Transport proteins
1/15
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
16 Terms
Transporters
Things that carry materials across the membrane rather than information
Can use this therapeutically to prevent the transport across the membrane —> E.g Blocking SERT prolongs the action of the nt at the synapse
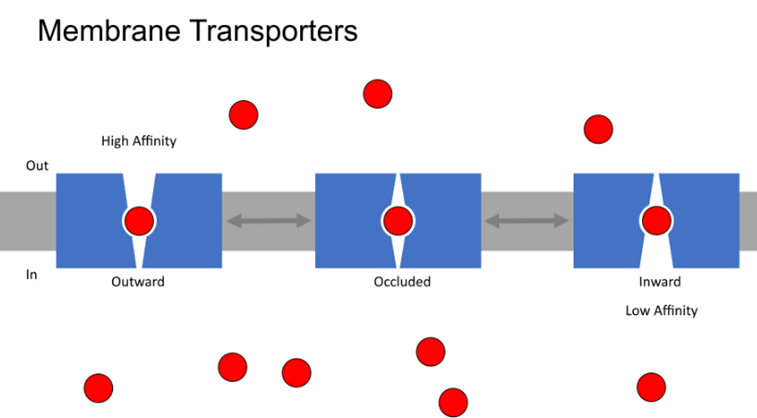
Have an outward facing binding site, with low concentrations of molecules which requires high affinity binding (outward facing)
Also need the protein to be open at no point otherwise will be a channel and dissipate the concentration gradient, so takes on this occluded conformation where the substrate is enveloped
In the inward facing state, we have to reduce the affinity of the transporter for the substrate so that its released
Required energy to drive this process —> either by ATP or by an electrochemical gradient
Types of transporters
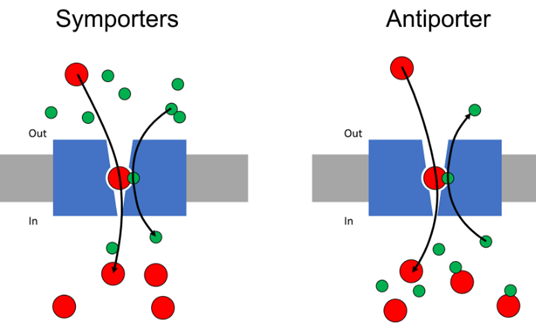
Symporters – the molecule being transported moves in the same direction as the molecule creating the gradient
Antiporters – the molecule being transported is moving in the opposite direction of the molecule that generates the gradient
Either one of these mechanism may be favoured depending on the charge of the molecule being transported —> E.g: if you have a negatively charged compound being moved, may favour symport with a proton to cancel out the charge difference
Solute carrier proteins
>50 structurally related subclasses, 300 individual proteins
Involved in numerous processes:
Nutrient uptakes
Elimination of drugs
Vesicle loading
Neurotransmitter release
Can be targets for:
Neurological conditions
Diuretics
Diabetic
Structure of transport proteins
Two main families of transporters: MFS and LeuT have similar structures and formed by the same mechanism
Duplication events give these highly symmetrical molecules – known as two inverted pseudo repeats
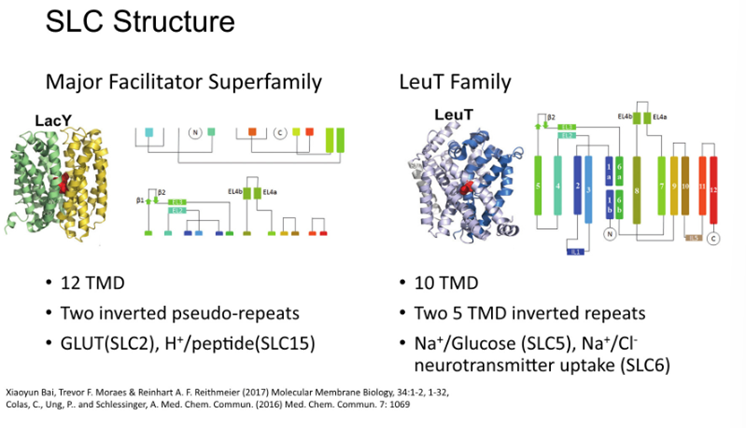
LeuT family – SLC (sodium chloride channel)
Involved in a whole range of processes
Types of transporters
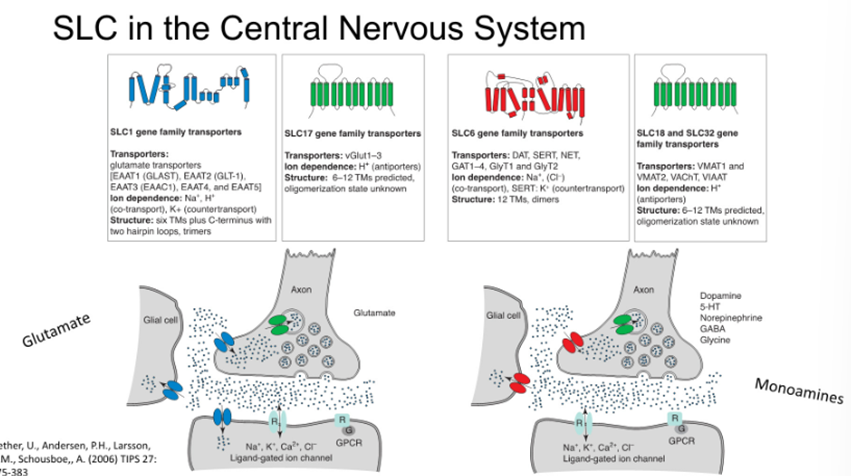
SLC1: Use the sodium and potassium electrochemical gradient to drive the transport of glutamate across the membrane
SLC17: glutamate antiporter using proton gradient to drive the transport of glutamate
SLC6: SERT transporter
SLC18/22: use proton gradients to transport molecules
Involved in interaction of the neurons with glial cells, loading of neurotransmitters etc
Critical to the function of the nervous system and therefore provides us with new opportunities to drug these targets
Vesicular transporters - members of the MFS family
Responsible for loading molecules into vesicles before their transport into the synapse
Uses a vesicular (v) ATPase to utilise ATP within the cell to generate a proton gradient
Depending on the substrate loaded into the vesicle depends on how the gradient is used
E.g: if cations like Ach is being used, the transport proteins are used to make use of the chemical (proton gradient) to assimilate Ach against their concentration gradient.
For zwitterions like GABA/Glycine, the transport proteins use a combination of the chemical gradient and the charge gradient
For anions like glutamate, the charge gradient across the membrane is used to assimilate substrate inside vesicles
Structure of vGLUT transporters
Mapping of helices and AA residues shows how the duplication event leads to the dimeric structure involved in the transport of glutamate
Environment that transporters are in will change significantly depending on whether its in a vesicle or fused with the membrane, also important that we are able to turn off these processes when its embedded in the membrane.
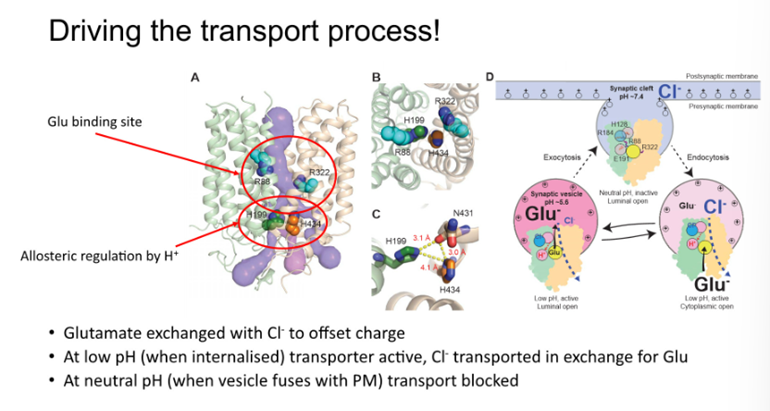
Driving the transport process
Activity is tightly regulated by the proton concentration
Antiport of the chloride ions with glutamate —> negative cl- transported in the opposite direction of a negatively charged glutamate so the charge remains the same
At low pH the transport process is relatively active —> when the H199 is protonated in the vesicle
When fused with the plasma membrane, the pH is at pH7 and the activity drops due to a change in ionisation state
The activity of the transport protein intimately coupled with where the protein is localised
SLC6 - Plasma membrane transporters
Utilised widely in the body and can be exploited for treatment of:
Epilepsy
Austism
Schizophrenia
CV disease
Retinal blindness
Renal function
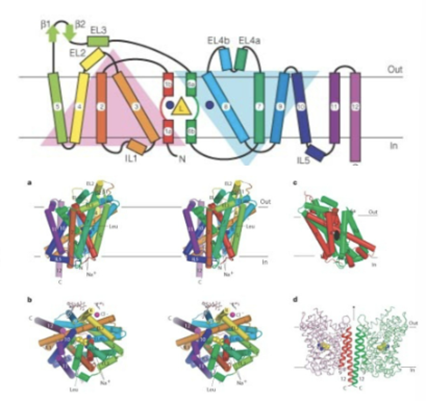
Duplication of two blocks of 5 —> 10TM domains of the transporter parts, plus 2 additional flanking TM domains
5 TM domains in each repeat are long blocks of alpha helices, and the fifth outer one has breaks which is important for the formation of ligand binding site therefore transport of proteins
Serotonin transporter
Conformationally flexible, which can make crystallography challenging
Identified mutations which proved the thermostability of the protein —> Attempted to use antibody fragments to stabilise a particular state
1st and 6th helix broken in the middle, with an internal binding ste and an allosteric binding site
Na+/Cl- down their concentration gradient is coupled very closely with the transport of serotonin; therefore, Na+/Cl- are part of the binding site and so unable to couple this transport
Substarte binding site
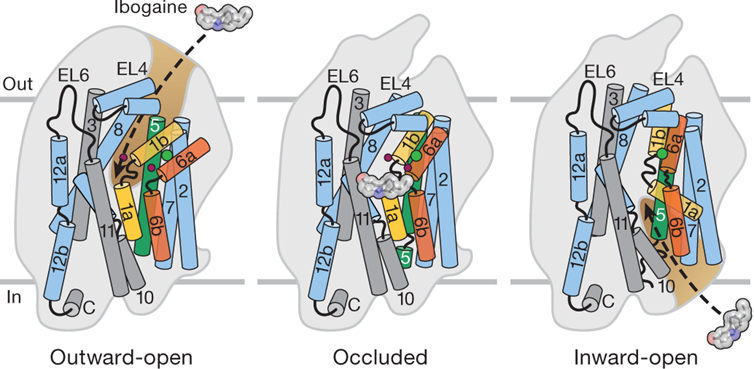
To study this, studies the binding of natural agonists (serotonin), artificial agonists (antidepressants) and antagonists (ibogaine)
Affinity in the nanomolar range showing high affinity binding —> useful to know because if we want to create a drug it needs to have a higher affinity
Common structure of two rings with a chain coming off, or fluorinated ring structure
Some conserved architecture but not so clear how these molecules will bind
Development of compounds targets 3 specific subsites, which when bound, give high affinity binding
A: central ring structure
B: aromatic ring structure with a halide ion
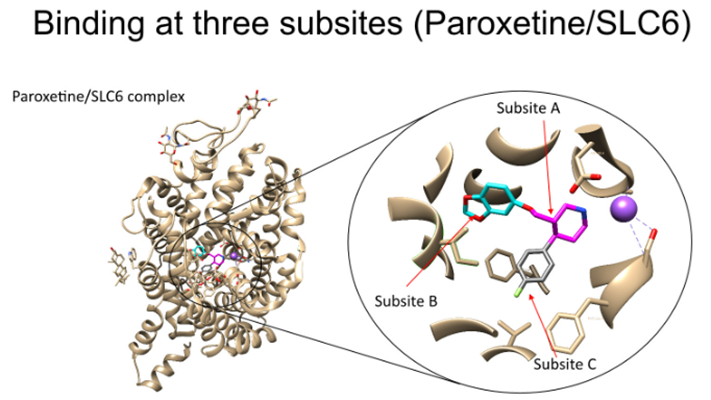
Interactions that stabilise binding
Halide ions (electronegative atoms) which interact with hydrogen bond donors (Ser/Thr)
Aromatic groups interaction of Phe and Try forming base stacks which helps to improve the affinity
Understanding the catalytic transport cycle
Understanding the conformational change that the transport protein undergoes allows us to potentially identify new binding sites which arise during this catalytic cycle
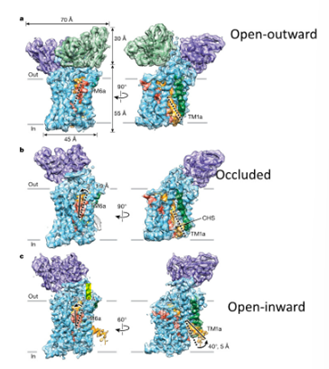
Identification of conditions which allow you to resolve the structure of the serotonin transporter in all three states using Cryo-EM
Found that in high NaCl conditions, favours the outward facing state, in low NaCl/KCl concentrations, favours the inward facing state
Used single particle analysis techniques to then resolve these structures
Electron microscope used to diffract the electron beam causing a diffraction pattern, and use this to back calculate the electron density
However, there’s no guarantee that the transport proteins you’re doing single particle analysis on will all be aligned so you group photos, align them and start to average them
Benefit of using electron diffraction is that it gives you a lattice structure so you can see the orientation of molecules relative to others
Images taken, averaged and aligned relative to each other which allows you to make out the overall shape of the transport protein
Transport cycle
TM helices 1 and 6 important for generating the binding site
Half channel leading to the central cavity surrounded by 6a and 1b
Transport of the molecule associated with the movement of 6 inwards (occluded state) and the open inward state associated with the movement of 1a outward
Broken helices next to the binding site are used as gates to allow the molecule in and out of the binding sites
Challenges of drug design and cryo-EM
Challenging to get a high-resolution structure with cryo-EM, but the molecules that we bind are about 6 angstroms in size —> need to be able to map high-resolution data onto the cryo-EM structure
Analyse all the possible orientations of the molecule in the binding site and calculate the interaction energy
Can then identify the best orientation of the molecule relative to the binding site
Can then to molecular dynamics to generate a family of molecular structures which you compare with the electron density that you’ve previously obtained
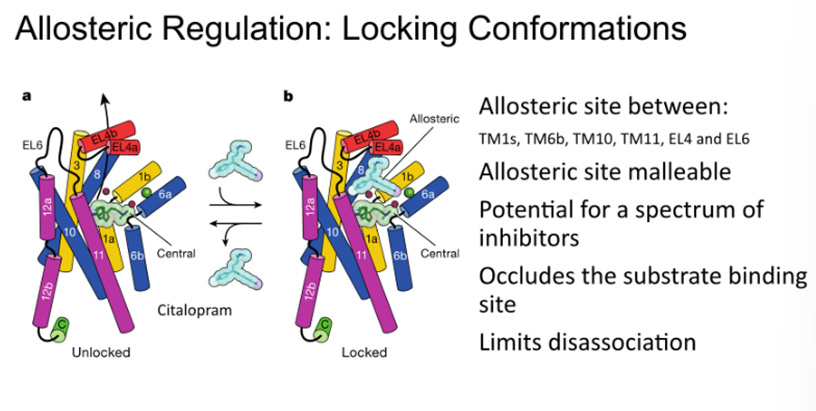
Allosteric regulation: locking conformations
Allosteric binding site which is important for inhibiting the binding of other molecules into the active site
Also locks the transport protein in its outward-facing conformation, therefore inhibiting the reuptake of serotonin
Identification of these allosteric binding sites may be important for the targeting of specific subtypes of receptors