DRAD
1/153
Earn XP
Description and Tags
Not completed
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
154 Terms
Define ‘Radiography’
The techniques involved in producing various radiographic images
Define ‘Radiology’
The interpretation of various radiographic images
Define ‘Attenuation’
Reduction in the intensity of the main X-ray beam caused by absorption and scattering
Define ‘Scattering’
Change in direction of a photon with or without a loss of energy
Define ‘Absorption’
Deposition of energy, i.e. removal of energy from the beam
Define ‘Ionisation’
Removal of an electron from a neutral atom producing a negative ion (the electron) and a positive ion (the remaining atom)
Define ‘Radiolucent’
Less dense materials that permit X-ray beams to pass through, hitting the receptor
Define ‘Radiopaque’
Dense materials that absorb or stop X-ray penetration
What affects the shadow density of an object?
specific type of material of which the object is made
thickness or density of the material
shape
intensity of xray beam
position of object in relation to the xray beam and image receptor
sensitivity and type of image receptor
What is superimposition?
The overlapping of anatomical structures on a radiograph due to the projection of three-dimensional structures onto a two-dimensional image
Key limitations of a 2D radiographic image
superimposition
loss of depth information - cannot tell true spatial relationships
distortion - can be stretched or compressed
magnification - objects can appear larger
geometric inaccuracies - shapes and lengths may not be true to reality
limited detail of complex anatomy - 3D structures are flattened
What factors affect overall image quality of a 2D radiograph?
contrast
image geometry
characteristics of the xray beam
image sharpness and resolution
Define ‘Contrast’
The visual difference between the various black, white, and grey shadows
Define ‘Image geometry’
The relative positions of the image receptor, object, and xray tubehead
Describe the positioning of the Image Receptor, Object, and X-ray Beam
the object and the image receptor should be in contact or as close together as possible
the object and the image receptor should be parallel to one another
the X-ray tubehead should be positioned so that the beam meets both the object and the image receptor at right angles
Ideal characteristics for an X-ray beam
sufficiently penetrating, to pass through the patient and react with the film emulsion or digital sensor and produce good contrast between the different shadows
parallel, i.e. non-diverging, to prevent magnification of the image
produced from a point source, to reduce blurring of the edges of the image (penumbra effect)
Two main groups of traditional dental radiographs
intraoral
extraoral
How are X-rays described in terms of energy?
X-rays consist of wave packets of energy called photons, with each photon representing one quantum of energy
What is a photon?
A single packet (quantum) of X-ray energy
Define ‘Atomic number’
The number of protons in the nucleus of an atom
Define ‘Neutron number’
The number of neutrons in the nucleus of an atom
Define ‘Atomic mass number’
The sum of the protons and neutrons in an atom
Define ‘Isotope’
An atom with the same atomic number but different neutron number so therefore a different atomic mass
Define ‘Radioisotope’
An isotope with unstable nuclei which undergo radioactive disintegration
What determines the chemical behaviour of an atom?
The number of electrons determines the chemical behaviour of the element and forms the basis of the periodic table
Why are atoms in the ground state electrically neutral?
Number of positive charges (protons) is balanced by the number of negative charges (electrons), resulting in no overall charge
What is ‘excitation’ in an atom?
Excitation occurs when an electron moves from an inner shell to a higher energy outer shell. The atom remains neutral but is in an excited state
Main features of an X-ray Tube
cathode (negative)
anode (positive)
tungsten target
focusing device
glass vacuum tube
high voltage supply
protective casing (lead-lined)
x-ray window
Purpose of the Cathode in an X-ray tube
Consists of a heated filament of tungsten that provides the source of electrons
Purpose of the Anode in an X-ray tube
Consists of a tungsten target for electrons set into the angled face of a large copper block to allow efficient removal of heat
Purpose of the Focusing Device in an X-ray Tube
Aims the stream of electrons at the focal spot on the target
Why is the target in an X-ray tube made of tungsten?
high atomic number - high number of electrons, efficient production of x-ray
high melting point - withstand heating
Purpose of the Glass Vacuum Tube in an X-ray Tube
Maintains a vacuum so that electrons can travel freely and prevents collisions with air molecules
Purpose of the High Voltage Supply in an X-ray Tube
Creates a potential difference between the cathode and anode so the electrons can be accelerated at a high speed towards the target
Purpose of the Protective Housing of an X-ray Tube
Lined with lead which absorbs unwanted x-rays because x-rays are emitted in all directions
Contains insulating oil which facilitates the removal of heat
Describe how x-rays are produced in an x-ray tube
The filament is electrically heated and a cloud of electrons is produced around the filament.
The high-voltage (potential difference) across the tube accelerates the electrons at very high speed towards the anode.
The focusing device aims the electron stream at the focal spot on the target.
The electrons bombard the target and are brought suddenly to rest.
The energy lost by the electrons is transferred into either heat (about 99%) or X-rays (about 1%).
The heat produced is removed and dissipated by the copper block and the surrounding oil.
The X-rays are emitted in all directions from the target. Those emitted through the small window in the lead casing constitute the beam used for diagnostic purposes
What happens when high-speed electrons strike the tungsten target in an X-ray tube?
Two main types of collisions:
heat-producing collisions
x-ray-producing collisions
Describe a Heat-producing Collision
The incoming electron is deflected by the cloud of outer-shell tungsten electrons, with a small loss of energy, in the form of heat
The incoming electron collides with an outer-shell tungsten electron, displacing it to an even more peripheral shell (excitation) or displacing it from the atom (ionisation), again with a small loss of energy in the form of heat
Why are heat-producing interactions more common than x-ray-producing interactions?
There are millions of incoming electrons and many outer-shell tungsten electrons with which to interact
Each individual bombarding electron can undergo many heat-producing collisions resulting in a considerable amount of heat at the target
Why must heat be removed from the X-ray tube target, and how is this achieved?
Heat must be removed quickly to prevent damage to the tungsten target
Achieved by embedding the tungsten target in a copper block, which has high thermal capacity and good thermal conductivity
Describe an X-ray-producing Collision
The incoming electron penetrates the outer electron shells and passes close to the nucleus of the tungsten atom - it is dramatically slowed down and deflected by the nucleus with a large loss of energy, which is emitted in the form of x-rays
the incoming electron collides with an inner-shell tungsten electron, displacing it to an outer shell (excitation) or displacing it from the atom (ionisation),with a large loss of energy and subsequent emission of x-rays
What are the two types of X-ray spectra produced by X-ray-producing collisions?
Continuous spectrum (Bremsstrahlung radiation)
Characteristic spectrum
Describe the Continuous Spectrum
The continuous spectrum is a range of X-ray energies produced when high-speed electrons are slowed down (decelerated) near the nucleus of tungsten atoms - the amount of deceleration and degree of deflection determine the amount of energy lost by the bombarding electron/resultant emitted photon
Far from nucleus → small energy loss → low-energy X-rays
Very close → large energy loss → high-energy X-rays
Why is the Continuous Spectrum described as ‘continuous’?
electrons can lose any amount of energy
a wide range or spectrum of photon energies is therefore possible
What type of electron deflection is most common in continuous X-ray production and what does it produce?
Small deflections are most common and produce low-energy photons
Why do low-energy photons not contribute to the useful X-ray beam?
They have low penetrating power and are absorbed within the X-ray tube or patient, so they do not reach the detector - their removal is called filtration
What type of deflection produces high-energy photons and how common is it?
Large deflections (close interaction with the nucleus) produce high-energy photons, but these events are rare
What determines the maximum photon energy (Emax) in an X-ray tube?
The potential difference (kV) across the X-ray tube
Describe the Characteristic Spectrum
The characteristic spectrum consists of X-rays with specific (discrete) energies that are unique to the target material (e.g. tungsten)
High-energy electron hits inner-shell electron
A bombarding electron ejects an inner-shell (e.g. K-shell) electron from a tungsten atom
Vacancy is created
This leaves a gap in the inner shell
Electron from outer shell drops down
An electron from a higher energy shell moves down to fill the vacancy
Energy is released
The difference in energy between the two shells is emitted as an X-ray photon
Why is the Characteristic Spectrum described as ‘characteristic’?
The energy of the emitted X-ray depends on:
The difference between electron shell energy levels
These energy differences are unique to each element
➡ So the X-rays are characteristic of tungsten
What do the K and L lines represent in a Line Spectrum?
They are named according to the electron shell involved in the transition:
K lines → transitions to the K shell
L lines → transitions to the L shell
Which characteristic X-ray lines are diagnostically important and why?
Only the K lines are diagnostically important because L lines have too little energy to be useful
What is required to produce a characteristic K-line in tungsten?
The bombarding electron must have ≥ 69.5 kV to eject a K-shell electron
What is meant by the “critical voltage” in X-ray production?
It is the minimum voltage (69.5 kV for tungsten) required to produce characteristic K-line photons
What happens if the X-ray tube operates below the critical voltage?
No characteristic K-line photons are produced
What is the typical operating voltage range of dental X-ray machines?
Approximately 60–90 kV
What is meant by intensity and quality of an X-ray beam?
Intensity = number (quantity) of photons
Quality = energy of photons (penetrating power)
What factors affect X-ray beam intensity and quality?
Tube voltage (kV)
Tube current (mA)
Distance (d)
Time (t)
Filtration
Target material
Voltage waveform
How do X-rays travel in free space?
Travel in straight lines
Speed = 3 × 10⁸ m/s
Do not require a medium
What is the inverse square law?
Intensity is inversely proportional to the square of the distance:
👉 Doubling distance → intensity becomes ¼
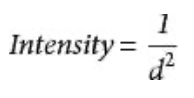
How does wavelength relate to X-ray energy?
Short wavelength → high energy → high penetration
Long wavelength (soft X-rays) → low energy → low penetration
What happens when X-rays interact with matter?
They are attenuated by:
Absorption
Scattering
Why are X-rays considered hazardous?
They are ionising radiation, capable of causing biological damage
Can humans detect X-rays?
No — X-rays are undetectable by human senses
How are X-rays used to produce images?
1. Affect film emulsion → radiographic image
When X-rays pass through the body, different tissues absorb them differently.
The remaining X-rays hit the film (or sensor).
On traditional film, X-rays change the film emulsion chemically, creating a visible image after processing.
Areas that get more X-rays appear darker, and areas that absorb more X-rays (like bone) appear lighter.
2. Cause fluorescence → used in screens and digital sensors
X-rays can make certain materials glow (fluoresce).
This glow is used in:
Intensifying screens (to expose film more efficiently)
Digital sensors (which convert X-rays into light, then into an electronic image)
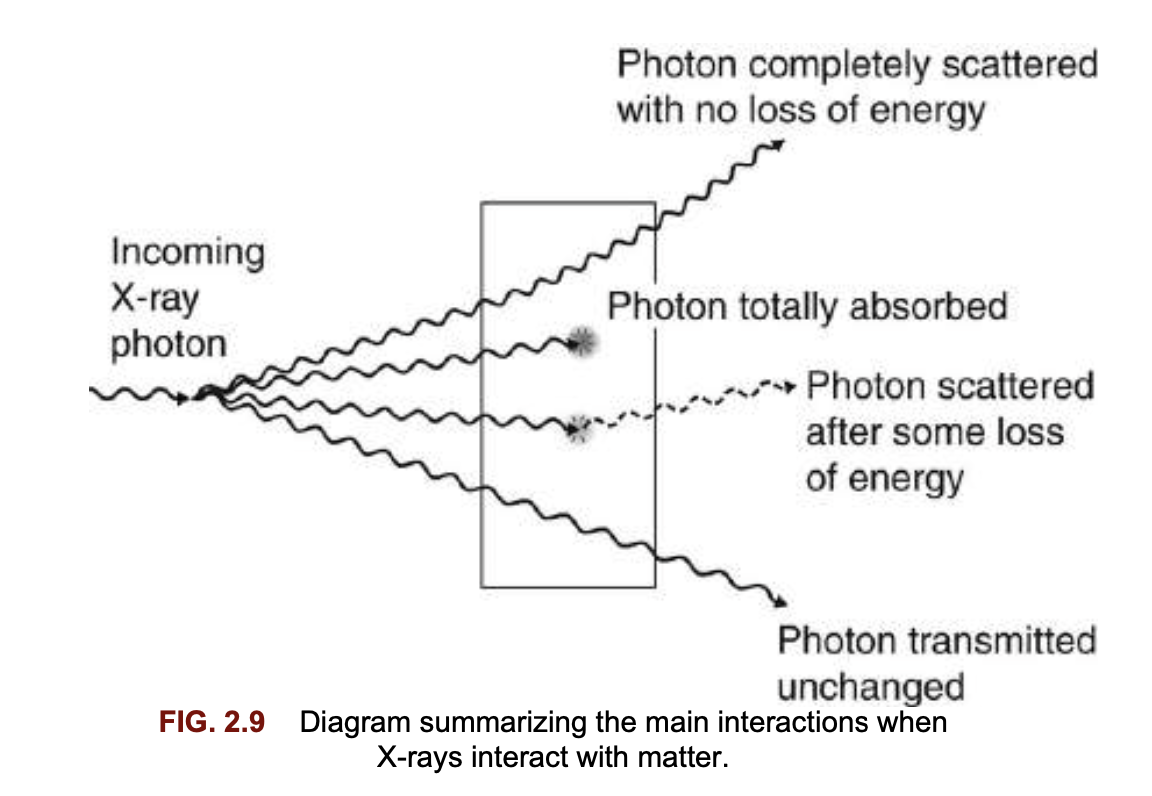
What are the four possible fates of X-ray photons when they strike matter?
Completely scattered with no loss of energy
Completely absorbed (total loss of energy)
Scattered with partial absorption (loss of energy)
Transmitted unchanged
What are the four main interactions of X-rays with matter at the atomic level?
Unmodified (Rayleigh) scattering – pure scatter
Photoelectric effect – pure absorption
Compton effect – scatter + partial absorption
Pair production – pure absorption
What is Rayleigh scattering?
A low-energy interaction where the photon is scattered without loss of energy (no ionisation)
What is the photoelectric effect?
The photon is completely absorbed, ejecting an inner-shell electron → ionisation
What is the Compton effect?
The photon is partially absorbed and scattered, losing some energy and ejecting an outer-shell electron.
What is pair production?
A high-energy interaction where the photon is absorbed and produces an electron–positron pair (not relevant in dental X-rays)
Which two X-ray interactions are important in the dental energy range?
Photoelectric effect
Compton effect
Why are only the photoelectric and Compton effects important in dentistry?
Because dental X-ray energies are such that:
Rayleigh scattering is minimal
Pair production requires much higher energy and does not occur
Summary of the Stages in the Photoelectric Effect
1. The incoming X-ray photon interacts with a bound inner-shell electron of the tissue atom.
2. The inner-shell electron is ejected with considerable energy (now called a photoelectron ) into the tissues and will undergo further interactions.
3. The X-ray photon disappears having deposited all its energy; the process is therefore one of pure absorption.
4. The vacancy that now exists in the inner electron shell is filled by outer-shell electrons dropping from one shell to another.
5. This cascade of electrons to new energy levels results in the formation of very low energy radiation (e.g. light), which is quickly absorbed.
6. Atomic stability is finally achieved by the capture of a free electron to return the atom to its neutral state.
7. The high-energy ejected photoelectron behaves like the original high-energy X-ray photon, undergoing many similar interactions and ejecting other electrons as it passes through the tissues. It is these ejected high-energy electrons that are responsible for the majority of the ionization interactions within tissue, and the possible resulting damage attributable to X-rays.
What energy condition is required for the photoelectric effect to occur?
The X-ray photon energy must be equal to or slightly greater than the binding energy of the inner-shell electron
How does atomic number (Z) affect the photoelectric effect?
The probability is proportional to Z³
➡ Higher Z = more photoelectric interactions
Why is lead effective in radiation protection?
Lead has a high atomic number (Z = 82), making it a strong absorber of X-rays via the photoelectric effect
Why do bone and soft tissue appear different on radiographs?
Bone (Z ≈ 12) → higher photoelectric absorption
Soft tissue (Z ≈ 7) → lower absorption
➡ Creates radiographic contrast
How does X-ray energy (kV) affect the photoelectric effect?
Probability is proportional to 1 / kV³
➡ Lower kV = more photoelectric absorption
What is the effect of low kV on radiographs?
✅ Higher contrast
❌ Increased patient dose
What is the overall result of the photoelectric effect?
Ionisation of tissues
How do intensifying screens use the photoelectric effect?
They absorb X-rays and emit light, which then exposes the film
Summary of the Stages in the Compton Effect
The incoming X-ray photon interacts with a free or loosely bound outer-shell electron of the tissue atom.
The outer-shell electron is ejected (now called the Compton recoil electron ) with loss of some of the energy of the incoming photon, i.e. there is some absorption. The ejected electron then undergoes further ionizing interactions within the tissues (as before).
The remainder of the incoming photon energy is deflected or scattered from its original path as a scattered photon.
The scattered photon may then:
– Undergo further Compton interactions within the tissues
– Undergo photoelectric interactions within the tissues – Escape from the tissues – it is these photons that form the scatter radiation of concern in the clinical environment.Atomic stability is again achieved by the capture of another free electron.
What is the energy relationship in the Compton effect?
The incoming X-ray photon has much greater energy than the binding energy of the outer-shell electron
Is the Compton effect dependent on atomic number (Z)?
No — it is independent of atomic number, as the photon interacts with free or loosely bound electrons
Why does the Compton effect provide poor diagnostic information?
Because it is not dependent on Z, there is little differentiation between tissues, resulting in low image contrast
At what energies does the Compton effect predominate?
At higher X-ray photon energies (high kV)
How does high kV affect radiographic image quality?
❌ Reduced contrast
❌ More scatter (Compton effect)
What happens to the energy of the photon in the Compton effect?
The scattered photon has less energy than the incoming photon, as some energy is transferred to the recoil electron
How does scatter direction relate to photon energy?
High-energy photons → forward scatter
Low-energy photons → back scatter
Why is forward scatter important in radiography?
It can reach the image receptor and degrade image quality
What is the overall result of the Compton effect?
Ionisation of tissues
3 main components of Dental X-ray Equipment
Tubehead
Positioning arms
Control panel and circuitry
Ideal Requirements of Dental X-ray Equipment
Safe and accurate
Capable of generating X-rays in the desired energy range and with adequate mechanisms for heat removal • Small
Easy to manoeuvre and position
Stable, balanced and steady once the tubehead has been positioned
Easily folded and stored
Simple to operate and capable of both film and digital imaging
Robust
What are the main components of a dental X-ray tubehead?
Glass X-ray tube (filament, copper block, target)
Step-up transformer
Step-down transformer
Lead shielding
Oil (for cooling)
Aluminium filtration
Collimator
Spacer cone / beam-indicating device
What is the function of the step-up transformer?
Increases mains voltage (~240 V) to high kV needed to accelerate electrons
What is the function of the step-down transformer?
Reduces voltage to provide low voltage, high current to heat the filament
Why is lead shielding used in the tubehead?
To minimise leakage radiation
What does the collimator do?
Shapes and restricts the beam:
Rectangular (image receptor size) OR
Round (max 6 cm diameter)
What is the function of the spacer cone (beam-indicating device)?
Directs the X-ray beam
Maintains correct focus-to-skin distance (FSD)