The Reproductive System
1/79
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
80 Terms
pulsatile signaling, feedback regulation, cell-cell cooperation, precise timing
what are the core principles of reproductive physiology and genetic sex?
SRY gene
sex-determining region of the Y chromosome (also regulates other genes) that converts non-differentiated gonad medulla into a testis
developing gonads after egg is fertilized looks same for male and female (bipotential), but have the potential to develop into testis or ovaries (and turns into testis if this is present)
sertoli cells: spermatogenesis
leydig cells: function in early days; secrete testosterone, which can be converted to DHT (both hormones important in development of male phenotype)
what two cells develop when testis develop?
testosterone
develops male internal genitalia; associated with testes, develop structures like vas deferens, epididymis, seminal vesicles (glands that secrete large amounts of semen)
newly formed tests secrete this
2 duct system
Wolffing duct and Mullerian duct; initially there are both, but depending on male hormones (or not), one will be disintegrated and one won't
dihydrotestosterone (DHT)
determines which sex external genitalia will develop; key for external genitalia development associated with testes, including prostate gland (contributes to small portion of semen)
lack of enzyme that converts testosterone to this = lack of external gentialia development
male: Mullerian duct gets disintegrated after Wolffing duct starts developing → structures like epididymis and vas deferens stem from this, which are tubes to transfer sperm out
female: without testosterone, no Wolffing duct develops, ovaries develop from bipotential gonads, Mullerian develops into tube and uterus/upper vagina (internal female genitalia)
***also lack of DHT means female external genitalia will develop
how does the development of male vs female internal genitalia look after determination of the fate of the 2 duct system?
anti-mullerian hormone/Müllerian inhibiting substance (MIS)
leads to mullerian ducts degenerating; secreted by male individuals to suppress development of internal female genitalia
newly formed testes also secrete this, and can be an indicator of male fertility issues if too high
without DHT, you can't have male external genitalia growth (bc signal is missing) and female external genitalia will develop instead
even without DHT, if you have testosterone male internal genitalia (ducts and glands) should be present
SRY = male gonads (testis) are present
answer the question and explain

one polar body and one mature egg, 4 haploid spermatids being produced
female egg doesn't do meiosis II until fertilization (meet with a sperm), but males do right after meiosis I
***most eggs are primary oocytes in females bc they don't divide until they are fertilized
female meiosis II ends in ___, and male meiosis II ends in ___. female oocyte does meiosis II when ___, and male spermatocyte does meiosis II when ___
begin with diploid germ cells
involve mitosis and meiosis
produce haploid gametes
require meiosis I and meiosis II
hormonally regulated
what are the similarities between spermatogenesis and oogenesis?
timing
arrests
product
pattern
what are the differences between spermatogenesis and oogenesis?
female division is longer; by month 5 of fetal development all female mitosis is finished (all eggs a female will ever have are produced), and later she starts to lose them → once puberty hits some eggs will continue on to meiosis I
males continue to have mitosis after birth, and have a longer reproductive life bc they have continual meiotic division (after puberty)
describe the timing difference between spermatogenesis and oogenesis phases
female is different to prioritize resource allocation into egg developing and differentiating (unlike sperm); in meiosis I, the dividing cell (primary oocyte) splits into a polar body (nonfunctional) and a secondary oocyte because you want all resources to go into one egg at a time
what is the reason for the timing difference between spermatogenesis and oogenesis phases?
by the 5th month of fetal development: mitosis is completed and chromosome duplication → primary oocyte
resume at puberty:
1. first meiotic division: secondary oocyte and a first polar body
2. second meiotic division: only if fertilization happens; one egg and a second polar body
describe the timeline of female oogenesis; when do mitosis and each meiotic division occur, and what products do you get at the end of each?
before birth: only mitosis
at puberty: mitosis resumes, meiosis starts (chromosome duplication and two divisions)
1. chromosome duplication: primary spermatocytes
2. first meiotic division: two secondary spermatocytes
3. second meiotic division: four spermatid
describe the timeline of male spermatogenesis; when do mitosis and each meiotic division occur, and what products do you get at the end of each?
hypothalamus: secrete GnRH (gonadotropin releasing hormone)
anterior pituitary: secrete FSH and LH when triggered by GnRH
gonads (testis/ovaries): secrete sex steroids (testosterone/estradiol) and inhibins (inhibit FSH secretion)
what are the roles of the hypothalamus, anterior pituitary gland, and gonads in hormonal control of reproduction?
sex steroids inhibit GnRH (ultra long loop) and FSH/LH secretion
describe the negative feedback loop in hormonal control of reproduction
pulsatile signaling
gonadotropins are not secreted when GnRH is not released in a oscillating fashion; if this is inhibited, development in puberty is altered (early or later impacts onset of puberty)
before puberty normal ___ release of GnRH is inhibited, but during puberty you do have it
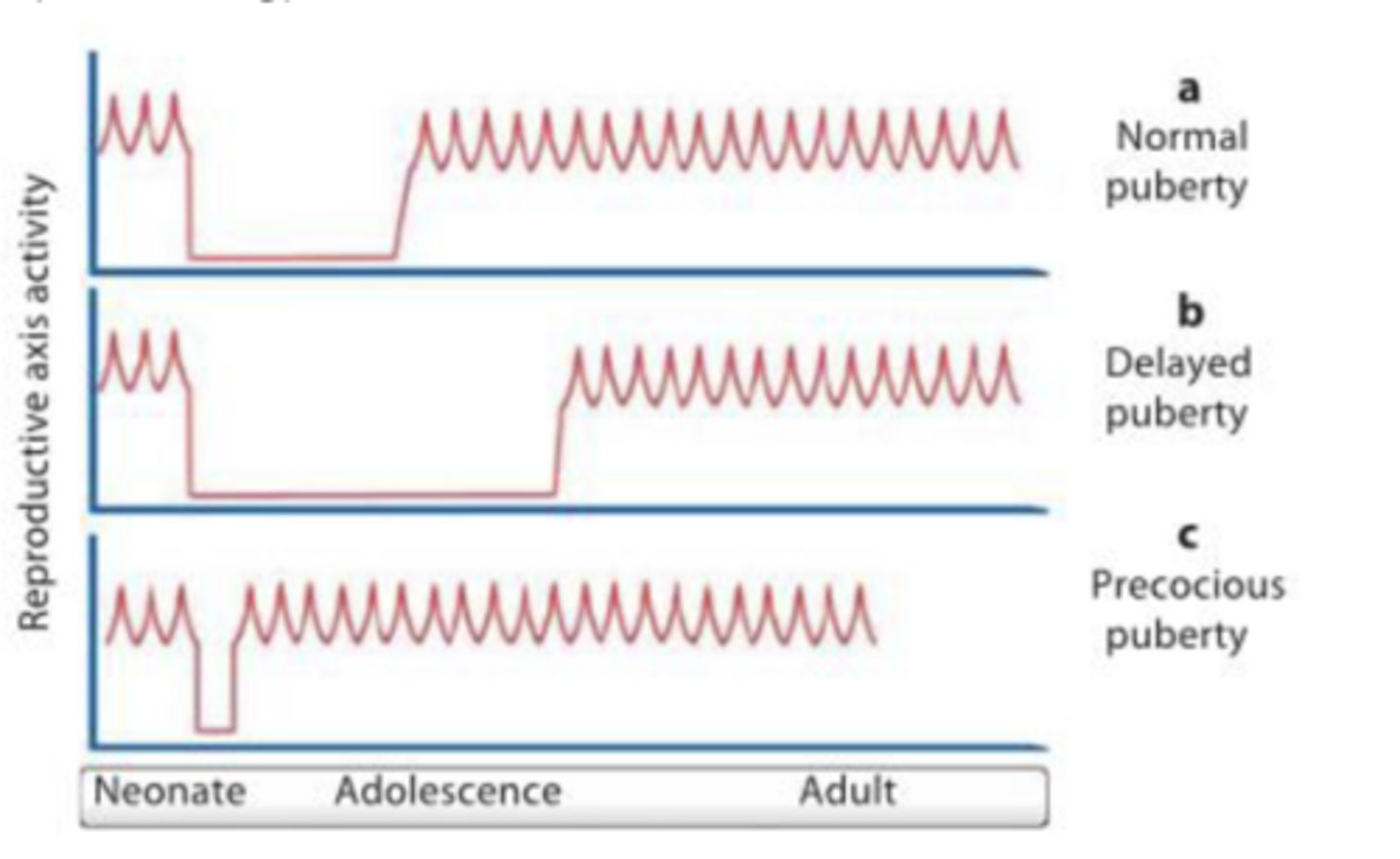
cells in anterior pituitary (release LH and FSH) respond to GnRH → if you constantly release GnRH the receptors will lose sensitivity to it
GnRH acts on both FSH and LH release → if you fluctuate slower FSH is released, if you fluctuate faster LH is released
answer the question and explain

seminiferous tubule
highly convoluted tubule where sperm develop (spermatogenesis)
epididymis
sits on top of testis and where sperm fully mature; sperm from seminiferous tubules are released into this
ductus deferins
muscular layer of tubing that transports sperm out
sperm production, production and secretion of testosterone
as sperm develop/mature they go from more peripheral to lumen (inner) → when released from lumen they go into epididymis
what are the main functions of the testis? how do sperm travel through them?
sperm are most immature in the most peripheral region and mature more as they move more inner
from outermost/peripheral (least mature) to innermost (most mature) in seminiferious tubule: stem cell spermatogonium → mitosis into primary spermatocyte → first meiosis into secondary spermatocyte → second meiosis into spermatid → spermiogenesis into spermatozoa
describe the phases of spermatogenesis throughout the seminiferous tubules
spermiogenesis
maturation of spermatids (immature haploid gametes) into sperm cells; genetically identical, but morphological changes occur where it develops an acrosome and flagellum (head and tell), receptors to detect where eggs are
basal compartment: where only mitosis happens
adluminal compartment: after sertoli cells "let go of each other's hands," main location where cells differentiate
differentiate the basal and adluminal compartments of the seminiferous tubules
blood testis barrier
very tightly controlled barrier that keeps developmental stages of spermatogenesis out of outside control (blood)
sertoli cells
promote and sustain sperm development (spermatogenesis); secrete growth factors for sperm development, secrete androgen binding protein, blood testis barrier, phagocytosis of defective sperm
very large cells that provide structure and are in close contact with developing sperm
secrete androgen binding proteins, inhibin, and MIS
sertoli cells "hold hands" with adjacent ones, creating tight junctions with neighboring cells → when spermatogonium go through mitosis and differentiate into primary spermatocytes, the "hands" release temporarily so cells can move through and differentiate as they move towards lumen
describe how sertoli cell junctions change as sperm differentiate

androgen binding proteins
because sertoli cells don't generate testosterone themselves, to have high concentrated level they produce these
helps sperm develop
support spermatogenesis; these cells express receptors for FSH and their functions rely on them → if FSH drops, these cells can't survive and thus can't support spermatogenesis
in response to FSH and testosterone sertoli cells...
leydig cells
produce and secrete testosterone, developing male secondary sex characteristics; close to blood vessels in peripheral testis
triggered by LH, also important for spermatogenesis
they're active in fetal life to support gonadogenesis (develop internal genitalia), more inactive after birth, and active again at puberty and onwards to support sexual maturity/function/secondary sex characteristics
how does leydig cell activity change throughout a male's lifespan?
FSH acting on Sertoli cells and LH-driven testosterone from Leydig cells
feedback loop in sertoli cells: FSH acts on sertoli cells → release inhibin → less FSH
feedback loop in leydig cells: LH triggers testosterone secretion → inhibition of LH secretion
***at a higher level testosterone can inhibit GnRH
spermatogenesis requires coordination between...
describe the feedback loops involved in this
secondary sex characteristics
sex-specific features that are not essential for reproduction, but are part of the sexual phenotype → impacted by testosterone and DHT
general body shape, including pattern of muscle development, body/facial hair, deep voice, male sexual behavior (libido)
spermatogenesis and spermiogenesis, CNS, metabolism, and accessory glands/organs are also affected by T/DHT
excess testosterone can increase DHT, which increases inhibition of FSH and LH (feedback loop)
testosterone becomes estradiol via aromatase activity → with excess testosterone you will have excess estradiol (and thus estrogen)
answer is A
case study: a male has been taking testosterone because he believes it will help enhance the results of his intense weight training, but has been experiencing trouble with having a successful pregnancy with his wife. due to this they consult an infertility specialist.
what will his bloodwork reveal? (see answer options in the picture)

because of decreased FSH, which is important for control of spermatogenesis → without it testis will shrink, sertoli cells will not function, spermatogenesis significantly decreases
case study: a male has been taking testosterone because he believes it will help enhance the results of his intense weight training, but has been experiencing trouble with having a successful pregnancy with his wife. due to this they consult an infertility specialist.
they find that he has a low sperm count, but he is confused because he has read online that T stimulates spermatogenesis.
why does he have a low sperm count?
A is dependent on NO, GC, etc. (ANS) → this pathway is not hormonal
polycythemia (overproduction of RBCs) can be caused by increased testosterone (ex. performance enhancing)
bc you have more estrogen being converted, this can cause gynocomastia
enlargement of prostate (NOT atrophy) because of excess DHT, but testis become smaller
case study: a male has been taking testosterone because he believes it will help enhance the results of his intense weight training, but has been experiencing trouble with having a successful pregnancy with his wife. due to this they consult an infertility specialist.
what other effects would be caused by injected testosterone? (see answer options in the picture)

male: continuous gamete production, stable negative feedback
female: cyclic gamete production, negative AND positive feedback
what are the differences in pattern of gamete production and feedback between males and females?
ovarian cycle
females have cyclic patterns of FSH and LH release; their secretion fluctuates dynamically across this with
LH has bigger role in gamete production and surges due to positive feedback
2 phases with event in between (follicular, ovulation, luteal)
about 28 days
not only inhibitory (negative) but also stimulatory (positive)
the long-loop feedback action exerted by the ovaries on the hypothalamo-pituitary axis is ___
pituitary gonadotropins
work on gonads (like ovaries)
FSH and LH
FSH (female effect)
stimulates granulosa cell growth (cells that surround oocytes, like sertolis), stimulates synthesis of aromatase (key enzyme), and induces LH receptors on granulosa cells
aromatase
converts androgen into estradiol
LH (female effect)
stimulates theca cell growth and promotes oocyte maturation (remove inhibition so you can complete meiosis I), triggers ovulatory (inflammatory-like) events, stimulates progesterone synthesis
***oocyte development and maturation are diff hormones
ovulation: lots of structural barriers are in place before ovulation, so enzymes help break them down so oocytes can leave ovaries into fallopian tubes and after this, the cells left behind are luteinized
progesterone: theca cells become corpus luteum that secrete hormones due to LH
how does LH trigger ovulation and progesterone synthesis?
follicle
functional unit of ovary; has egg inside and surrounding cells help egg's growth and development
granulose cells
more impacted by FSH, cells that surround oocytes and help its growth and development
in later stage respond to LH
theca cells
more impacted by LH, outer layer
synthesize androgen, which later gets converted by aromatase
answer is D
FSH increases LH receptors in the granulosa cells
LH is responsible for B, C, and E
***Luteinization = converting tissue to corpus luteum
answer the question and explain

leutinization
converting tissue to corpus luteum; theca cells left behind from follicles become corpus luteum
answer is C; LH matures follicle to allow maturation of oocyte
normally LH does luteinize cumulus cells (the ones left behind that get converted to become progesterone secreting), BUT in this case that is not why you give LH (bc this occurs later on after ovulation happens)
answer the question and explain

follicular phase
progressive growth of ovarian follicles, resulting in outgrowth of just one large one of them (dominant follicle) which buds on ovarian surface
ovulation
dominant follicle bursts and the oocyte is expelled into fallopian tubes
luteal phase
most of cells that formed the follicle wall before remain in ovary and give rise (change shape and function) to secretory structure called corpus luteum
corpus luteum degenerates spontaneously if pregnancy doesn't occur
at start follicles are few and underdeveloped (not secreting many steroid hormones) → lack of negative feedback causes increase in GnRH and gonadotropins → FSH causes growth of follicle and increase in estrogen → negative feedback causes a decrease in gonadotropins and rest of follicles (besides dominant one) are killed without FSH → the dominant follicle continues to secrete estrogen, which when it gets to a high level causes POSITIVE feedback and an LH surge (and FSH to a much smaller degree)
describe the steps of the ovarian cycle from having few follicles to making a dominant follicle
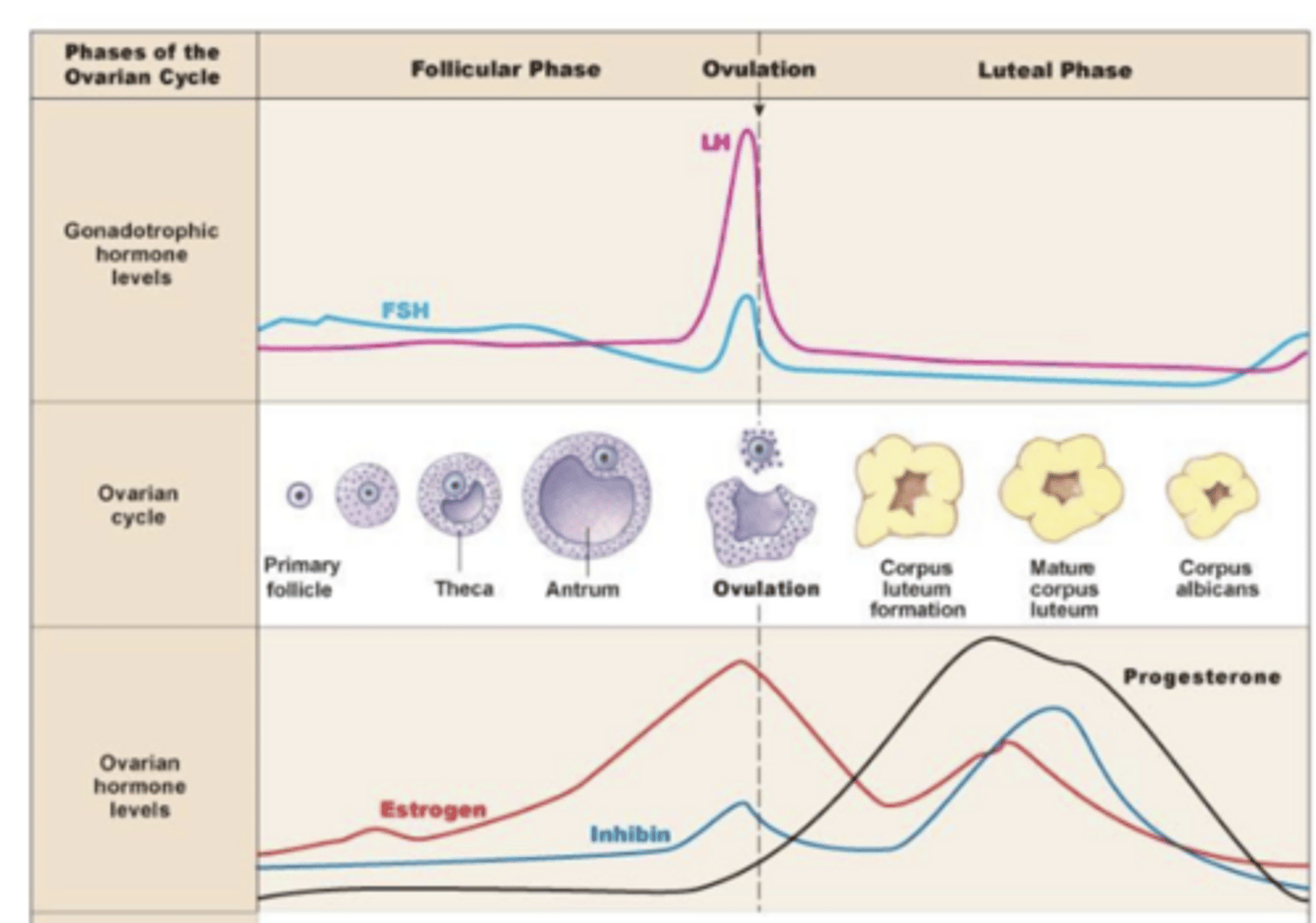
LH stimulates corpus luteum (from theca cells) to grow → "surge" of more secretion of estrogen and progesterone (higher than estrogen) → LH goes back down, and corpus luteum then degenerate → corpus albicans (degenerated version of corpus luteum, which can't secrete progesterone/estrogen)
describe the steps of the ovarian cycle from making corpus lute to corpus albicans
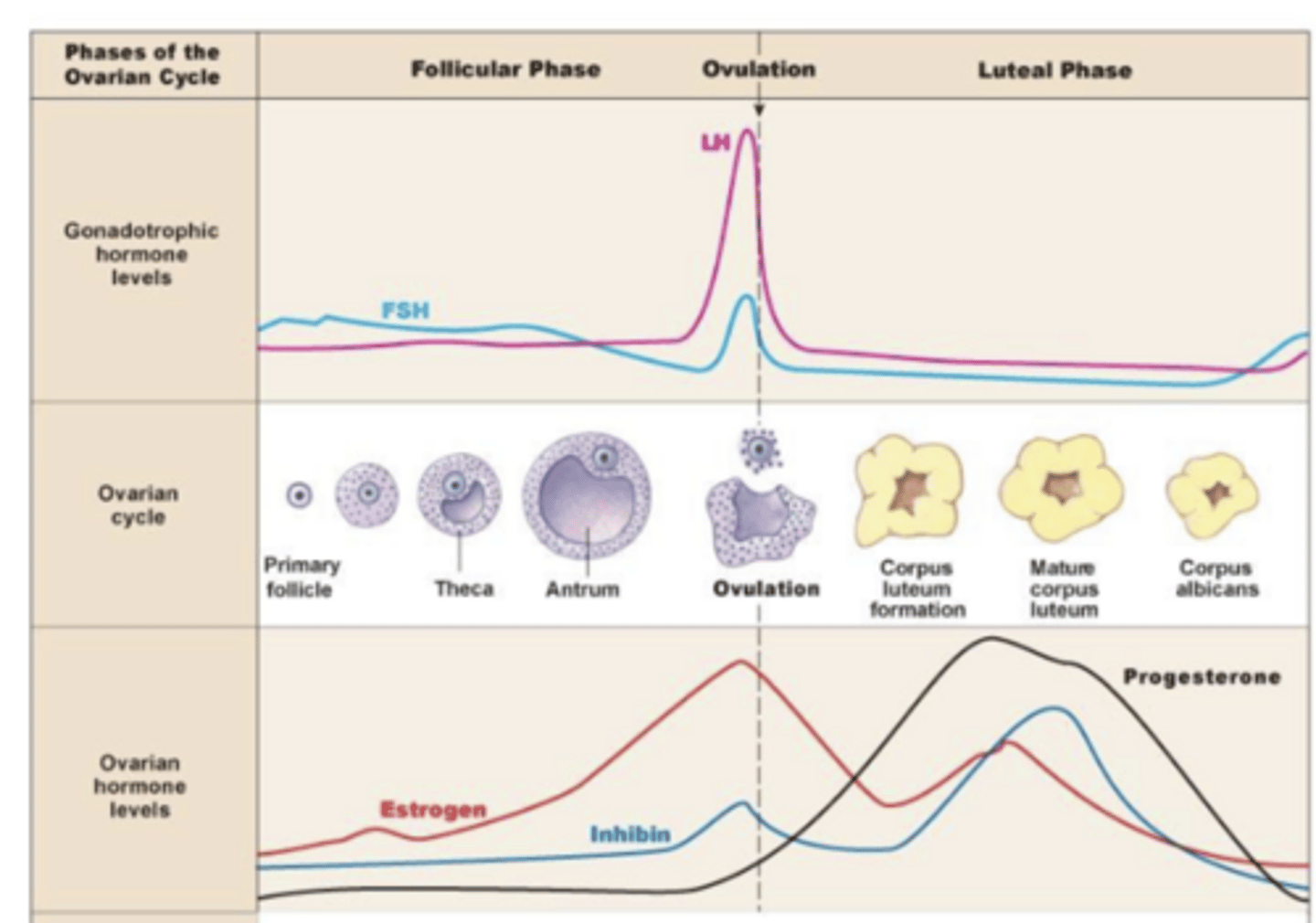
progesterone
keeps uterus stable and secreting, limits motility, negative feedback on gonadotropins
change in uterine lining
when gonads secrete the steroid hormones estrogen, progesterone, and inhibin, ___
uterine cycle
supports implantation of a fertilized egg and supports early stages of embryonic development
2 phases and 1 event (proliferative, secretary, menstruation)
proliferative (estrogen) phase
uterus wall grows; when estrogen is high, growing back stratum functionalis
secretory (progestational) phase
uterus wall grows and has lots of secretion; where a fertilized egg can implant bc wall is ready
perimetrium
outermost serous layer of uterus
myometrium
smooth muscle of uterus
endometrium
mucosal lining of uterus, contains stratum basalts and functionalis
stratum basalis
layer of endometrium that continues to be generated to promote growth
stratum functionalis
layer of endometrium that sheds and grows back in menstruation
sheds when steroid hormones are low
involution of corpus luteum causes drop in steroid hormone levels → driven by increasing amount of estrogen produced by the developing follicles → driven by estrogens and especially progesterone produced by the corpus luteum after ovulation
explain how drop in steroid hormone leads to shedding of the stratum funcitonalis
estrogen
from granulose cells in follicular phase and corpus lute in luteal phase
supports follicular development (early) and endometrial proliferation (later)
negative feedback mostly, but positive feedback pre-ovulation
progesterone
from corpus luteum
maintains secretory endometrium, decrease uterine contractility, supports early pregnancy
negative feedback on GnRH/LH
inhibin
from granulosa
modulates follicular recruitment
selectively inhibits FSH secretion
capacitation, acrosome reaction, zona penetration
when sperm is ejected into vagina it doesn't have fully capacity, but environment facilitates their ability to reach egg (locomotion and olfaction)
***if this doesn't happen it will be unable to actually properly fertilize an egg
acrosome reaction (zona penetration = when zona pellucida is broken down)
what is the solution to the problem of how sperm reach and penetrate the oocyte?
acrosome reaction
there is a barrier that can prevent sperm from coming in (zona pellucida), so when sperm bind to zona pellucida (olfactory receptors guide this), this occurs which allows enzyme layer of sperm heads to release the enzyme and to contact the zona pellucida and digest it and so sperm can make its way into oocyte
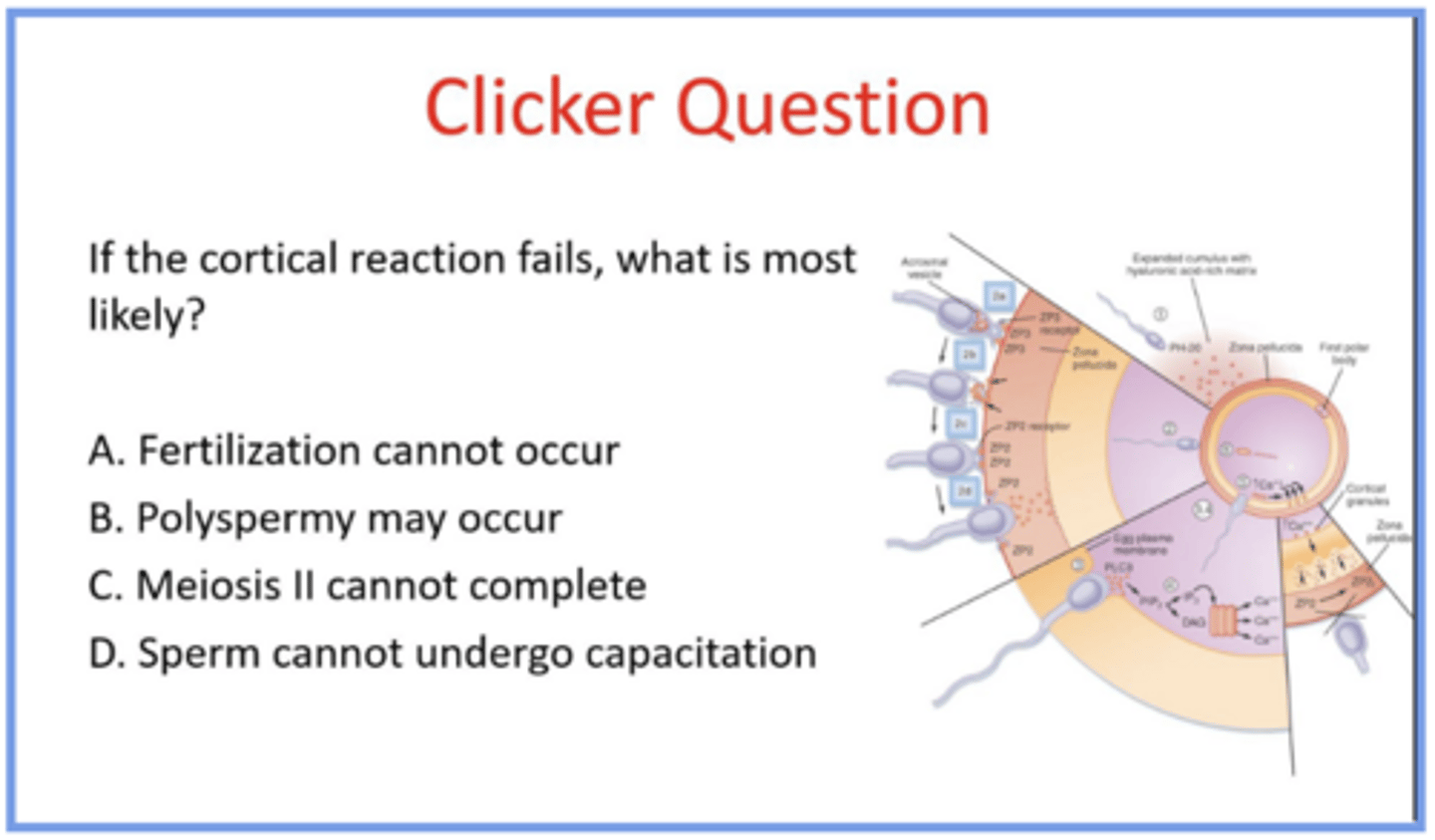
cortical reaction modifies the zona pellucida and blocks polyspermy
what is the solution to the problem of how the oocyte prevents polyspermy?
cortical reaction
after penetration of zona pellucida sperm binds to oocyte membrane, causing a Ca wave and release of cortical granules → this enzyme changes zona pellucida receptors so other sperm can't bind to it and thus only one sperm reaches the egg (block polyspermy)
the sperm cell membrane fuses with the oocyte membrane → triggers the completion of meiosis II in the oocyte → mature haploid egg has formed and a second polar body is extruded → zygote forms
what is the solution to the problem of how fertilization initiates development?
no cortical reaction = polyspermy may occur
answer the question and explain
fertilization occurs at ovulation → once egg is ovulated it does not last long (24-48 hours), but sperm can last around 5 days (can survive in female reproductive tract) → blastocyst ready to implant in uterine wall ~ 6 days after fertilization when progesterone level is very high and uterine endometrium is primed and ready for fertilized egg → implanted fertilized egg (developing embryo) starts to release human chorionic gonadotropin (hCG) to "rescue" corpus luteum to help embryo develop
explain the process of embryo implantation
human chorionic gonadotropin (hCG) works on gonads (ovaries) to replace LH when it drops and keep corpus luteum primed for another few months
this is why first 3 months of pregnancy is shaky bc embryo secretion of hCG is critical to keep uterus quiet (after 3 months placenta is developed and can help sustain)
describe how the corpus luteum is rescued when an egg is fertilized