5.4 nutrient cycles
1/19
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
20 Terms
What is the role of saprobionts in recycling chemical elements
Decompose/ break down organic compounds (protein, urea, DNA, organic waste)
How do saprobionts break down organic compounds
by secreting enzymes for extracellular digestions (saprobiotic nutrition)
Absorb soluble nutrients that are needed and release mineral ions (e.g phosphate ions)
What is mycorrhizae
A Symbiotic association between fungi and plant roots
What’s the role of mycorrhizae
fungi (hyphae) act as an extension of plant roots to increase surface area of root system
To increase the rate of uptake/ absorption of water and inorganic ions
In return, fungi receives organic compounds (e.g carbohydrates)
Give examples of biological molecules that contain nitrogen
amino acids
Proteins or enzymes
Urea
DNA or RNA
Chlorophyll
ATP or ADP
NAD or NADP
What are the key stages of the nitrogen cycle
Nitrogen fixation
Ammonification
Nitrification
Dentrification
Draw the stages of the nitrogen cycle
.
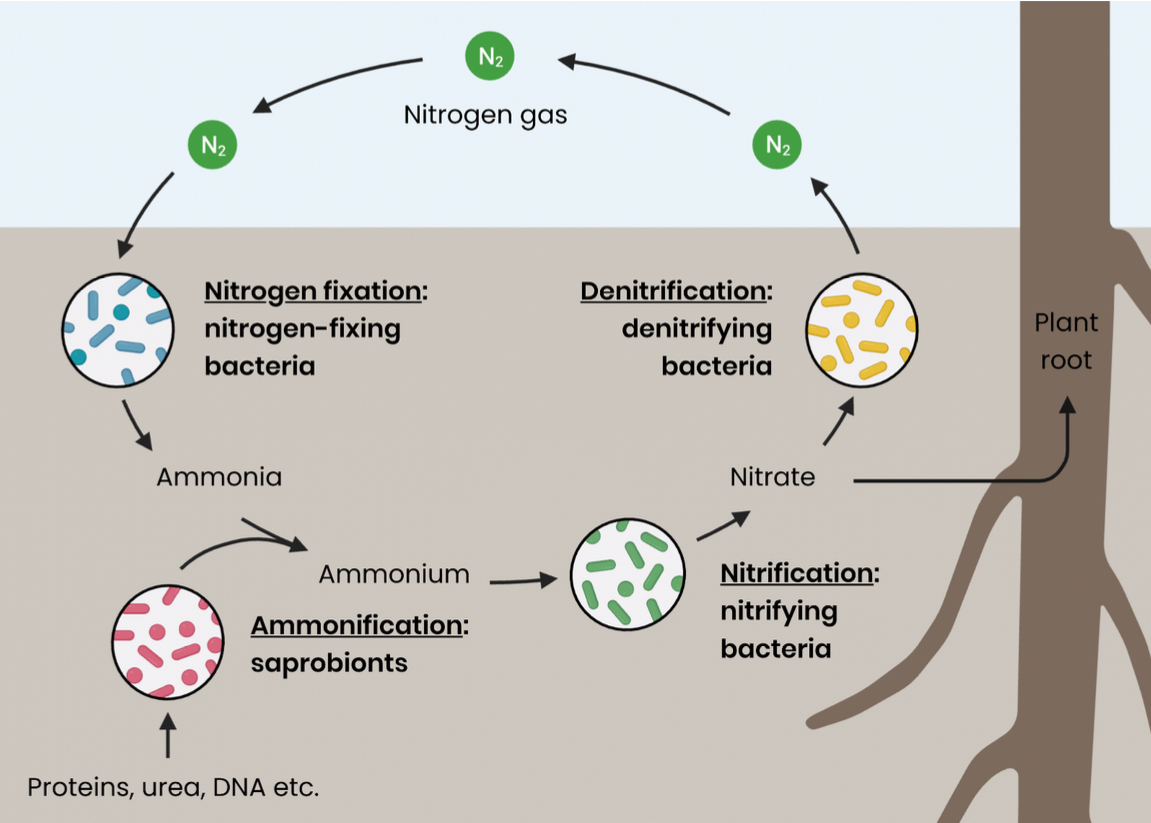
Describe the role of bacteria in nitrogen fixation
nitrogen gas (N2) is converted into ammonia (NH3) which forms ammonium ions (NH4+) in soil
This is done by nitrogen fixing bacteria, this may be found in root nodules
Describe the role of bacteria in ammonification
nitrogen-containing compounds (e,g proteins/ urea/ waste) are broken down/ decomposed
Converted to ammonia which forms ammonium ions in soil
This is done by saprobionts, which secrete enzymes for extracellular digestion
Describe the role of bacteria in Nitrification
ammonium ions in soil are converted into nitrites then nitrates via a two step oxidation reaction
Allows for uptake by plant roots hair cells by active transport
Done by nitrifying bacteria in aerobic conditions (oxygen)
Describe the role of bacteria in dentrification
nitrates in soil converted into nitrogen gas (reduction)
Done by denitrifying bacteria in anaerobic conditions (no oxygen, e.g waterlogged soil)
Why does ploughing/ aerating soil increase its fertility
more ammonium converted into nitrite and nitrate, more Nitrification, more active nitrifying bacteria
Less nitrate is converted to nitrogen gas, less denitrification, fewer active denitrifying bacteria
Give examples of biological molecules that contain phosphorus
Phospholipids
DNA or RNA
ATP or ADP
NADP
TP or GP
RuBP
Describe the phosphorus cycle
Phosphate ions in rocks are released by erosion/ weathering
These ions are taken up by producers/ plants/ algae and incorporated into their biomass (rate of absorption is increased by mycorrhizae)
Phosphate ions are transferred through food chain
Some are lost in water products/ excretion
Saprobionts decompose organic compounds releasing phosphate ions
Why are fertilisers used
to replace nitrates/ phosphates lost when plants are harvested and livestock are removed
As those removed from soil on incorporated into biomass can’t be released back into the soil through decomposition of saprobionts
So it improves efficiency of energy transfer, increasing productivity/ yield
What is a natural fertiliser
organic, e.g manure, compost, sewage
Ions released during decomposition of saprobionts
What are artificial fertilisers
contain inorganic compounds of nitrogen, phosphorus and potassium
What are the environmental issues from using fertilisers (1)
phosphates/ nitrates dissolve in water, leading to leaching of nutrients into lakes, rivers and oceans
This leads to eutrophication
What is eutrophication (2)
rapid growth of algae in ponds/ rivers (called algal bloom), so light is blocked
So submerged plants die as they cannot photosynthesise
So saprobionts decompose dead plant matter, using oxygen in aerobic respiration
So there’s less oxygen for fish to aerobically respire, so they die
What are the advantages of natural fertilisers
less soluble in water, so less leaching. Making eutrophication less likely
Organic molecules require breaking down by saprobionts, so there’s a slower release of nitrates/ phosphates