titration theory
1/3
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
4 Terms
equivalence point
precise moment when the amount of titrant added is stoichiometrically equal to amount of analyte, rxn is complete
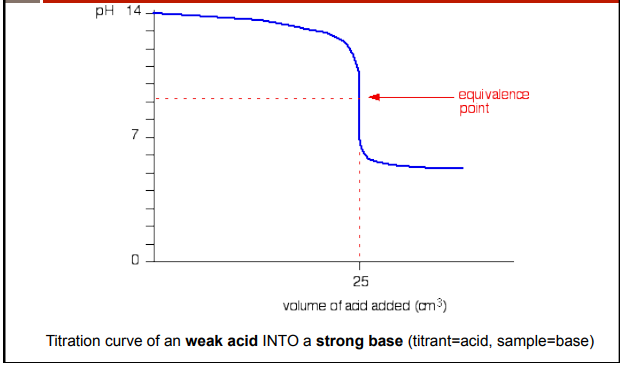
weak acid and strong base titrations
weak acid lets off some conjungate base (HA)
strong base fully dissociates into spectator cation and OH-
left over ions of weak acid act as a base
conjugate base + HA → H2O + A-
A- + H2O → HA + OH-
equivalence point at a pH greater than 7
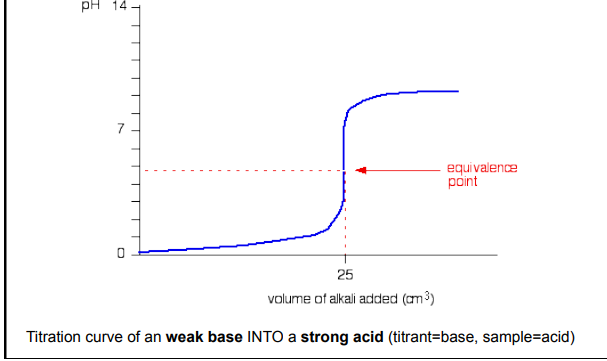
strong acid weak base titrations
strong acid dissociates fully into spectator ions and H+
weak base dissociates into conjugate acid
left over ions of the weak base act as an acid
conjugate acid + H+ → base
base + H2O → hydronium
equivalence point at a point less than 7
moles, volume concentration
na = nb
CaVa = CbVb