Infrared (IR) spectroscopy
1/16
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
17 Terms
What is IR spectroscopy all about?
The absorption of infrared radiation as bonds in a molecule absorb infrared radiation at characteristic wavelengths
Infrared radiation on the electromagnetic spectrum
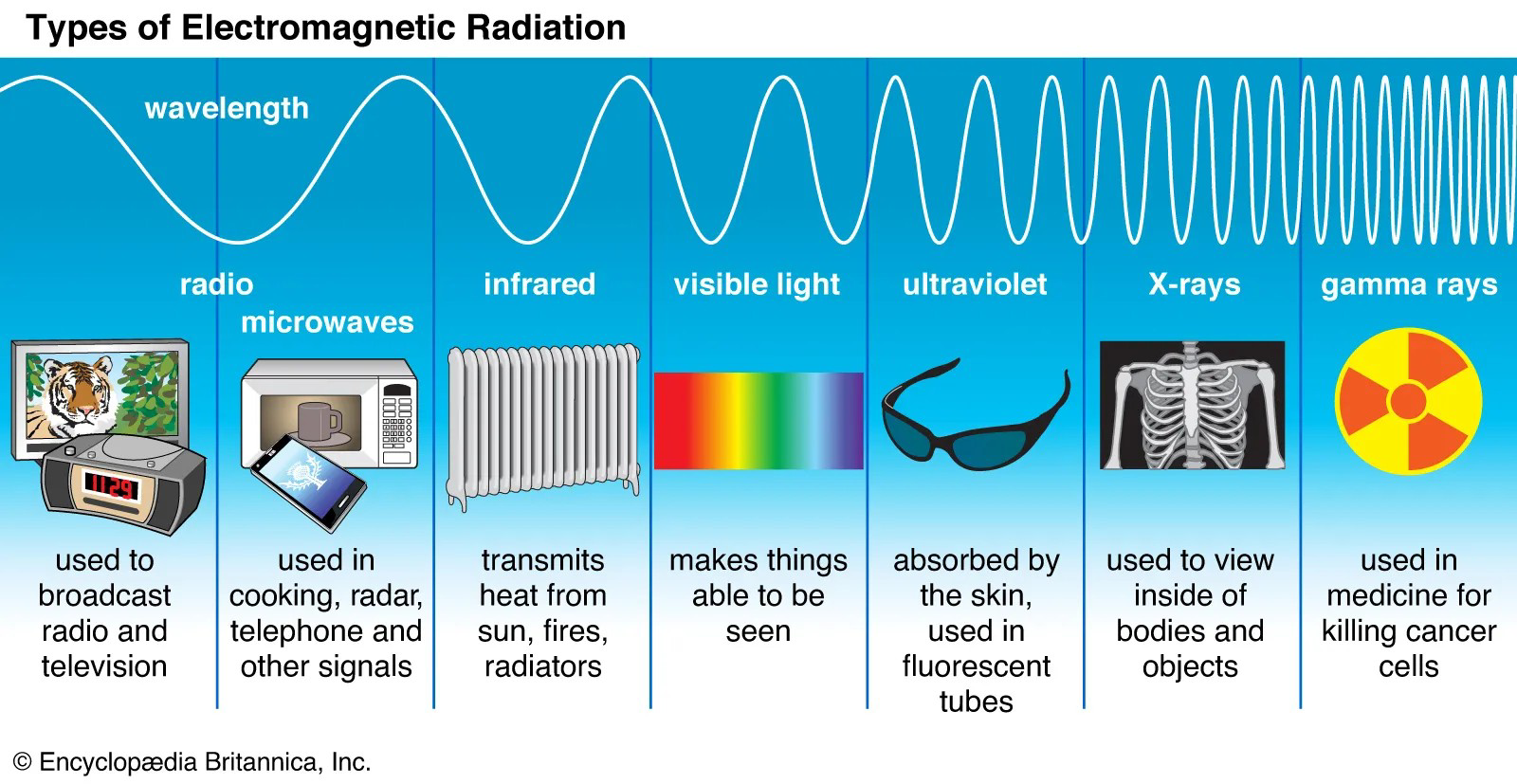
What is the core idea behind how IR spectroscopy works?
Infrared radiation comes from a source which hits a sample, for which some IR passes through the sample, some is absorbed by the sample completely and some is absorbed partially.
This radiation then hits a detector which is connected to a computer screen that shows an infrared spectrum as the computer read out
What is the radiation that passes through the sample referred as?
Transmitted radiation
What two things does the absorbance of IR radio action depend on?
Bond polarity and molecular vibrations
Is the absorption of IR radiation stronger or weaker in a polar bond?
Stronger
What will polar bonds show as in infrared spectrums?
As strong peaks
Why do C—C bond peaks hardly show up or not show up at all on infrared spectrums?
Because they are non-polar
Will C—H bonds show up on infrared spectrums and why?
Yes but as weak peaks because they are not entirely non-polar
Why may a C—H peak by large on an infrared spectrum?
Because there would no lots of these bonds present in the sample, lots of weak peaks will add together to give a stronger peak
What does it mean for molecules to vibrate?
A pair of atoms in a bond doesn't have a fixed position, and the bond doesn't have a fixed length.
The bond might stretch, and so the atoms will be further apart, which increases the bond length, or the bond might compress, and so the bond length will decrease.
This is called a stretching vibration which corresponds to the absorption of IR radiation
How else can molecules vibrate?
By bending or twisting
What does an infrared spectrum look like?
A graph with transmittance on the y-axis and wavenumber on the x-axis
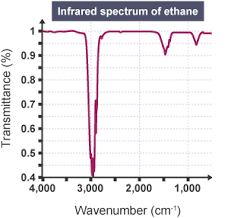
What does a transmittance of 100% mean? What would this look like on the graph?
No infrared radiation has been absorbed and so all of it has been transmitted. This would look like one straight flat line at 100%
Why is wavenumber used on the x-axis instead of frequency? What is particular about the values plotted on the x-axis?
Wavenumber is proportional to frequency. It makes the numbers easier to understand as frequencies can vary too greatly.
It starts at 4000 then decreases to 0
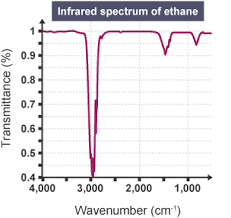
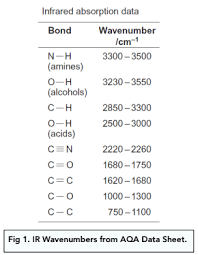
What do you use to identify the bonds when reading off the wavenumber values on a infrared spectrum?
A data sheet
What is the resonance frequency?
The specific frequency at which the molecules will vibrate to stimulate larger vibrations