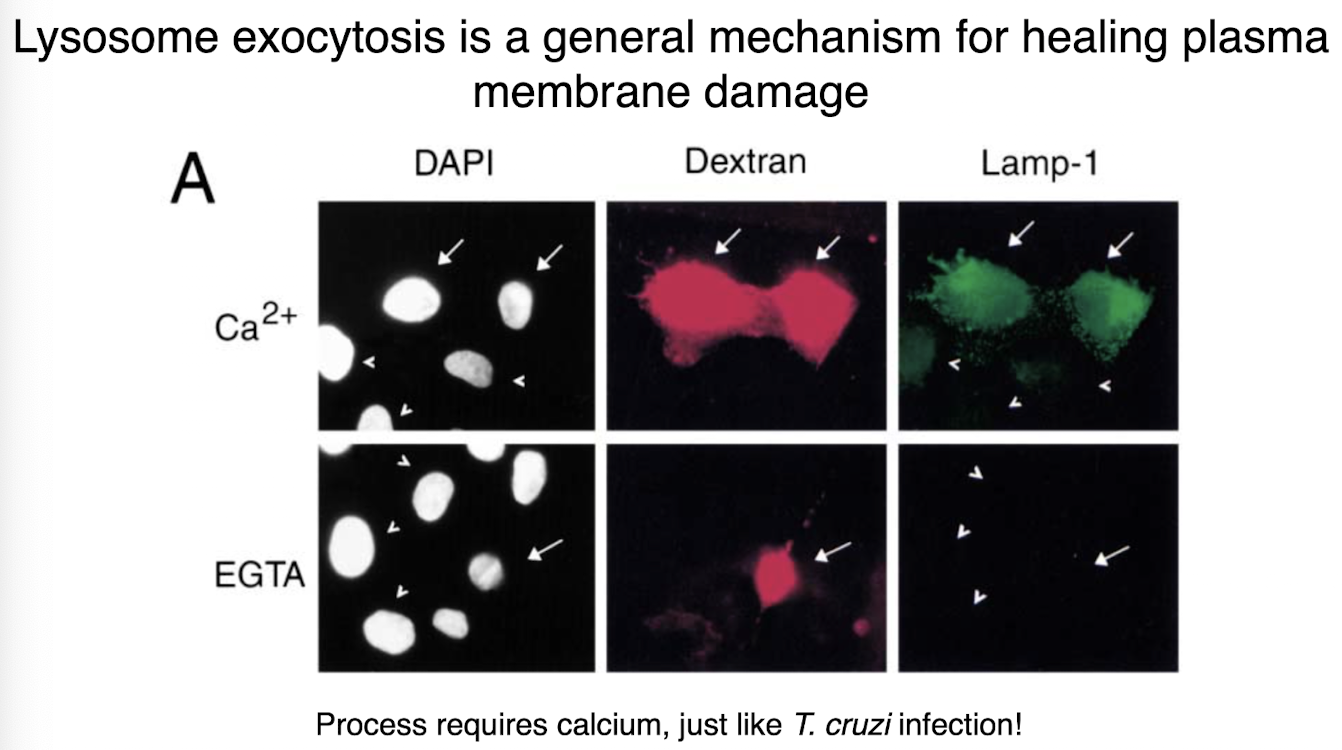
trigger changes in host cell cytosolic calcium levels
cannot enter host cells without extracellular calcium
trigger host cell death in absence of calcium
trigger lysosome fusion with plasma membrane
lysosome exocytosis
general mechanism for healing plasma membrane damage
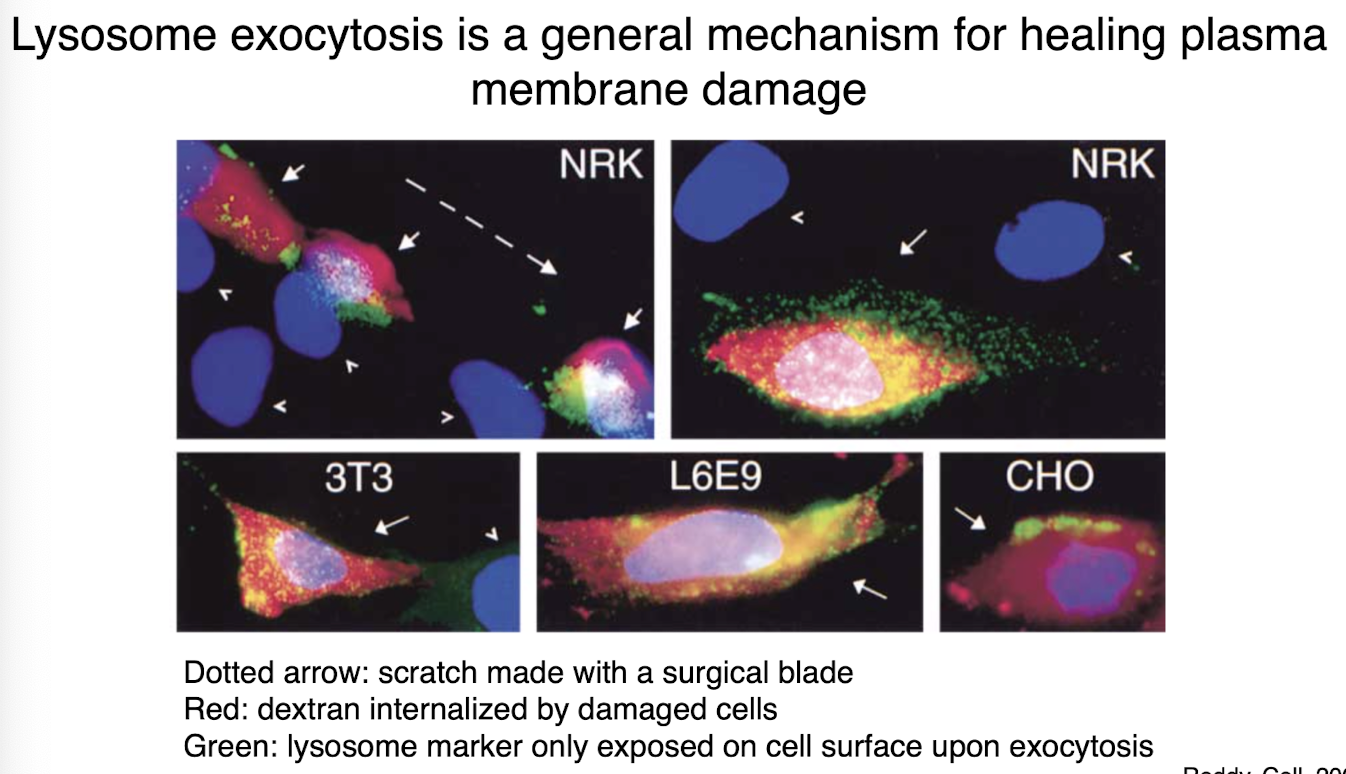
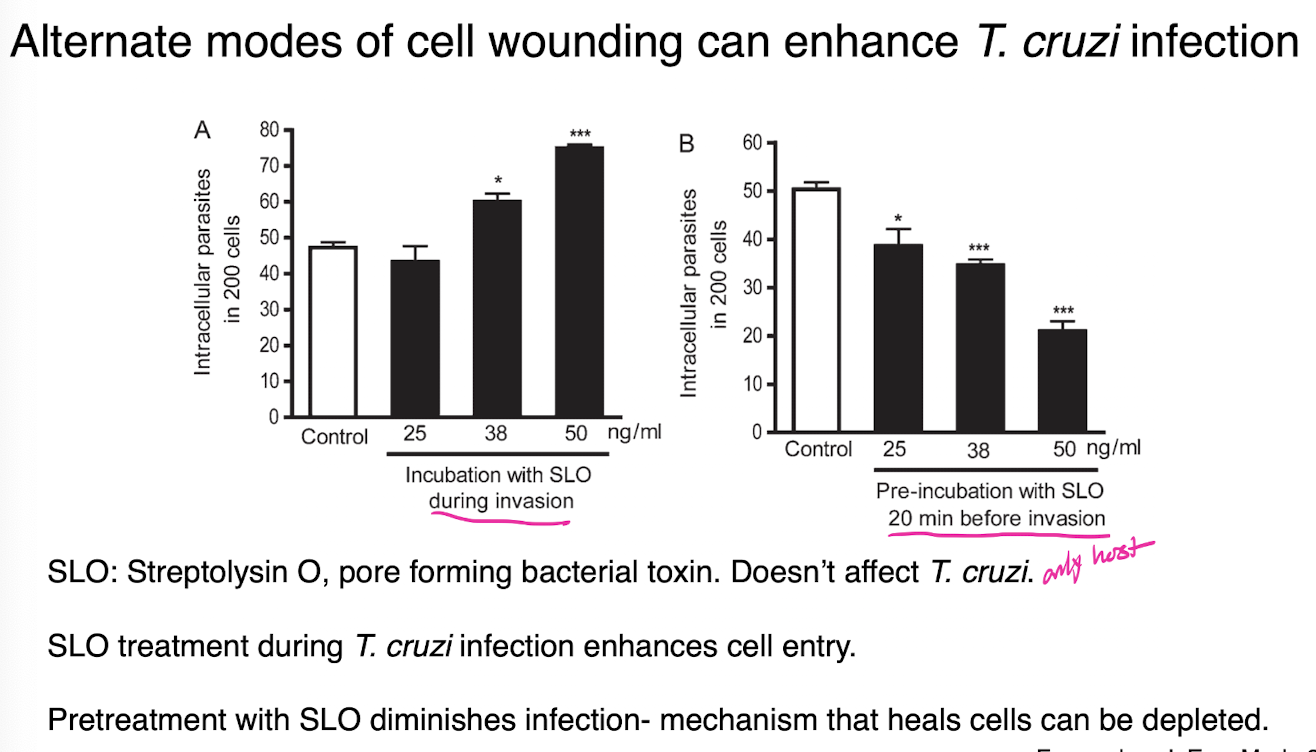
alternate modes of cell wounding t cruzi
cell wounding causes release of lysosomal acid sphingomyelinase
acid sphingomyelinase activity is necessary for t cruzi infection
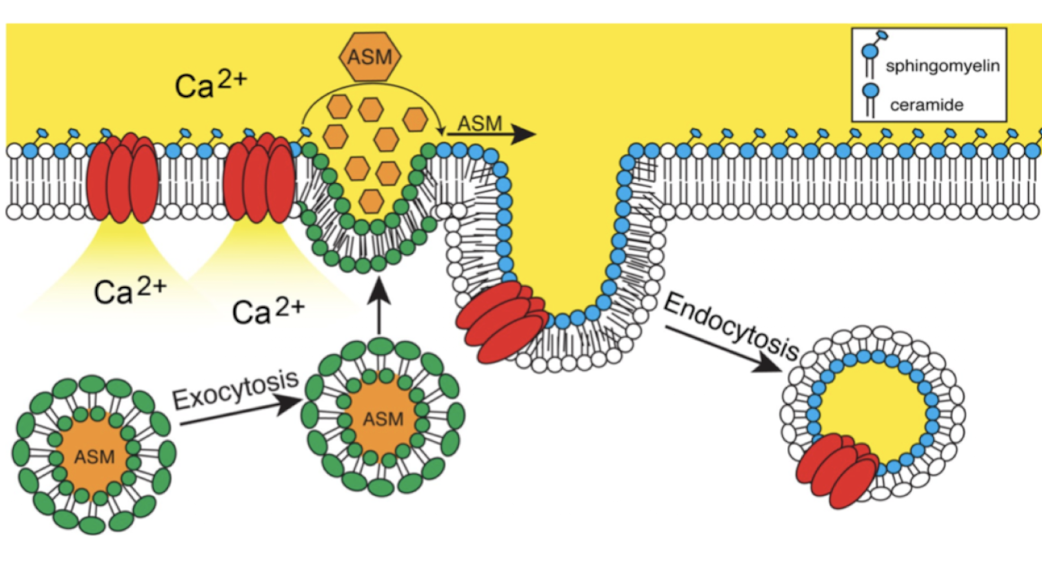
t cruzi cell wounding
triggers rapid membrane healing pathway
wound triggers calcium influx, leads to lysosomal fusion at plasma membrane, release of ASM, production of ceramide, and rapid endocytosis
extracellular t cruzi trypomastigotes wound cells
enter cells posterior first
intracellular t crudi remain highly motile
protruding t cruzi
still encased in host membrane
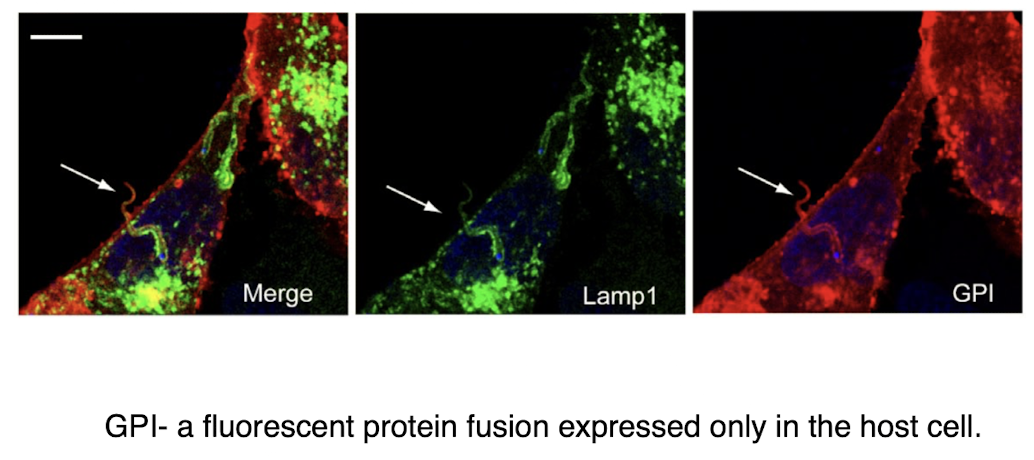
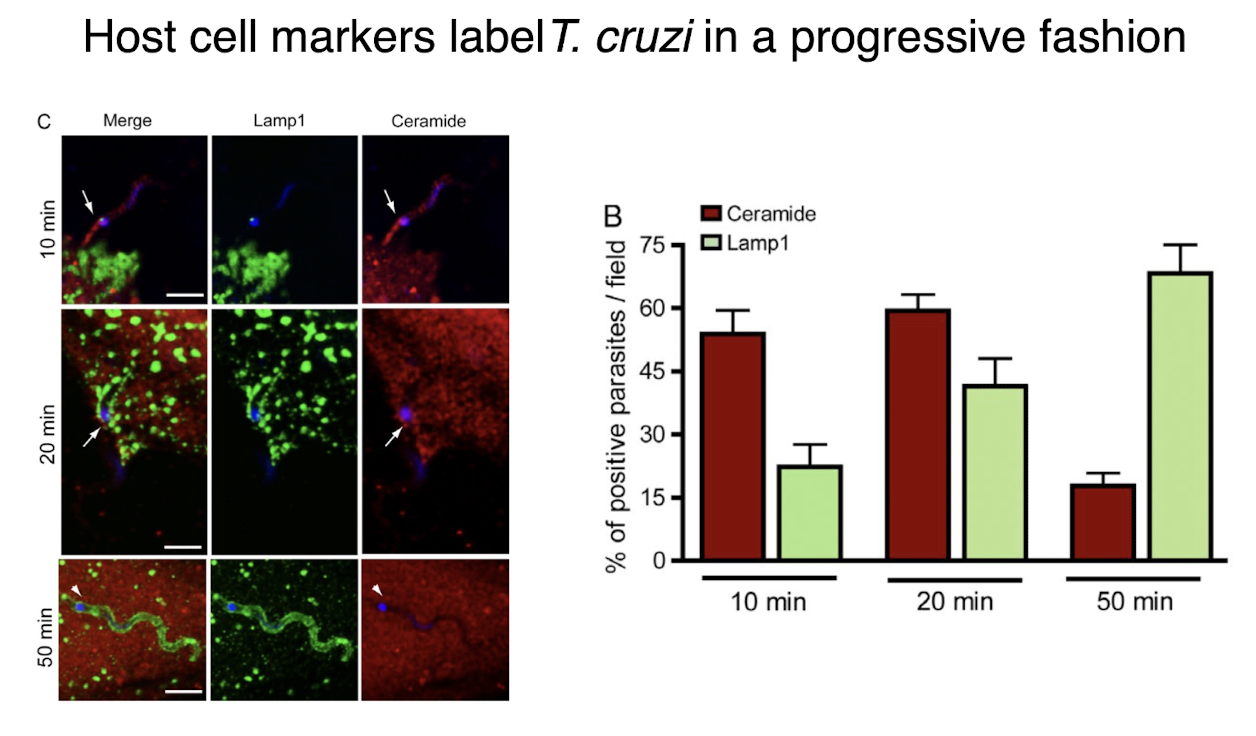
host cell markers t cruzi
label them in progressive fashion
cells early in infection process are labeled with ceramide, but not LAMP
cells later in infection process are LAMP positive, but lack ceramide
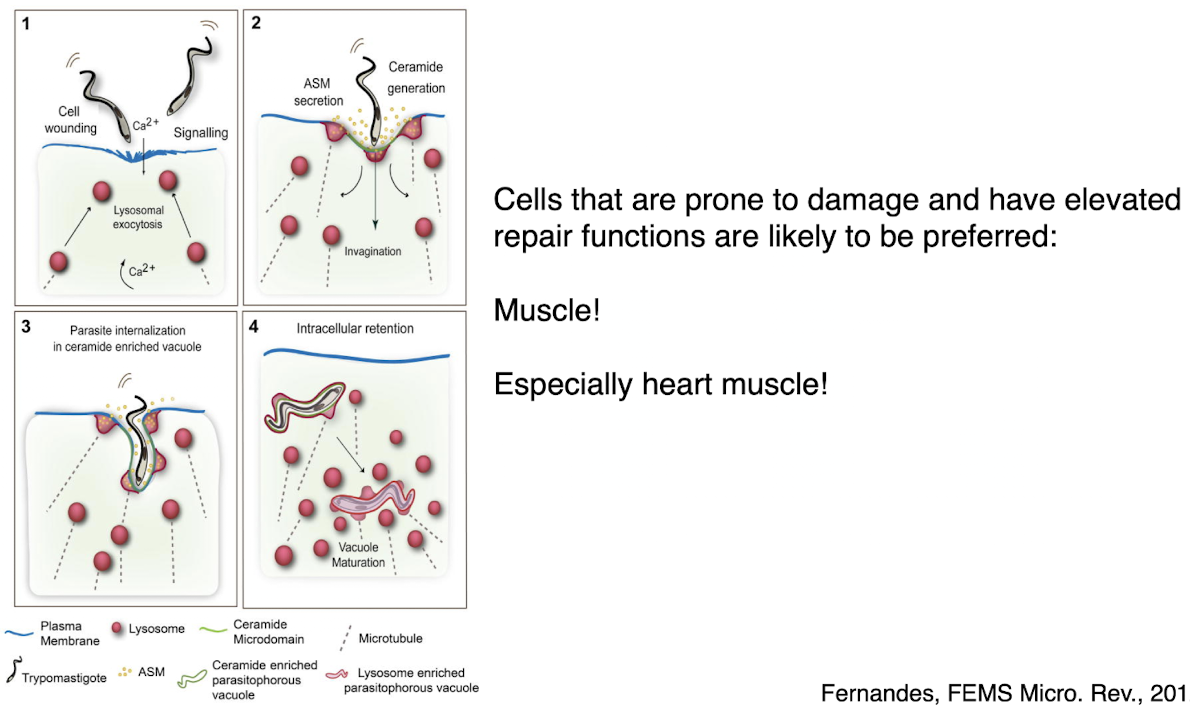
t cruzi cell invasion mechanism
cells that are prone to damage and have elevated repair functions are likely to be preferred: muscle and heart muscle
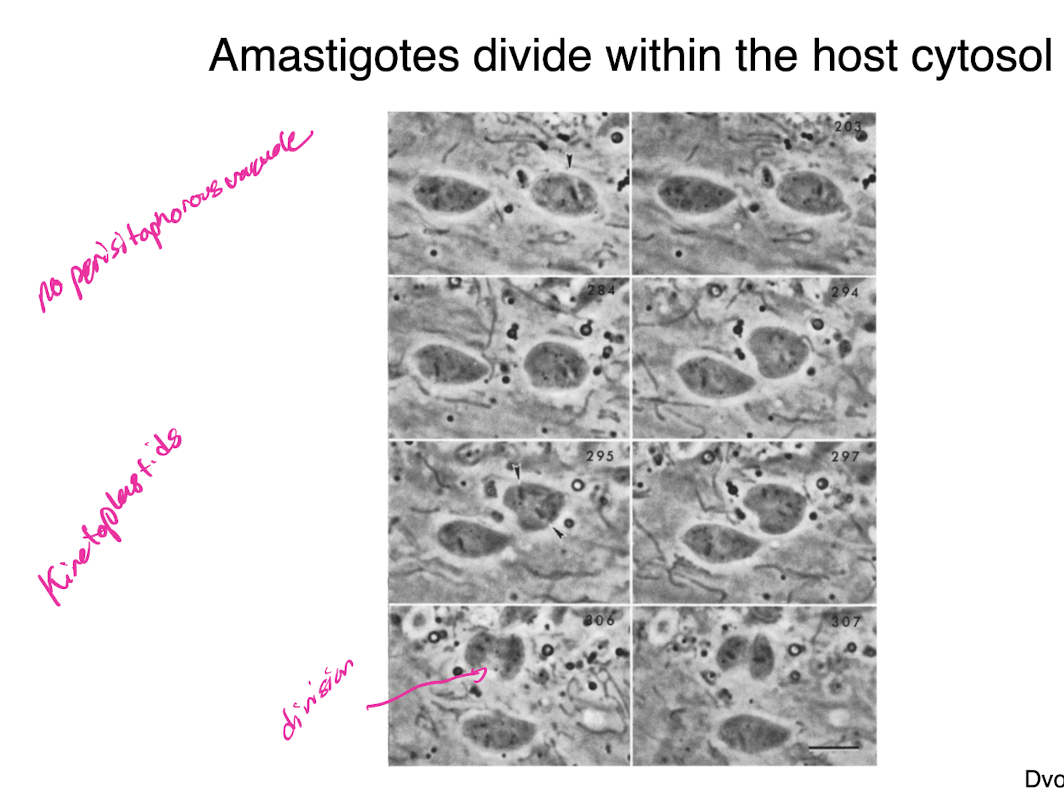
t cruzi amastigotes
divide within the host cytosol
become amastigotes from the trypomastigotes and can be triggered to do this conversion
no parasitophorous vacuole
t infestans in arequipa
is present more in urbanized parts than rural
spreads very slowly in urban setting
does not become proficient for transmission of t cruzi within a community effectively until a certain host density is reached
developing areas in contact with environment are LESS likely to be infested, develop t cruzi infection
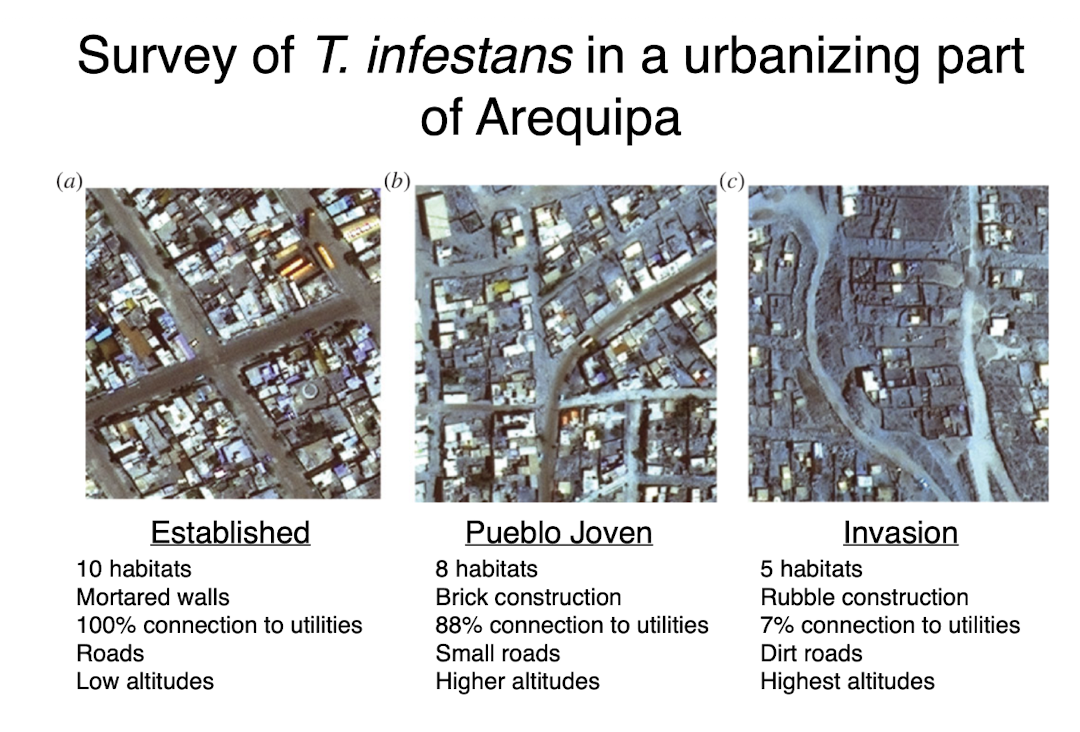

t cruzi in arequipa
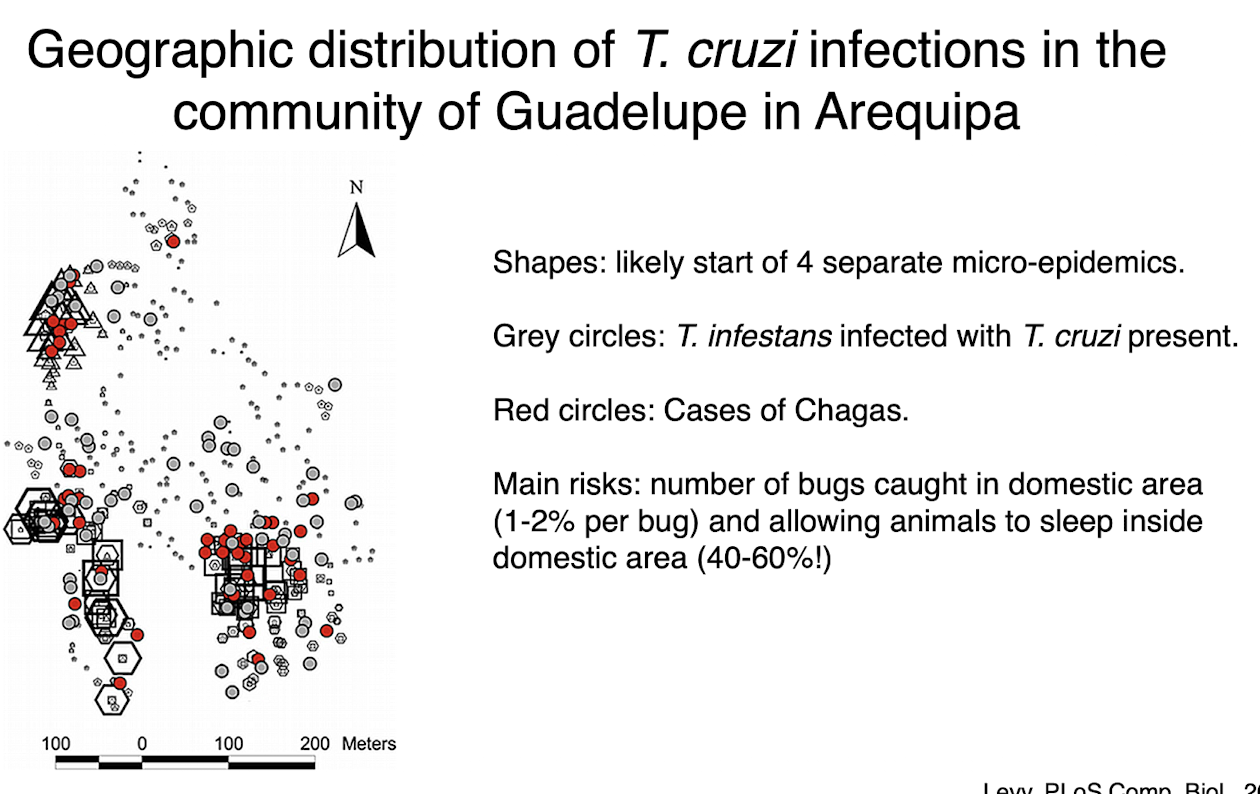
rates of t cruzi infection reached 5% of population before insect control introduced
earliest infection only 20 years old
low levels of chagas cardiomyopathy but likely a lot more in future
prophylactic drug treatment may be good before heart issues arise
benznidiazole clearing of parasites
does not clear all parasites in mice
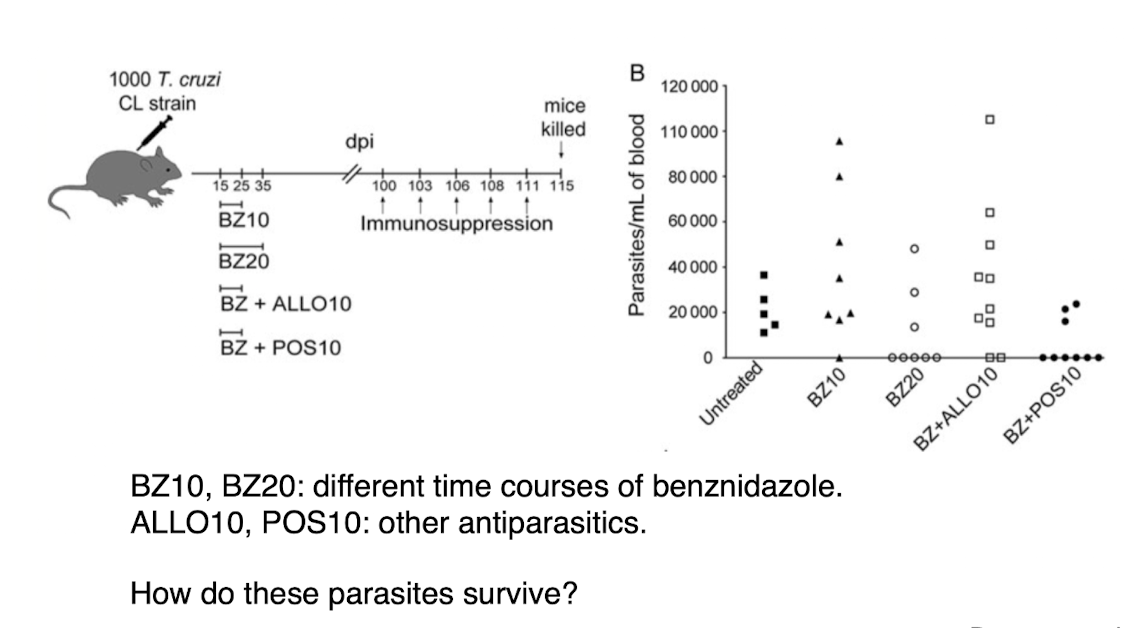
prolonged nifurtimox
selection identifies resistant strains of t cruzi
can select for resistant epimastigotes, but no longer mammalian-cell infectious
prolonged benznidazole
election identifies resistant strains of t cruzi
all clones show greatly diminished infection of mammalian cells
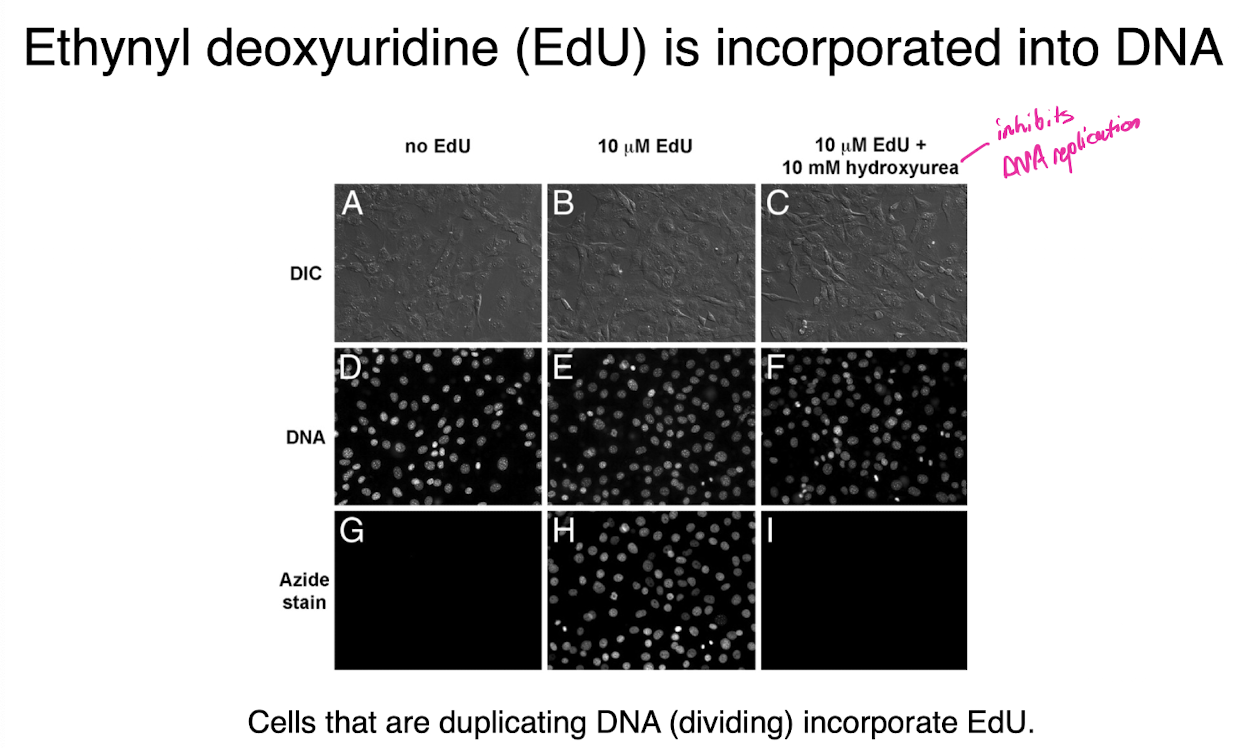
edu pulse
ethynyl deoxyuridine is incorporated into DNA
takes place of dT
ethynyl group can be selectively reacted with azides to produce covalent bonds, lots to see if DNA replication is occuring
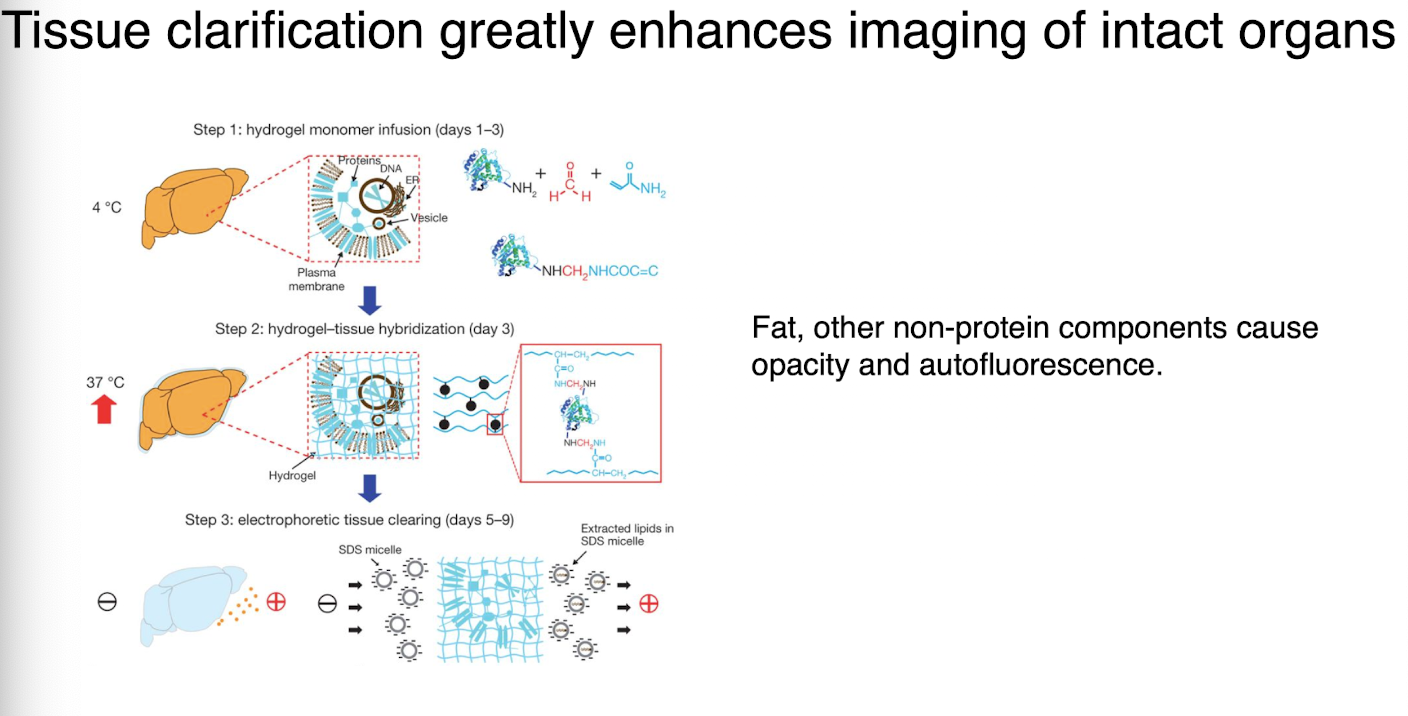
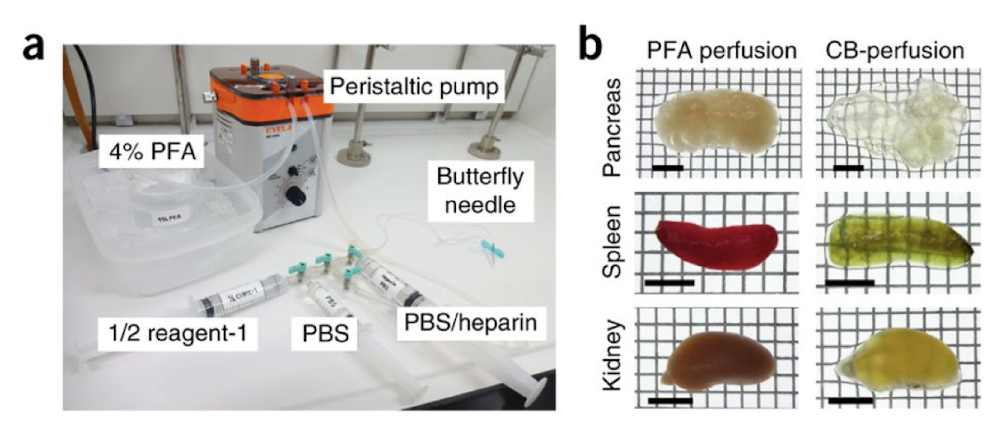
tissue clarification
greatly enhances imaging of insect organs
fat, other non-protein components cause opacity and autofluorescence
t cruzi amastigotes in mice
become dormant in mice and tissue culture cells
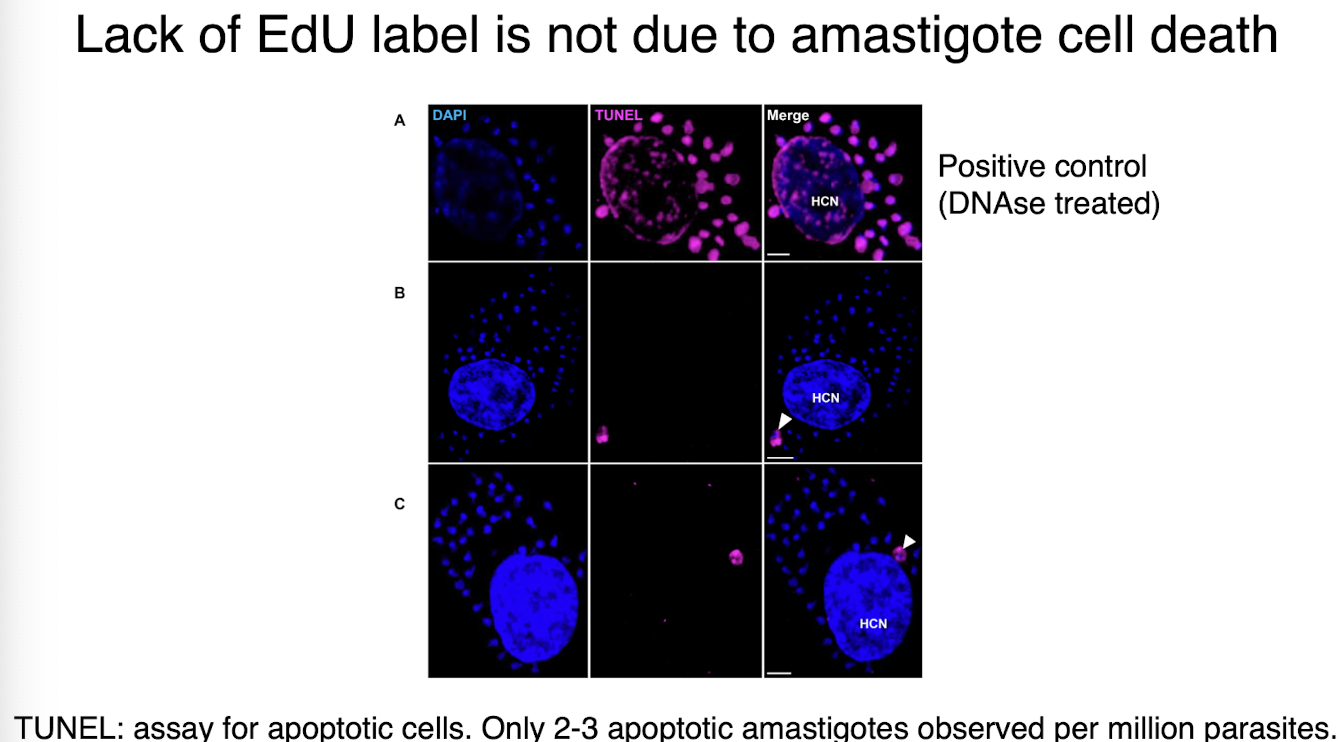
lack of EdU label is not due to amastigote cell death
TUNEL: assay for apoptotic cells, only 2-3 apoptotic amastigoes observed per million parasites
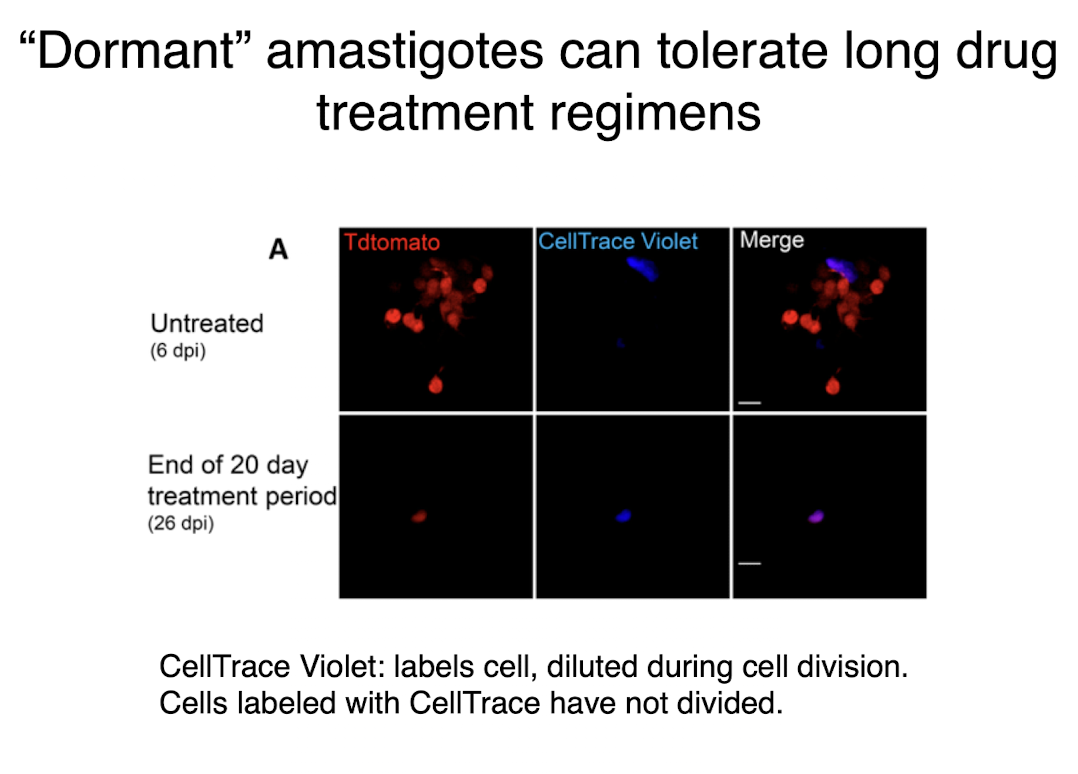
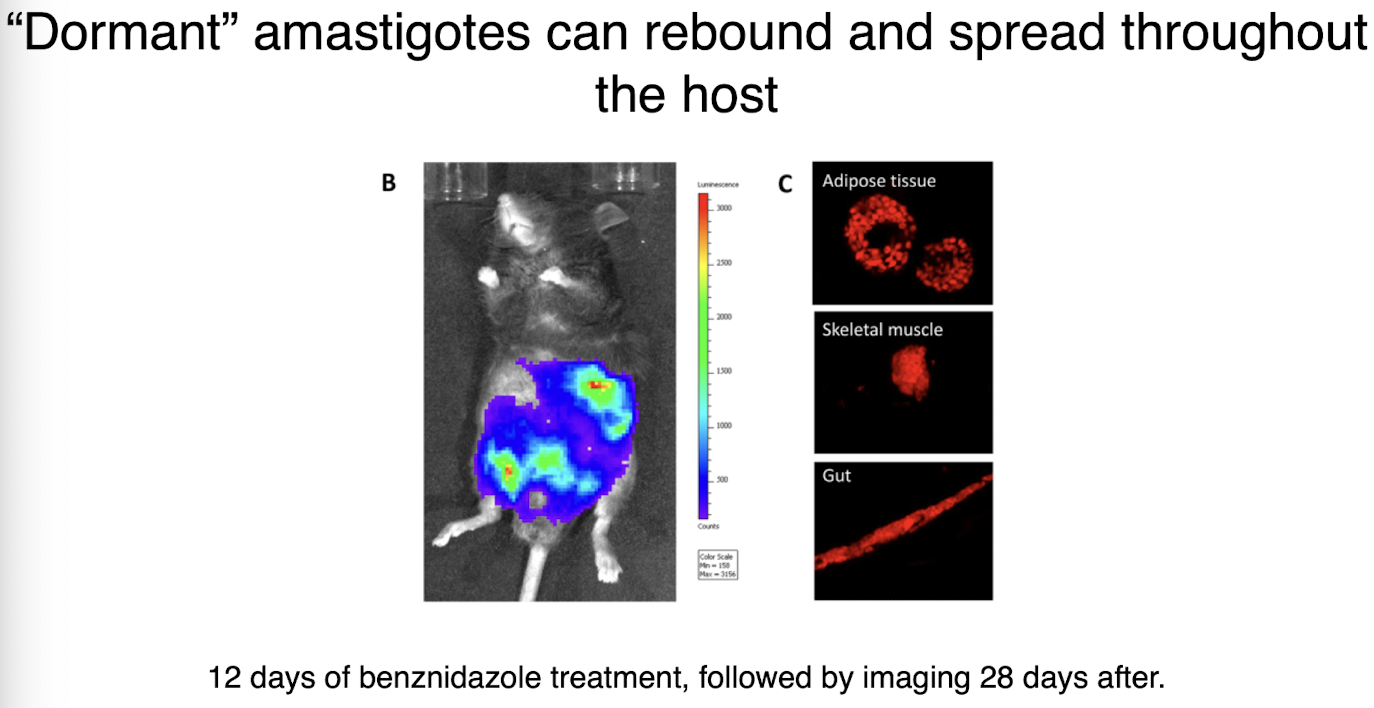
dormant amastigotes t cruzi
can tolerate long drug treatment regimens
can rebound and spread throughout the host
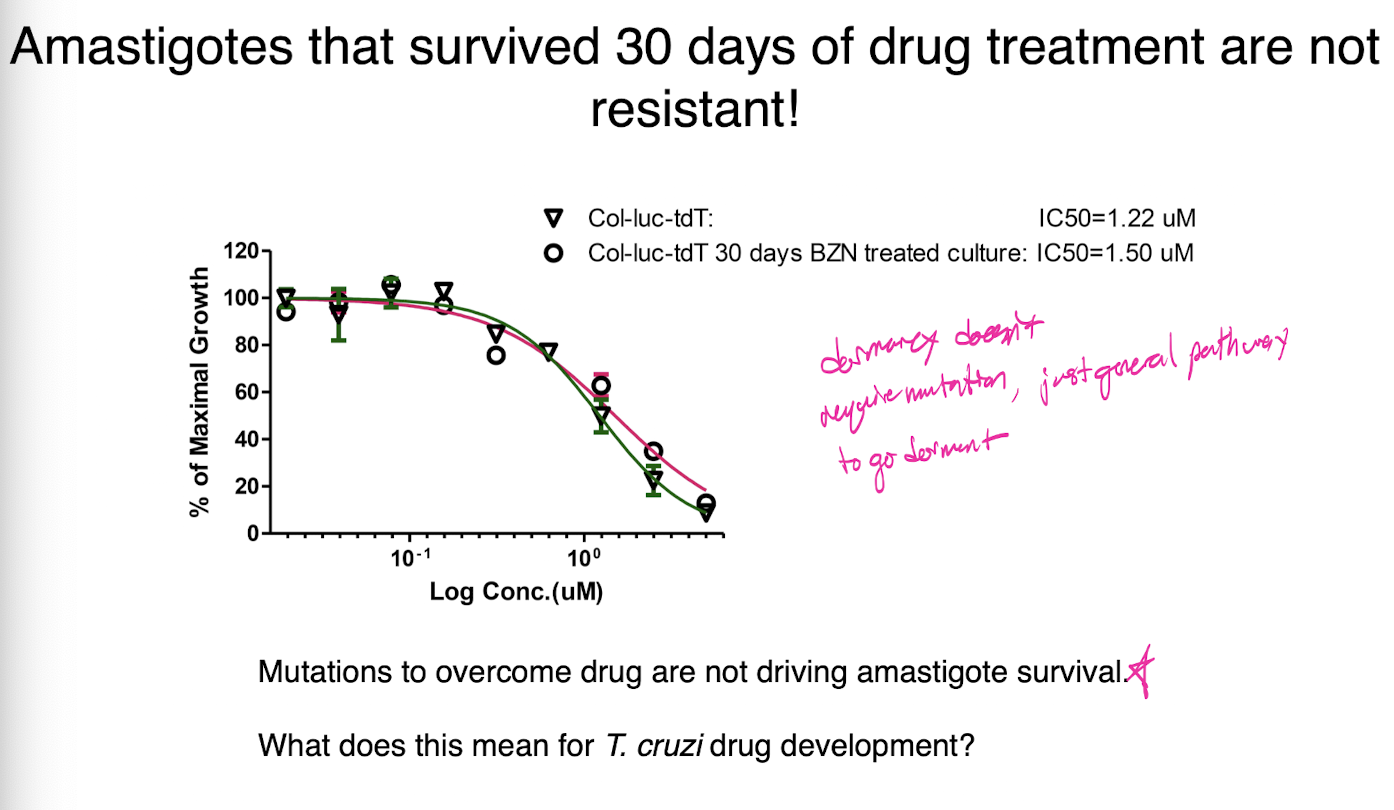
amastigotes that survived 30 days of drug treatment are not resistant
mutations to overcome drug are not driving amastigote survival
dormancy has limits though because extended courses of high-dose BNZ clears the infection
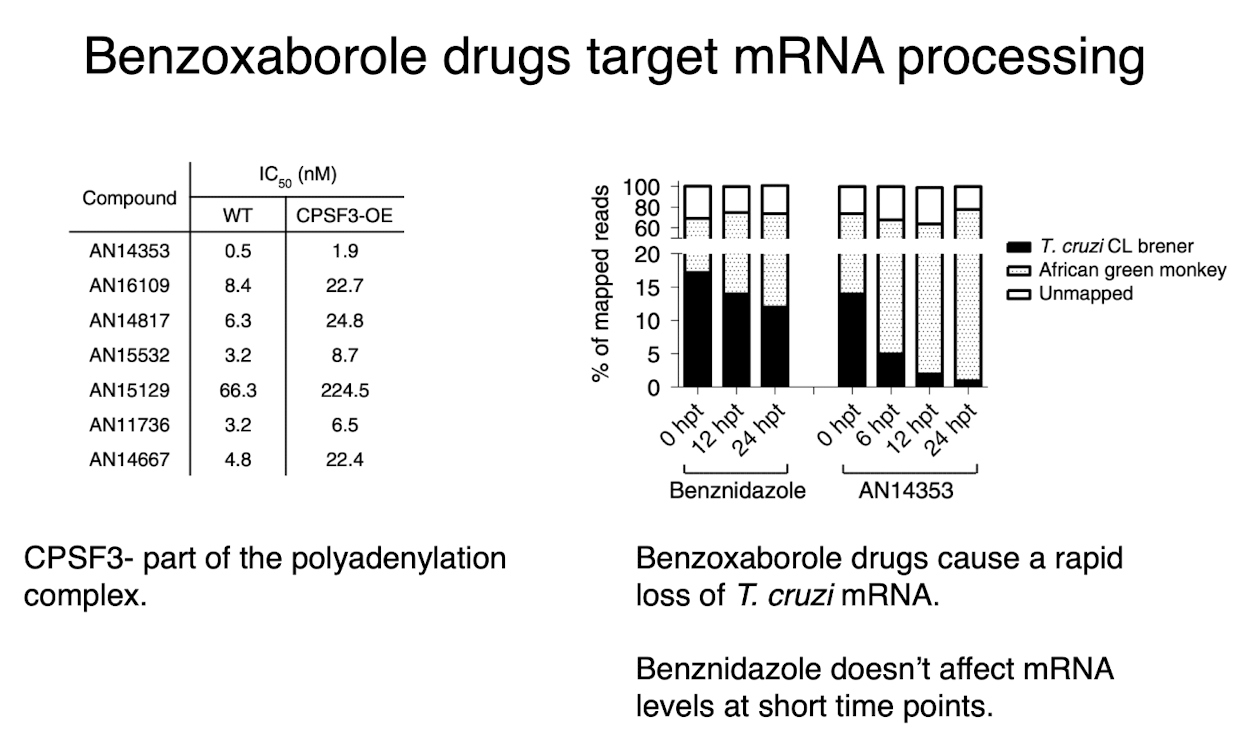
benzoxaborole
series of these drugs have potent activity against t cruzi
targets mRNA processing
benzoxaborole AN15368 is effective at clearing t cruzi infection in non-human primate model
ancient texts t brucei
describe wasting disease that kills livestock in Africa
Egyptians used to keep livestock with game animals
purebred livestock would die of wasting disease
Zulus called disease N’gana: useless/powerless
early sleeping sickness descriptions
first descriptions came from attendants on slaving ships sailing to americas
descriptions of people in coma-like state, alive but unable to communicate
Winterbottom’s sign - swelling of the lymph nodes int he neck precede the illness
David bruce
identified infectious agent that causes N’gana
noted that wasting disease affected horses, donkeys, dogs, and cows but had not effect on local species
essentially all animals died, only some cattle survived
was convinced tsetse fly was transmitting illness
took animals into tsetse infected regions to see if they would get infected
t brucei identification
Bruce showed bite of tsetse did not transmit poison
flies that had fed on infected animals could transmit
parasite was present in every sick host, but not healthy ones
fever in animals with nagana correlated with appearance of parasites
direct blood transfer from animal transmitted disease
he did think that mosquito was just a syringe to deliver which is not true
Forde
identified trypanosomes in humans
master of steamship River Gambia was admitted with a fever, lethargy
malaria was diagnosed, but quinine did not help fever
blood smear showed trypanosomes in blood
other trypanosomes
t congolense and t vivax cause nagana but don’t infect humans
t brucei rhodiense infects humans and causes a more sever disease than t brucei gambiense
trypanosomes were isolated from cerebrospinal fluid, suggesting how the parasite might cause the coma-like stage of disease
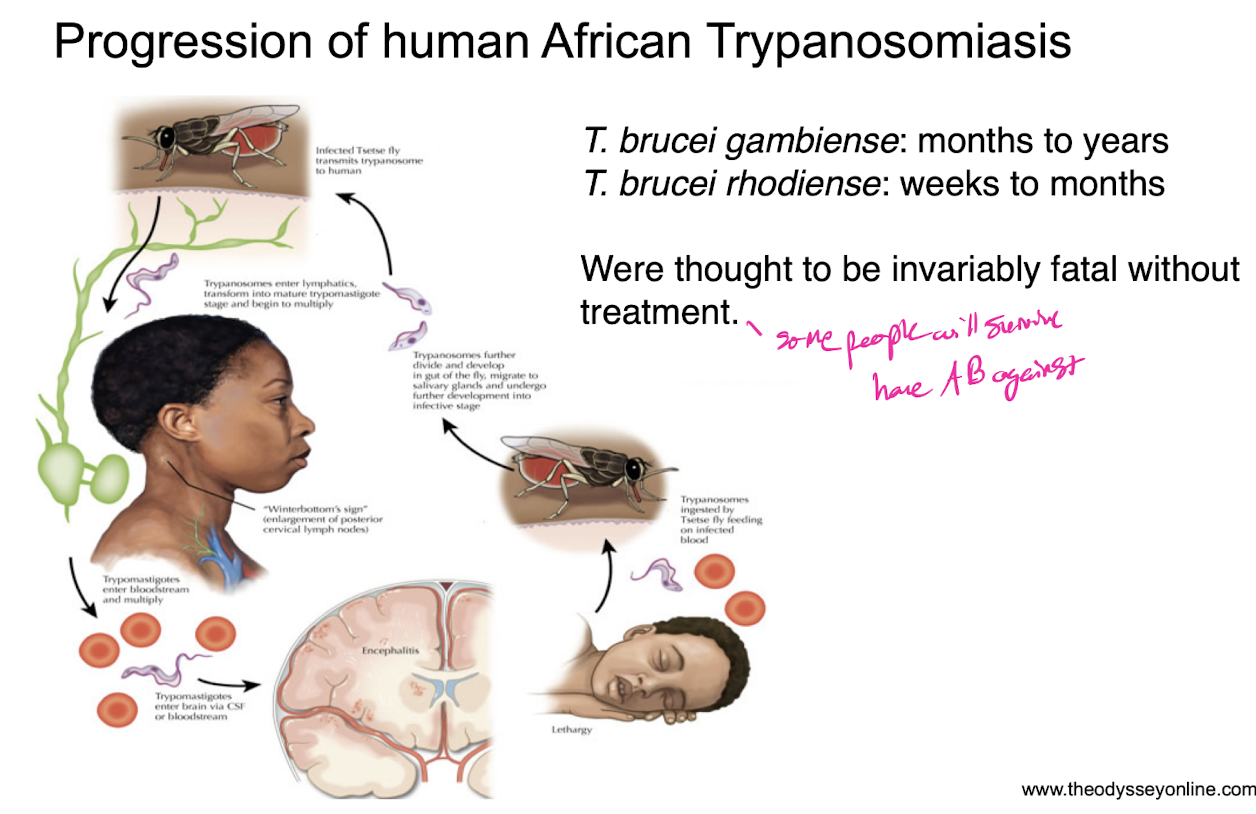
t brucei life cycle
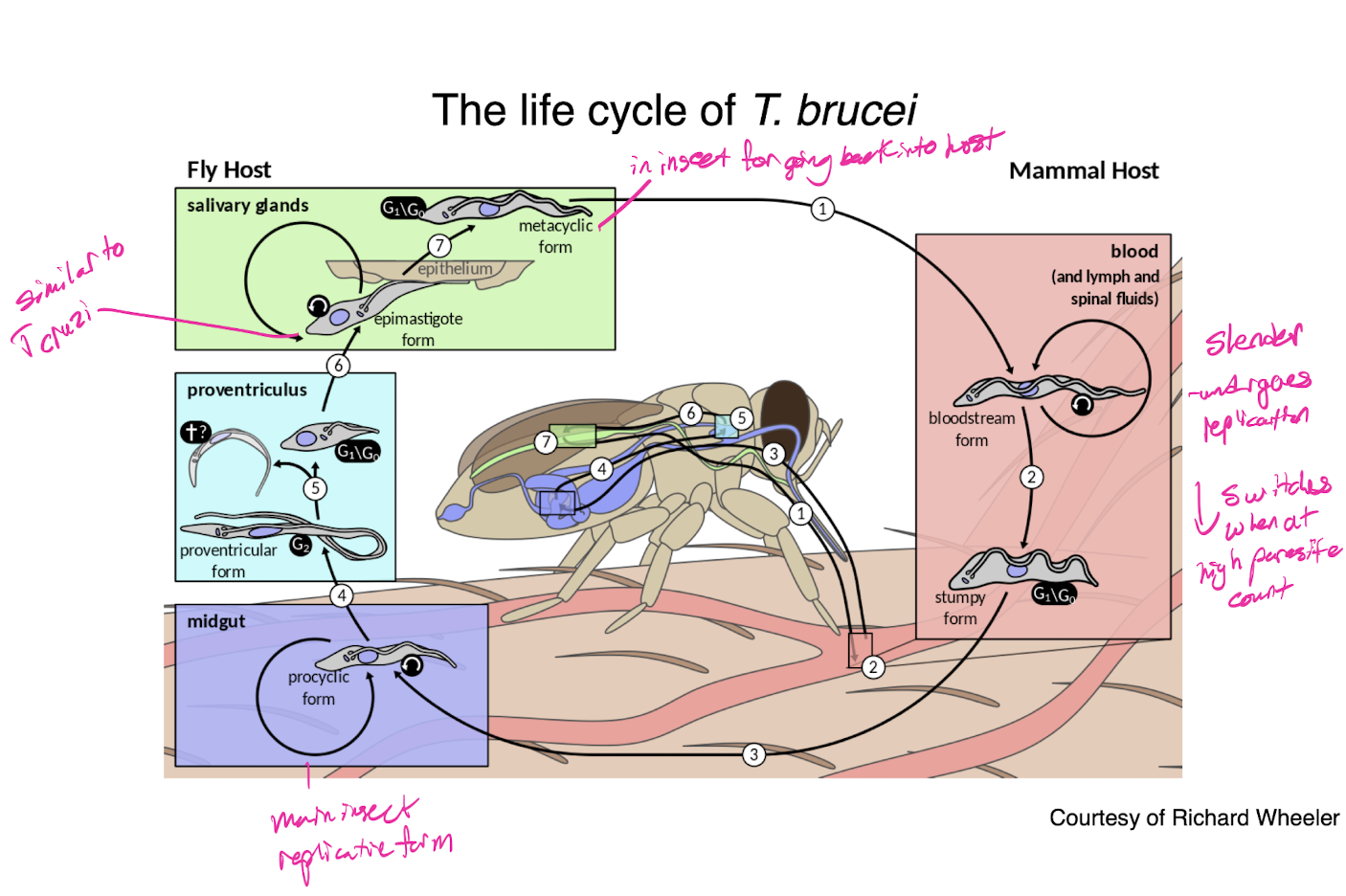
african trypanosomiasis progression in humans
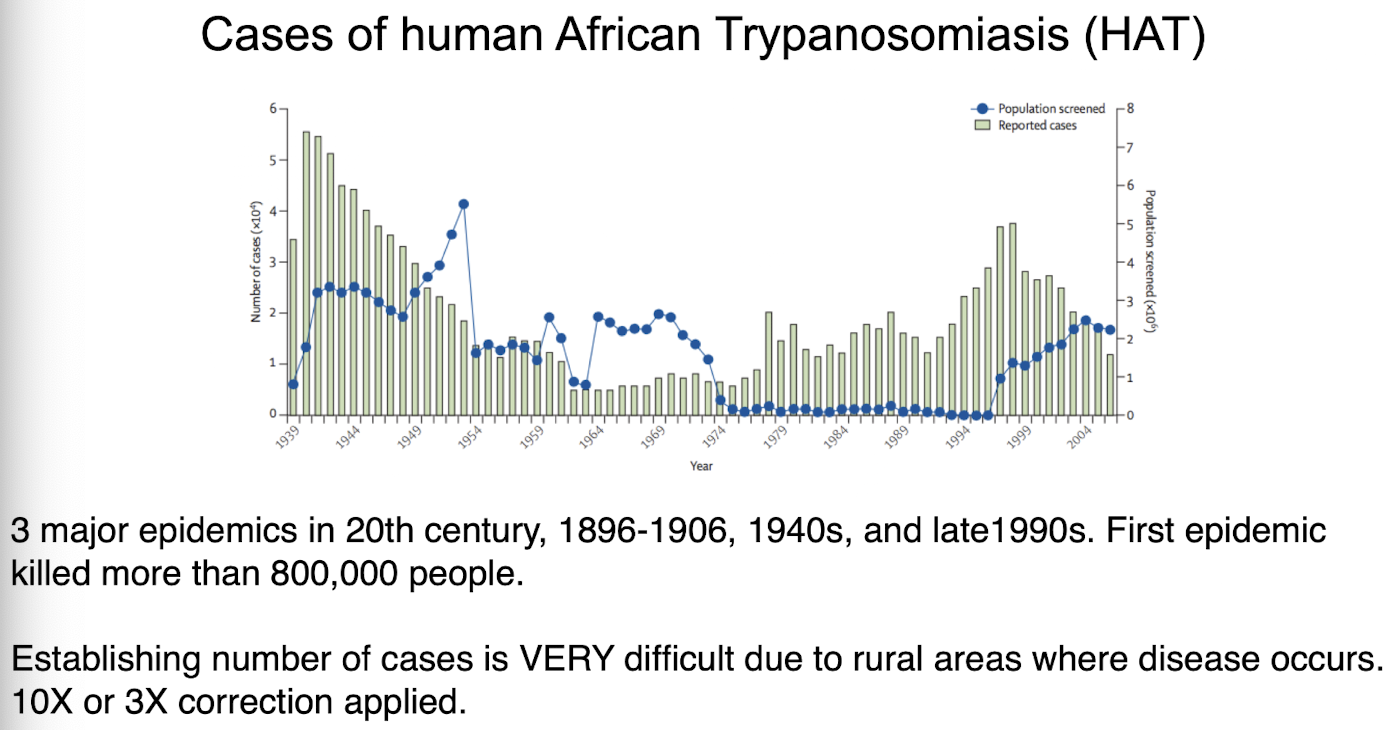
cases of HAT
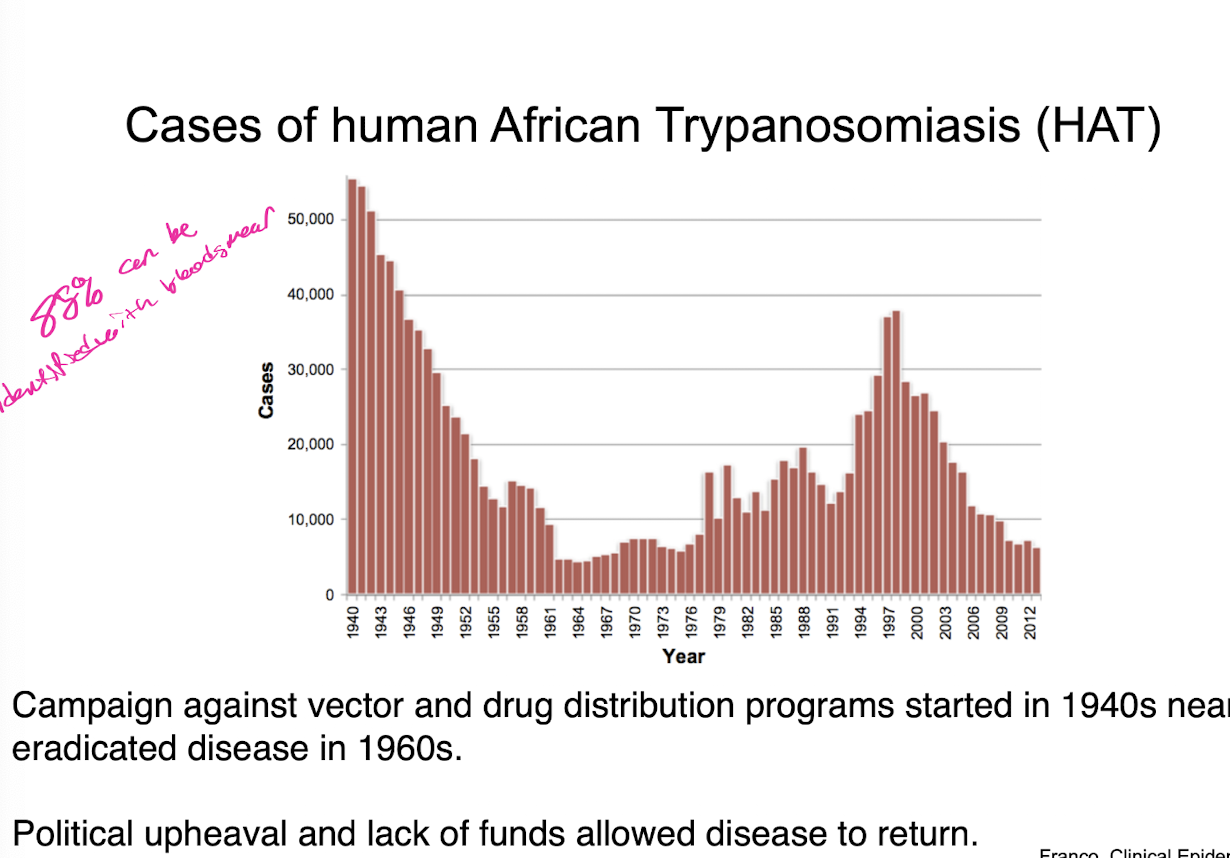
t brucei agricultural cost
livestock essential source of income for many families in sub-saharan africa
calculated cost of infection is around $2.5 billion, loss of 16% meat and 10% milk production
treating infected cattle is difficult, but suppressing tsetse fly using insecticides in effective
cattle function as important reservoir for t brucei rhodiense
drugs for HAT
trypan blue - effective for killing lab animals but turns them blue
1917: bayer 205 identified, colorless, very trypanocidial, still in use today for early-stage T. b. rhodiense infections. suramin
arsenicals - arsenic containing
ataxy: developed in 1960s, somewhat effective but blinds 2% of patients
melarsoprol: no blinding, effective against brain-stage HAT. causes severe encephalitis in 5-10%, kills 1-5%, damages veins when administered. still in use
nifurtimox: nitro prodrug, used to treat chagas
elfornithine: inhibitor of ornithine decarboxylase, blocks synthesis of polyamides in cells. originally chemotherapeutic. low toxicity, very effective
elfornithine
nearly lost as HAT treatment
rights to drug were sold several times
failed as chemotherapeutic, eventually sold for $750 per treatment, but eventually sold as a hair removal product
Doctors Without Borders and trypanosome researchers pushed BMS to provide free eflornithine for HAT
NECT treatment
for trypanosomiasis
combined elfonithine and nifurtimox
fexindazole
important treatment for HAT
orally bioavailable, 10-day course
low toxicity, as effective as NECT and doesn’t cross blood brain barrier
could significantly improve treatment course
still requires hospitalization because you need to make sure patient doesn’t have parasites in CSF. lumbar puncture
acoziborole
improved treatment for HAT
targets CPSF3- part of polyadenylation pathway
same target as compound for treating t cruzi
orally available, crosses blood-brain barrier, single-does, can use prophylactically, no need to monitor patients
3 mutations in CPSF3 can increase resistance
tsetse fly
gives live birth
feed “milk” to maggot, produces one at a time. needs additional blood meal to produce another offspring, takes about 9 days
susceptible to climate change and populations are rapidly declining
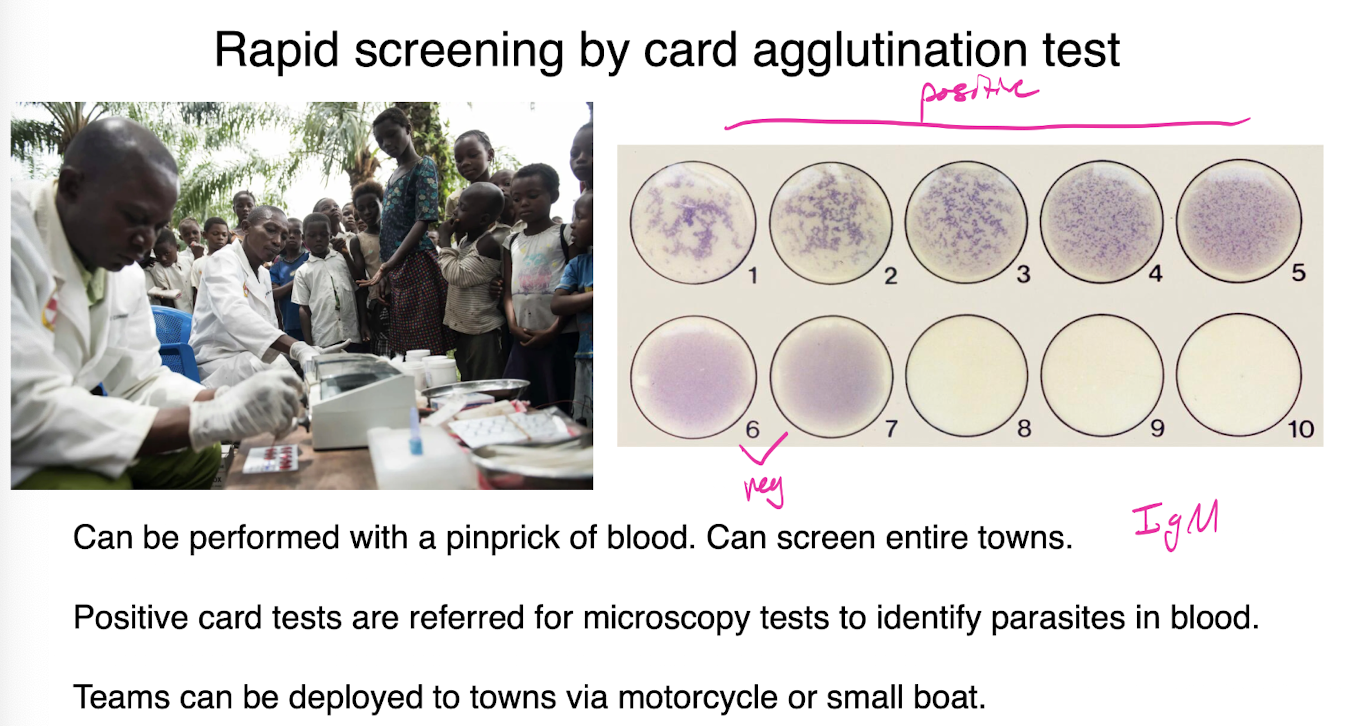
agglutination test
rapid screening for t brucei
can be performed with pinprick of blood, can screen entire towns
positive card tests are referred for microscopy tests to identify parasites in blood
teams can be deployed to towns via motorcycle or small boat
tsetse nets
tiny target nets effectively capture the flies
net deployment leads to 80% decline in tsetse population, 3-fold decline in disease prevalence. in control area, disease prevalence increased by 10-fold
t brucei extracellular
exclusively extracellular parasite
in blood, organs, and lymphatic system during early stages, then crosses blood-brain barrier in second stage illness
parasite is constantly exposed to host immune system
designed to swim in blood, motility is optimized for crowded environments and high viscosity. cells actually swim faster in blood than in media
t brucei parasitemia
shows cycles of high and low
waves tended to correlate with increased symptoms: fever, exhaustion
t brucei bloodstream form
have thick cell surface
variant surface glycoprotein (VSG) comprises most of the bloodstream form cell surface
VSG
variant surface glycoprotein
more than 10^7 copies of protein per cell
99% of cell surface covered
GPI anchored, not integral membrane protein
varying oligomerization states
~1600 copies of VSG gene in trypanosome genome
forms tight array on cell surface that minimizes the exposure of antigens to the host immune system
new VSG structures suggest wider diversity than previously thought and may have additional function beyond immune evasion
antigenic variation
t brucei can escape immune response
cells expressing one VSG grow to high density, but immune system recognizes cells and produces antibodies, almost all parasites cleared by antibody
low number of parasites “switch” to a different VSG gene, antibodies no longer function allowing them to survive
expresses only a single VSG variant at a time and maximizes the repertoire of VSGs in genome
switching is rare, successful immune response clears out all parasites with current VSG
antigenic variation mechanism
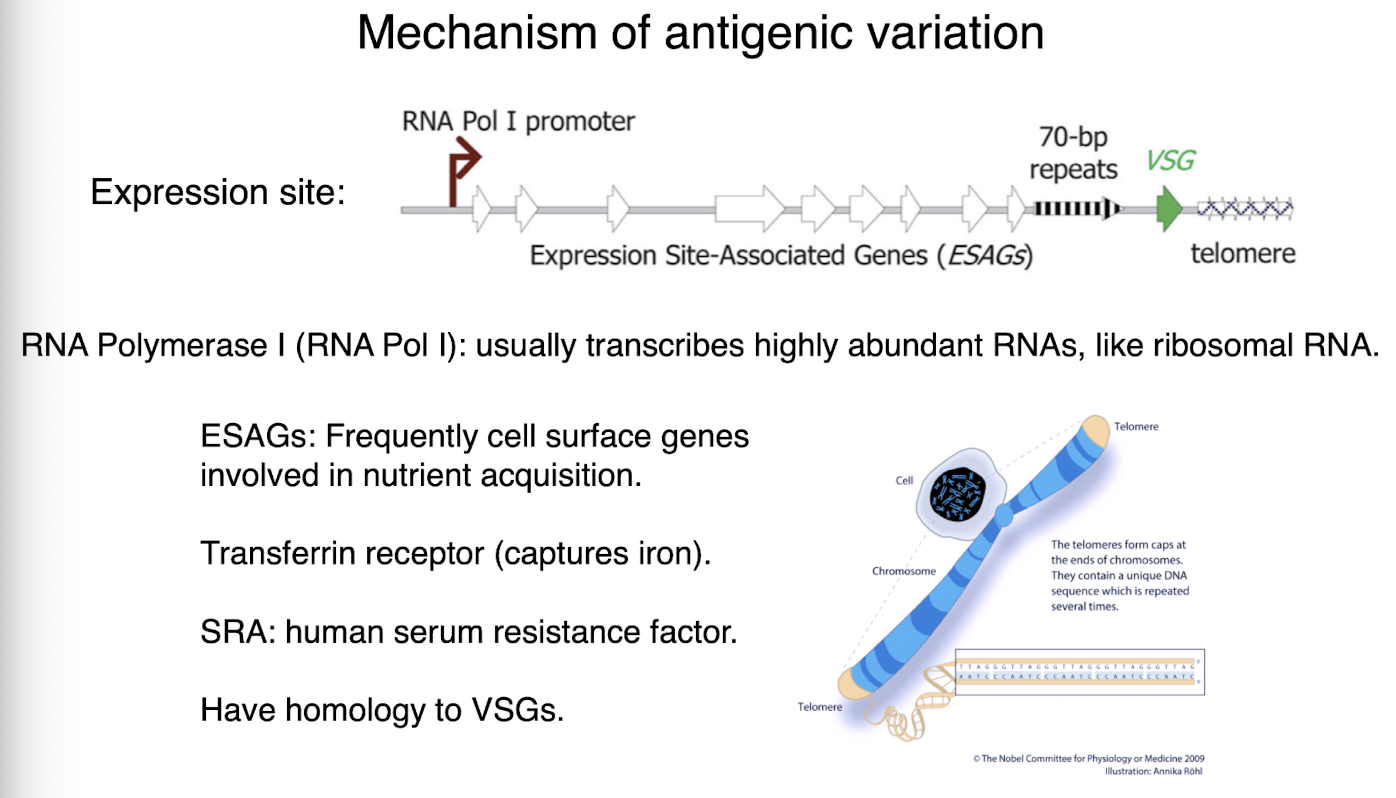
vsg in nucleus
other telomeric VSGs held inactive in nuclear periphery
active expression site body labeled with Pol I antibody
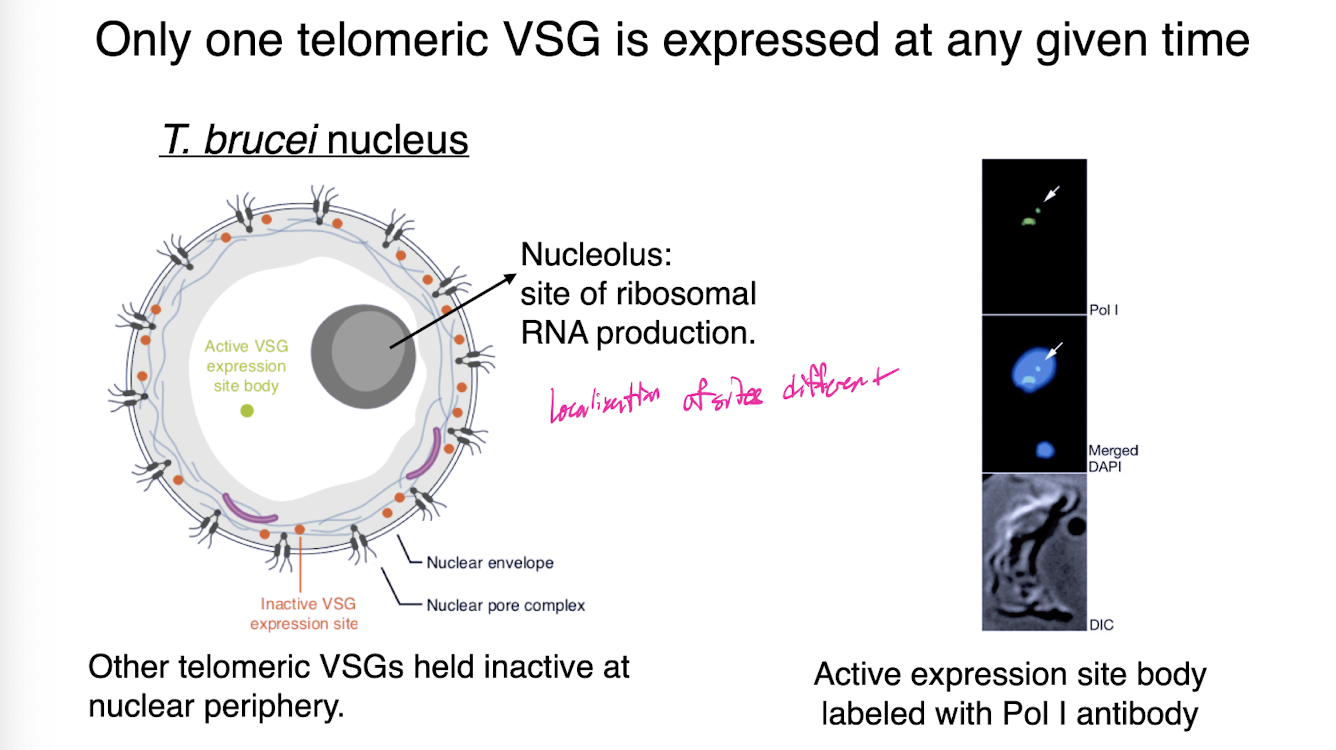
VSG switching can occur by switching to new VSG expression site
15 known sites in T brucei
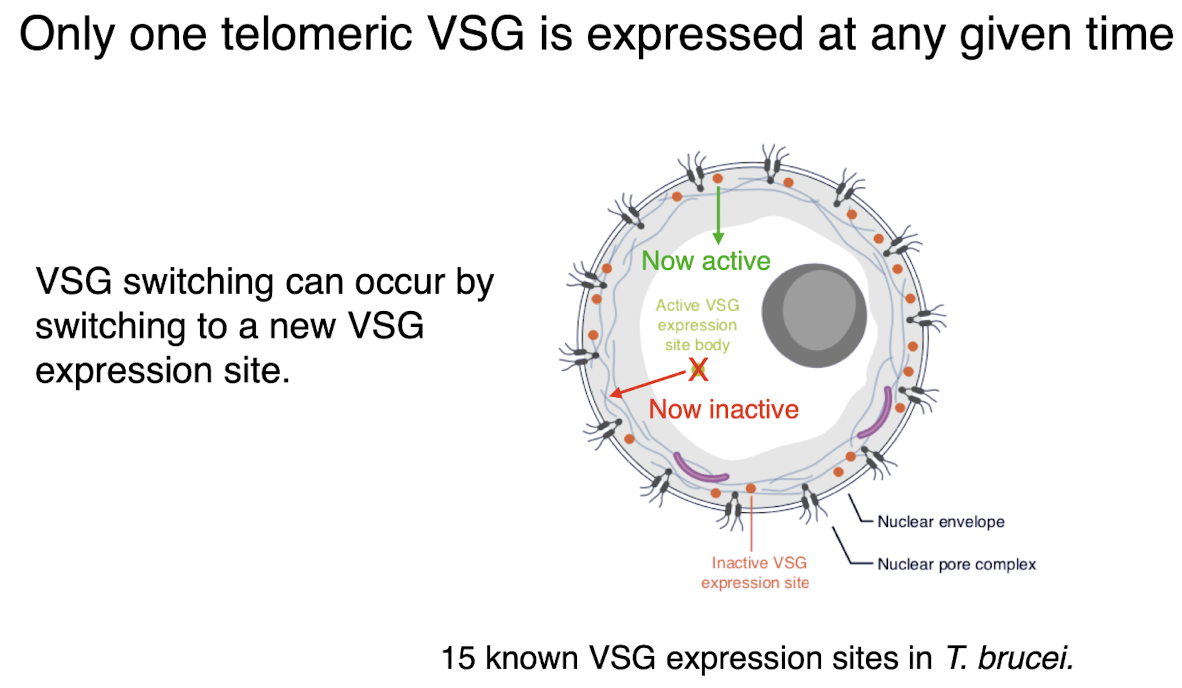
VSG switching in genome
can occur via homologous recombination at VSG
DNA break at VSG allows replacement of VSG gene
either VSG at other inactive site can replace VSG or other VSGs
t brucei has 11 primary chromosomes, inactive VSGs are maintained at “subtelomeric” (near telomeres) sites
minichromosomes contain hundreds of VSGs and function as repositories for “other” VSGs
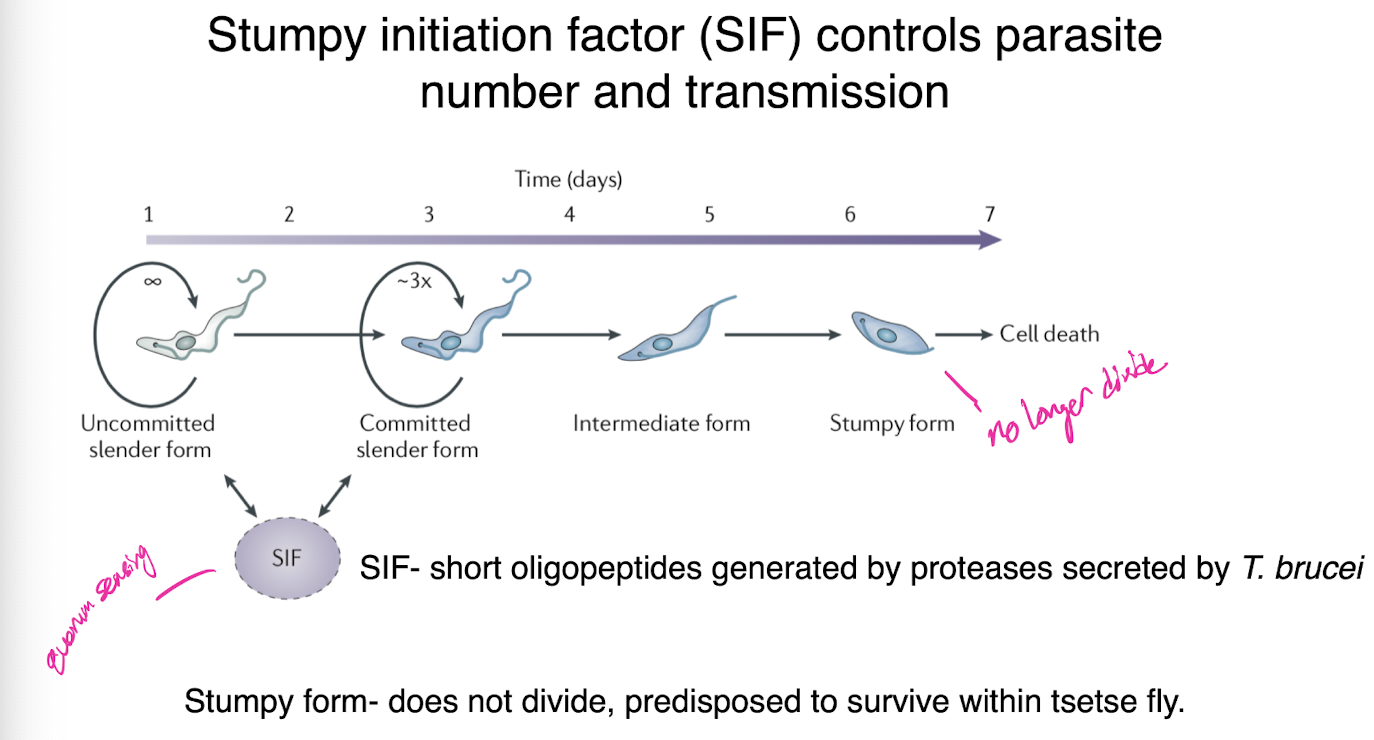
stumpy initiation factor
controls parasite number and transmission in T brucei
SIF-short oligopeptides generated by proteases secreted by t brucei
stumpy form - does not divide, predisposed to survive within tsetse fly
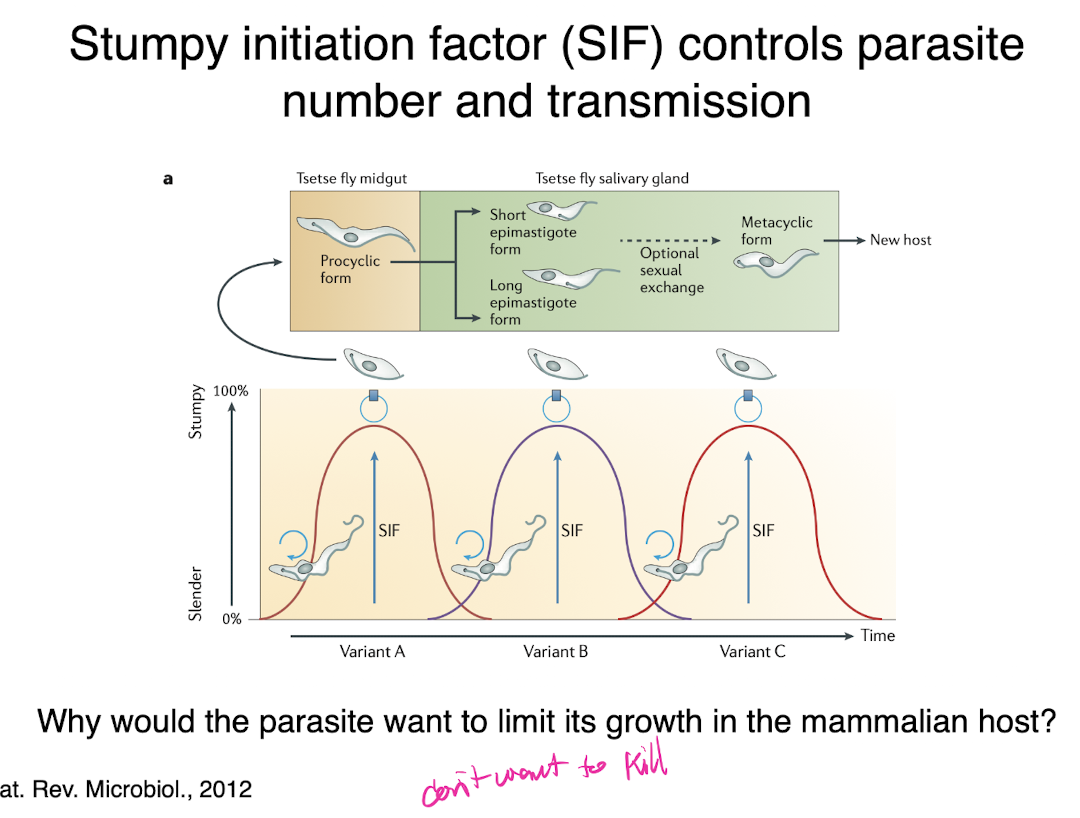
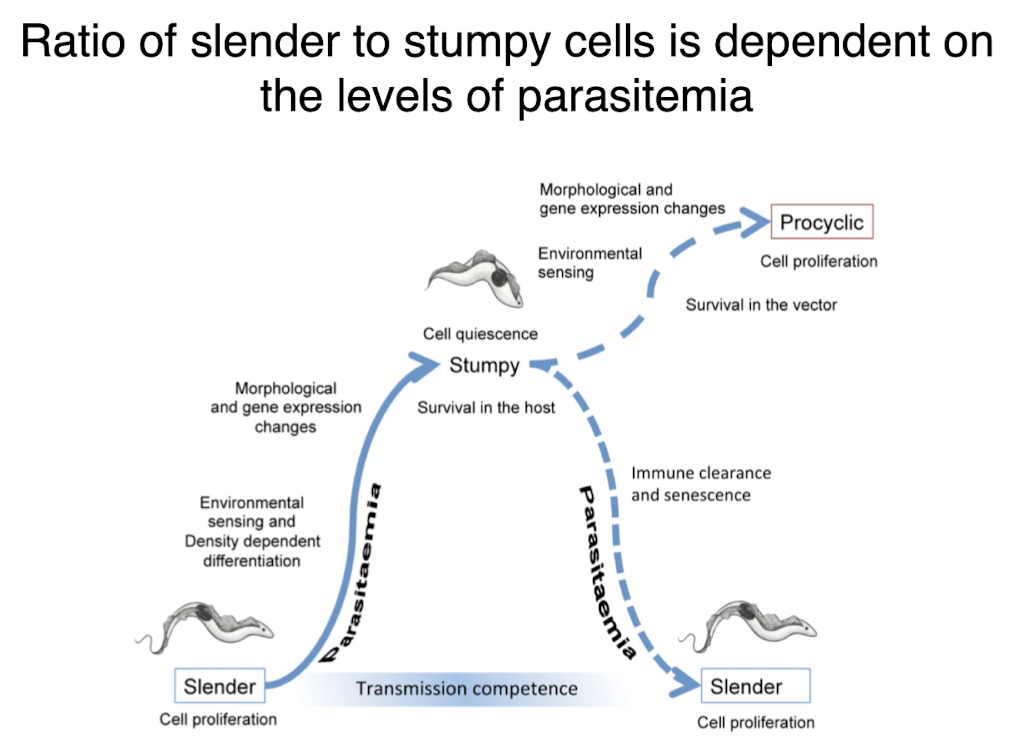
ratio of slender to stumpy t brucei
dependent on levels of parasitemia
t brucei subspecies hosts
congolense, brucei bruxei, vivas cannot cause infections in humans
brucei gambiense and brucei rhodiense can infect humans.
t brucei HDL
component of these particles in blood lyses certain trypanosome sub-species
particles filled with lipids and cholesterol
same VDL/HDLs that are tested for cholesterol levels
APOL-1 component of lipid particles
ApoL-1
effectively kills certain trypanosome sub-species
lysis appears to occur by swelling of the endosome/lysosome
similar appearance and kinetics compared to lysis with human serum
ApoL1-mediated lysis mechanism
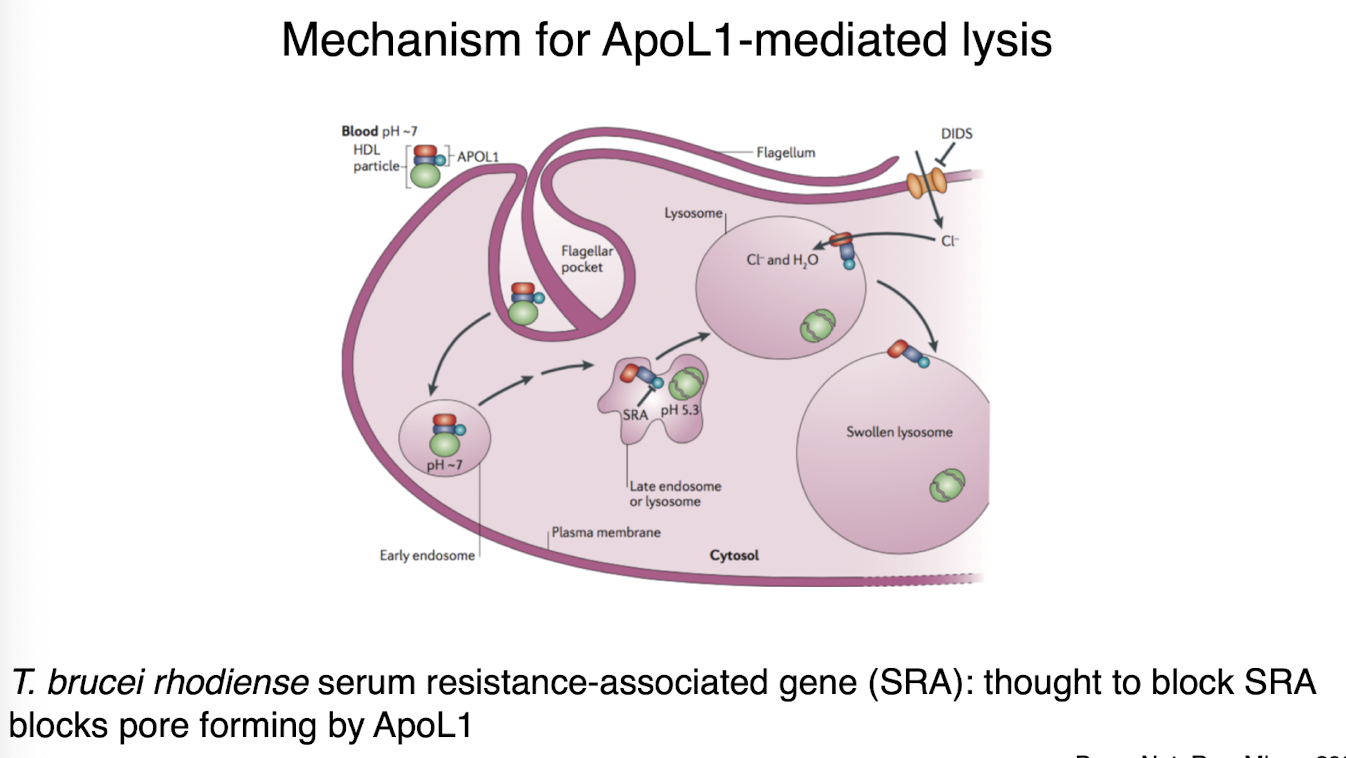
blocks ApoL1-mediated lysis in gHAT
TgsGP: T. b. gambiense-specific glycoprotein
stiffens membranes, thought to limit ApoL1 insertion into membrane
t brucei transmitted by bites
remain in nearby tissue
parasite associates with many cell types associated with skin
cells appear to be dividing and viable
abundant in skin after tsetse bite
alive and outside the vasculature
present in skin 12 days after infection in injection
t brucei eradication complications
skin resevoirs
people carrying skin infection had no symptoms of HAT
skin infection could have minimal/different symptoms
t brucei important resevoirs
fat and skin
fat resident ones stay primarily slender
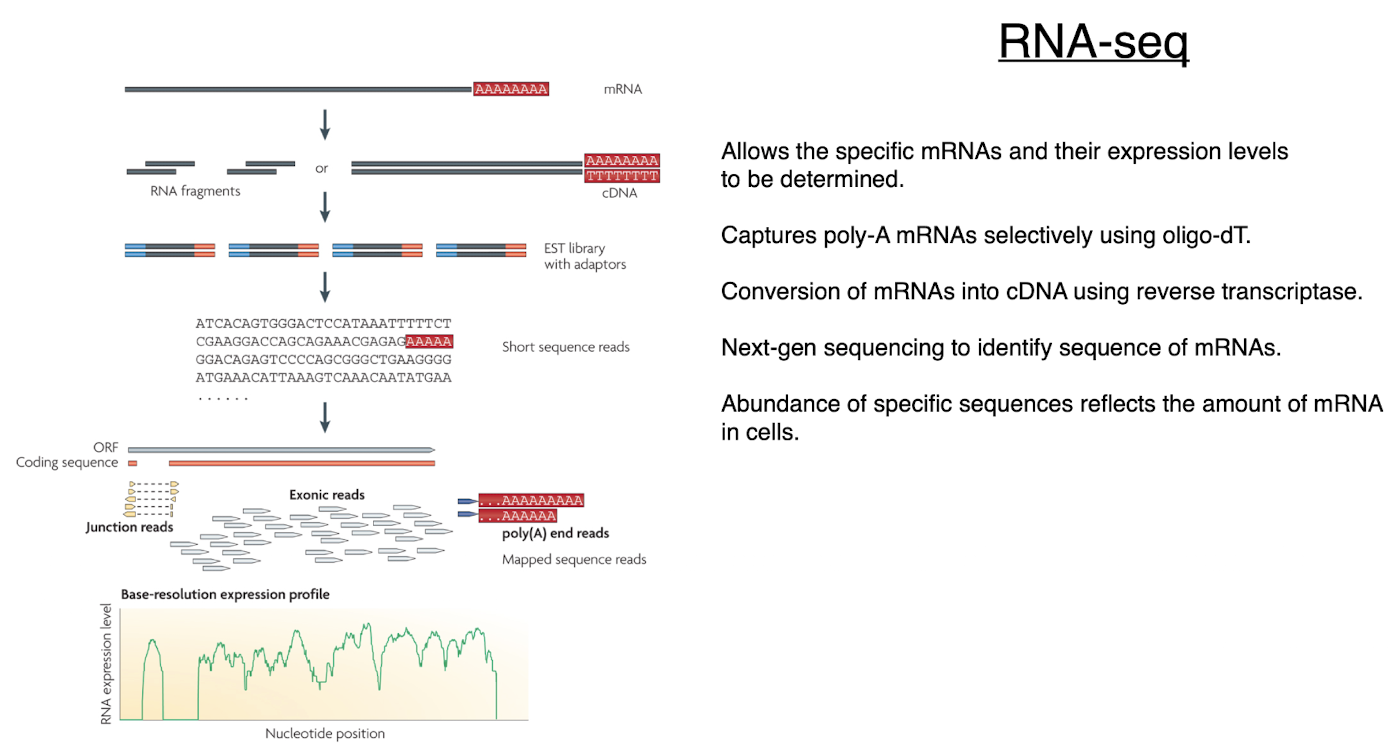
RNA-seq
allows specific mRNAs and their expression levels to be determined
captures poly-A mRNAs selectively using oligo-dT
conversion of mRNAs into cDNA using reverse transcriptase
next-gen sequencing to identify sequence of mRNAs
abundance of specific sequences reflects the amount of mRNA in cells
t brucei motility
essential for evading host acquired immune response
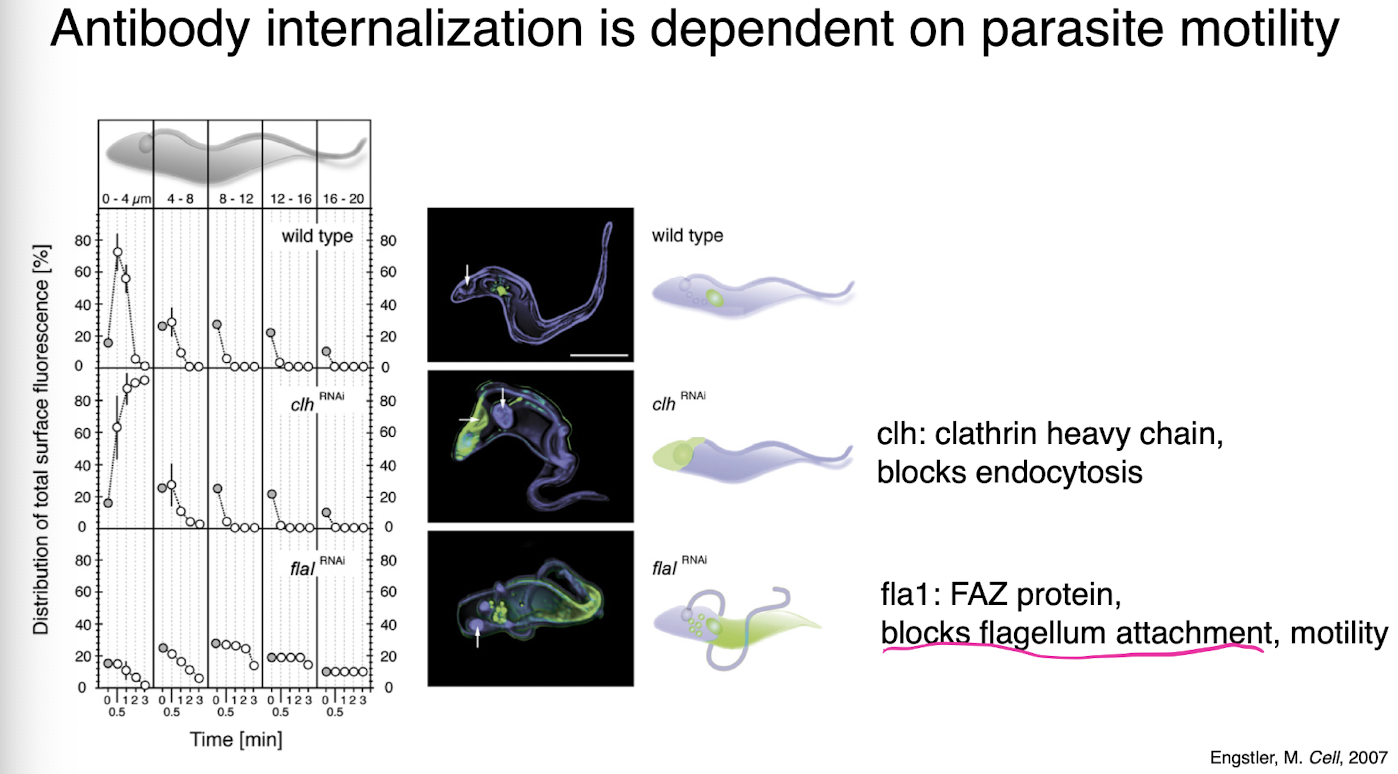
antibody internalization is dependent on parasite motility
t brucei antibody isotypes
display different internalization rates
hydrodynamic flow forces can drag antibody-VSG complexes towards cell posteior
larger antibody, more drag experiences so explains different rates of internalization for IgG, IgM, and Fab fragments
suggests that parasite motility is an important part of antibody clearance from cell surfaces
trypanosome swimming behavior
function of viscosity
higher viscosity gives higher motility
they can navigate crowded environments well
testing crowded environments
PDMS pillar arrays
biocompatible plastic
speed is affected by pillar spacing
tuned to size of erythrocytest
trypanosome cellular waveforms
affect their swimming speed in pillar arrays
zebra fish as host
t carassi can infect
attach to surfaces and erythrocytes
have all three modes of motility
sandfly
causes skin lesions
start small, but frequently ulcerate and develops “crateriform” borders
can take years to heal, leaving disfiguring scars
can also cause the formation of diffuse lesions and scarring
can cause erosion of mucous membranes
kala-azar
swelling of liver and spleen, weight loss, anemia, but no cyclical fevers or malaria present in blood smear
high degree of mortality noted
patients can resolve severe dermatitis after resolution
caused by leishmania
sir William boog leishman
identified parasites in spleens of malaria-like patients
identified small, ovoid cells in sample
noted “micronucleus” similar to what had previously been seen in t brucei
thought it was t brucei that had died and shriveled up
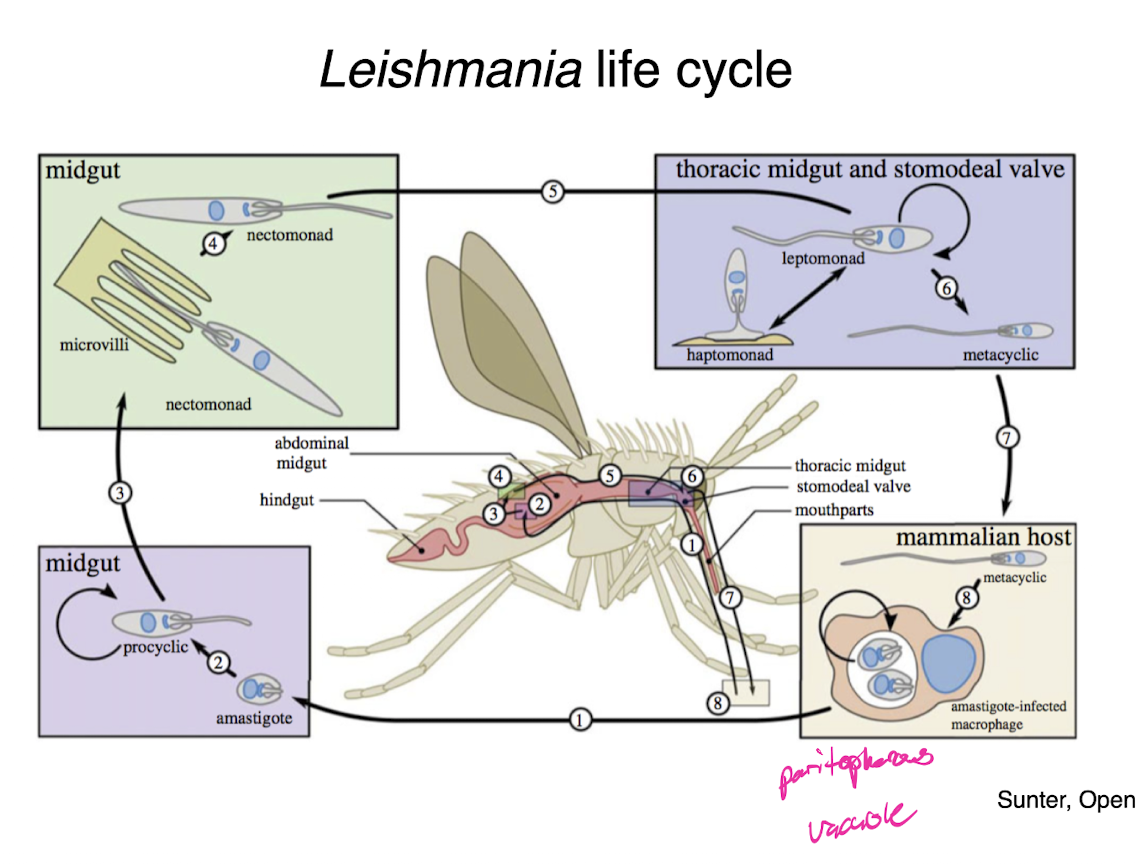
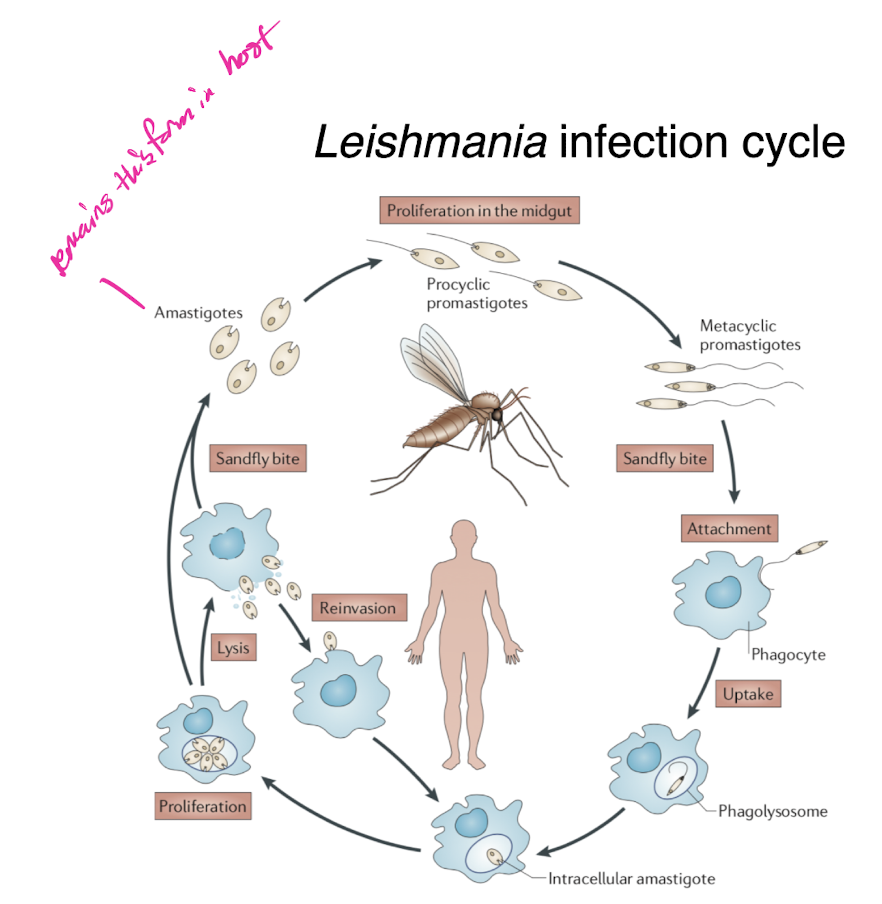
leishmania life cycle
ancient leishmania
conservation of life cycle stages and presence in sandfly argues that digenetic lifestyle is an ancient adaptation
presence in Asia and South America raises questions about dispersal of leishmania
Gondwana theory might give insight, ancestral leishmania developed there and two populations isolated during dissolution of supercontinent. developed separately afterwards
leishmania treatments
many different species, each of which has different sensitivities to drugs
some parasite may cause different diseases and we don’t know why
resistance is arising
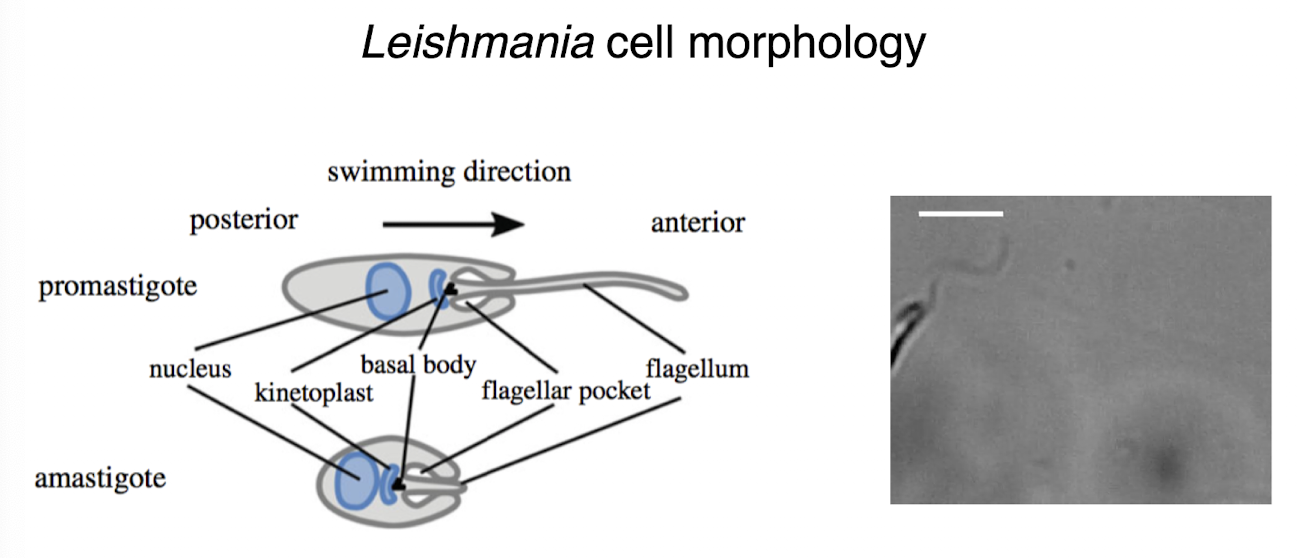
leishmania cell morphology
leishmania infection cycle
sandfly neutrophils
rapidly recruited to sandfly bite
neutrophils efficiently capture leishmania at sandfly bite site
internalized leishmania survive neutrophil lysis and can be recaptured
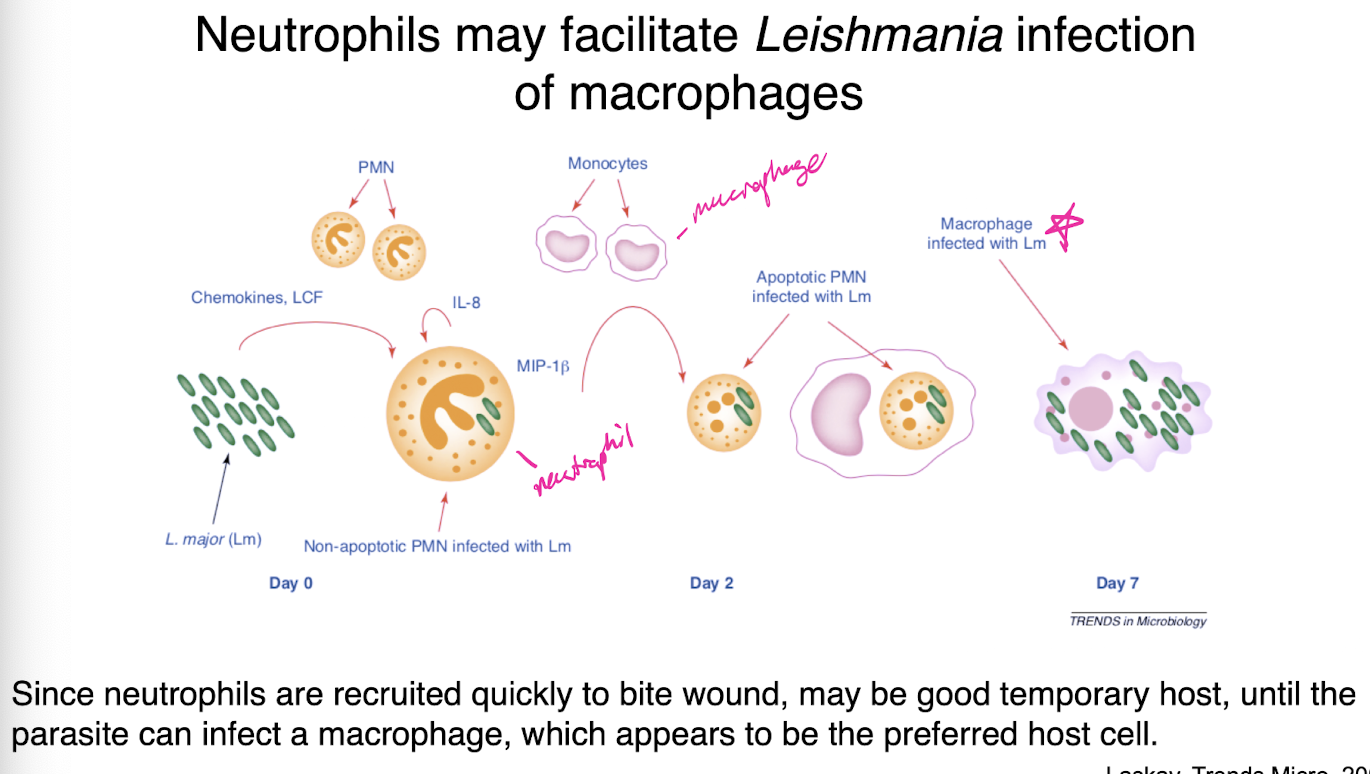
leishmania and neutrophils
depletion of neutrophils decreases number of leishmania in bitten ears
may serve as initial host cells. can be phagocytose by other immune cells, such as macrophages
neutrophils produce
NETs upon exposure to various pathogens
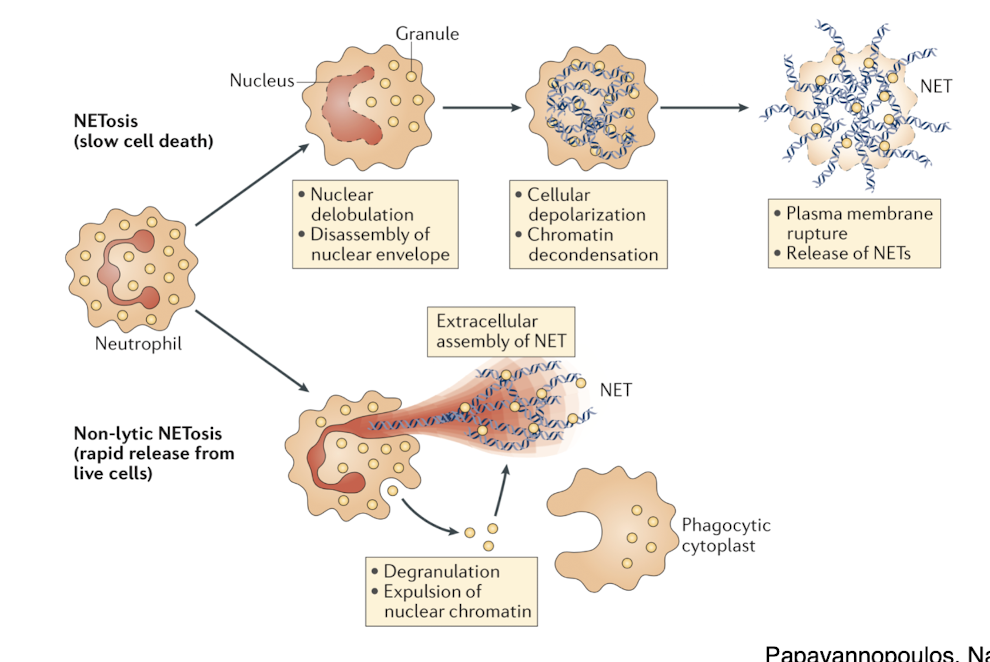
leishmania and NETs
leishmania LPG (lipophosphoglycan) blocks cell death caused by NETs
NET production is independent of LPG
leishmania in neutrophil
leishmania inside “loose” phagosome are killed
ones in “tight” phagosome tend to survive
recruitment of ER components to vacuolar membrane may block acidification and limit presence of lysosomal components
infected neutrophils are phagocytose by macrophages
neutrophils may facilitate leishmania infection of macrophages which are preferred host
leishmania and opsonization
opsonization facilitates uptake into immune cells