Comprehensive Flashcard Set on Water, Solutions, Acids, Bases, and pH for Chemistry
1/68
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
69 Terms
In a H2O molecule the oxygen is
partially negative
In a polar H2O molecule the Hydrogen is
partially positive
Water can form_______ _________ between molecules.
hydrogen bonds
Water molecules stick to each other
cohesion
Water molecules stick to other polar molecules
adhesion
H2O molecules on the surface form a skin because of
surface tension
It takes a lot of heat to raise the temperature of water because of
high specific heat
Water dissolves most things
universal solvent
Substance that can do the work of dissolving something else
solvent
Substance that can be dissolved
solute
Does not interact with water
hydrophobic
Interacts with water
hydrophilic
Ice floats on liquid water because
low solid density
Tastes sour, corrodes metals, react with bases
acids
releases H+ ions in water
acids
taste bitter, chalky, feel soapy, slippery, react with acids
bases
releases OH- ions
bases
based off of H+ concentration
pH scale
goes from 0-14, 7 is neutral
pH scale
10 pH is how many times as basic as a pH of 7
1000
A base combined with an acid creates
H2O + a salt
solute
the substance that is being dissolved in a solution; generally, the substance with the lesser amount present in solution
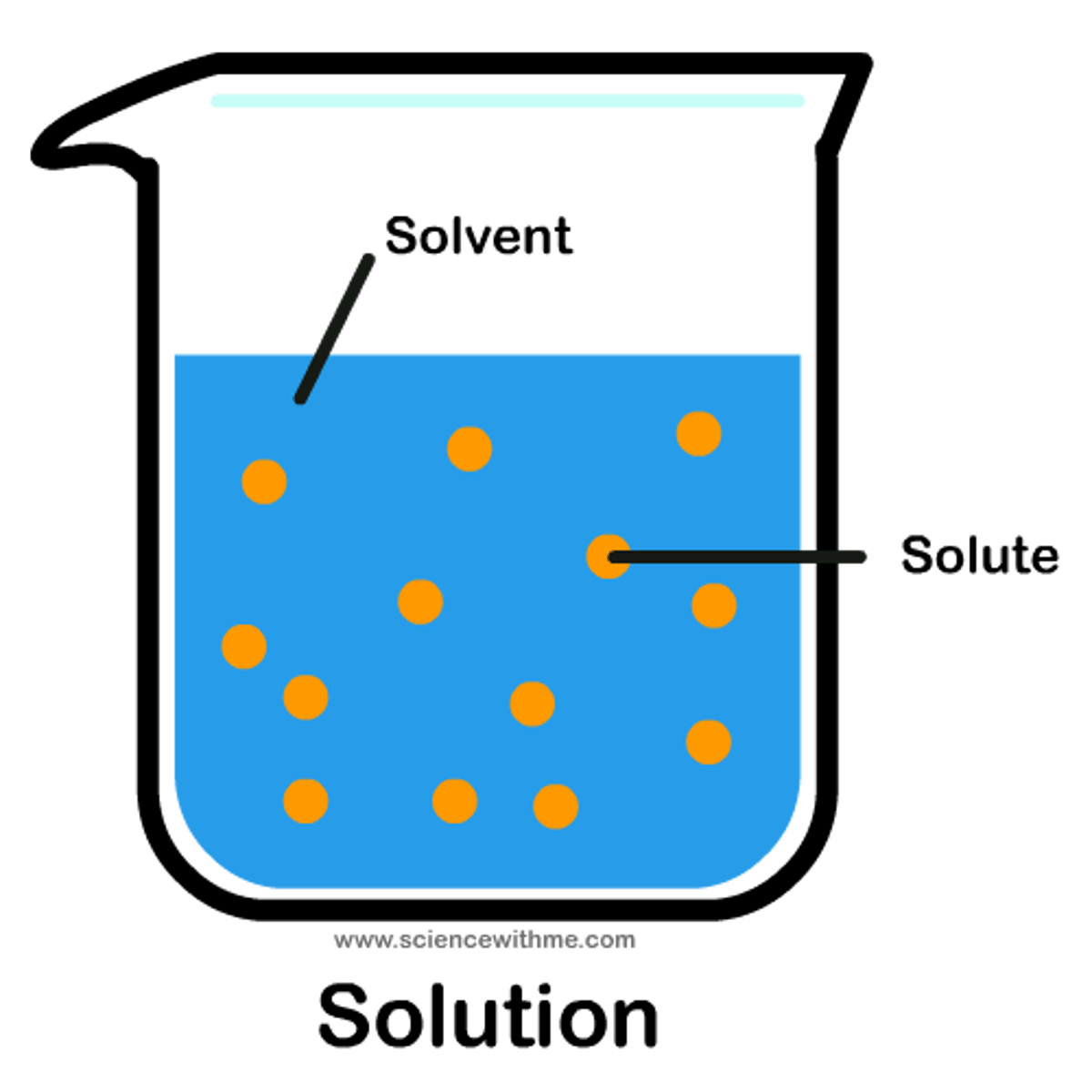
solvent
the substance that dissolves the solute; generally, the substance with the greater amount present in solution
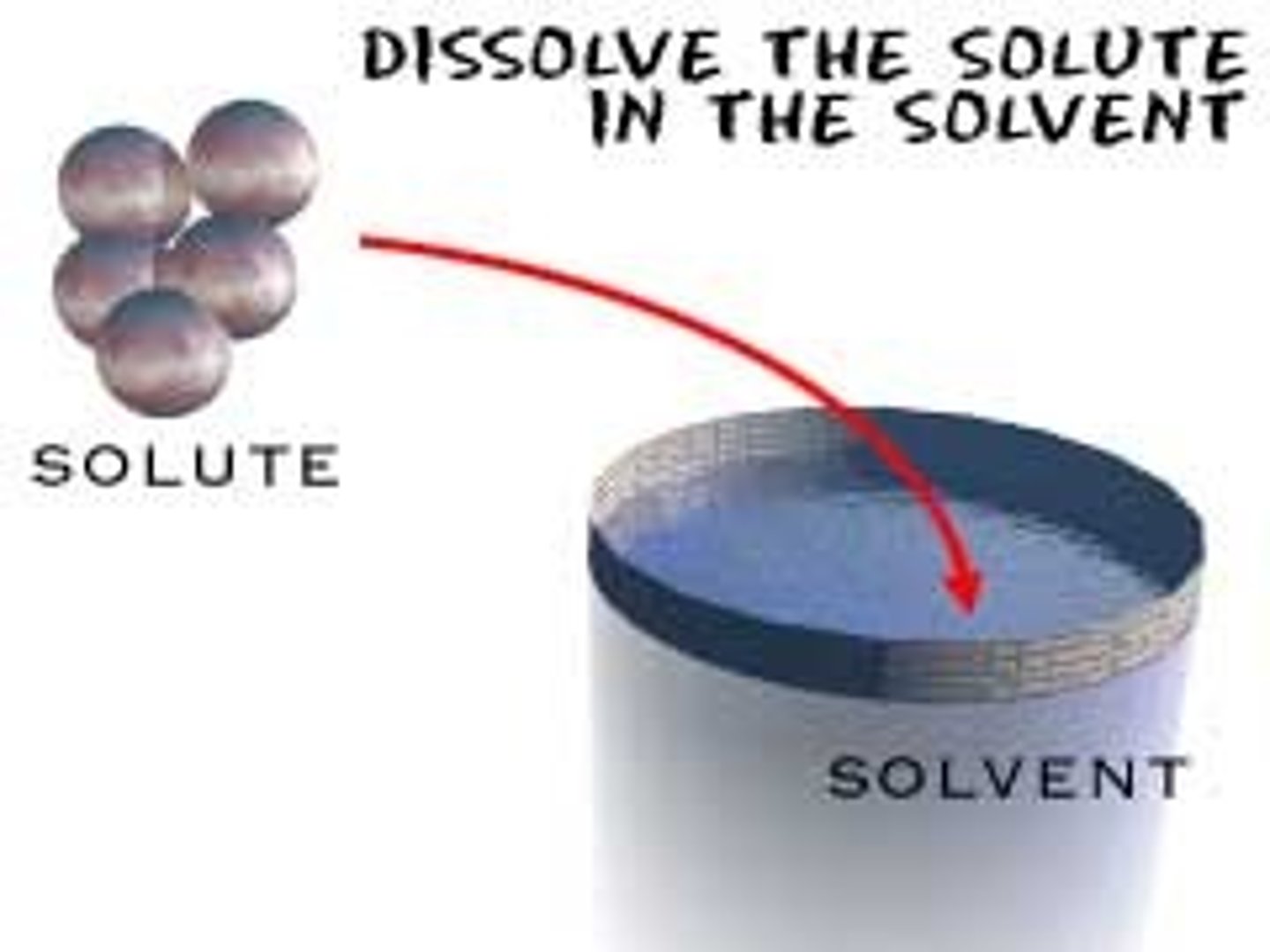
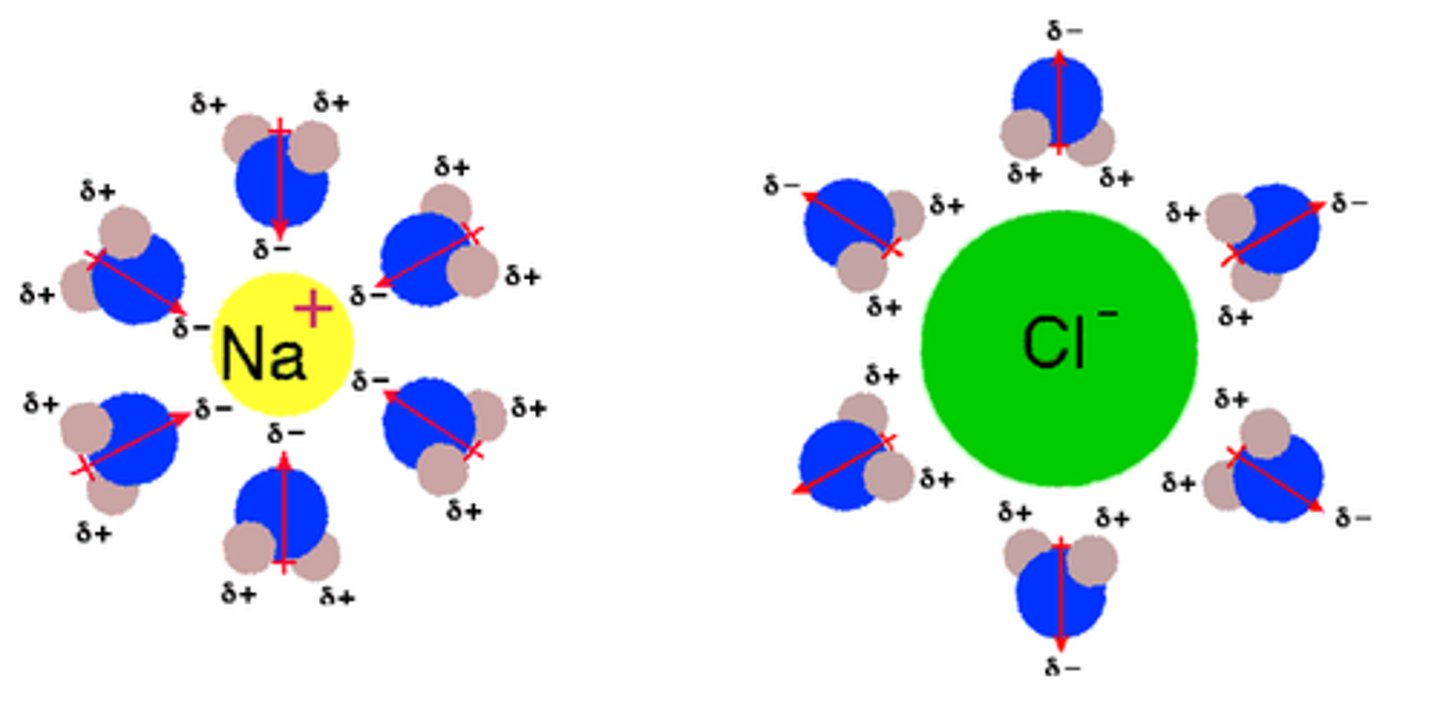
dissociation
the dissolving process that results in ionic compounds physically separating into component ions
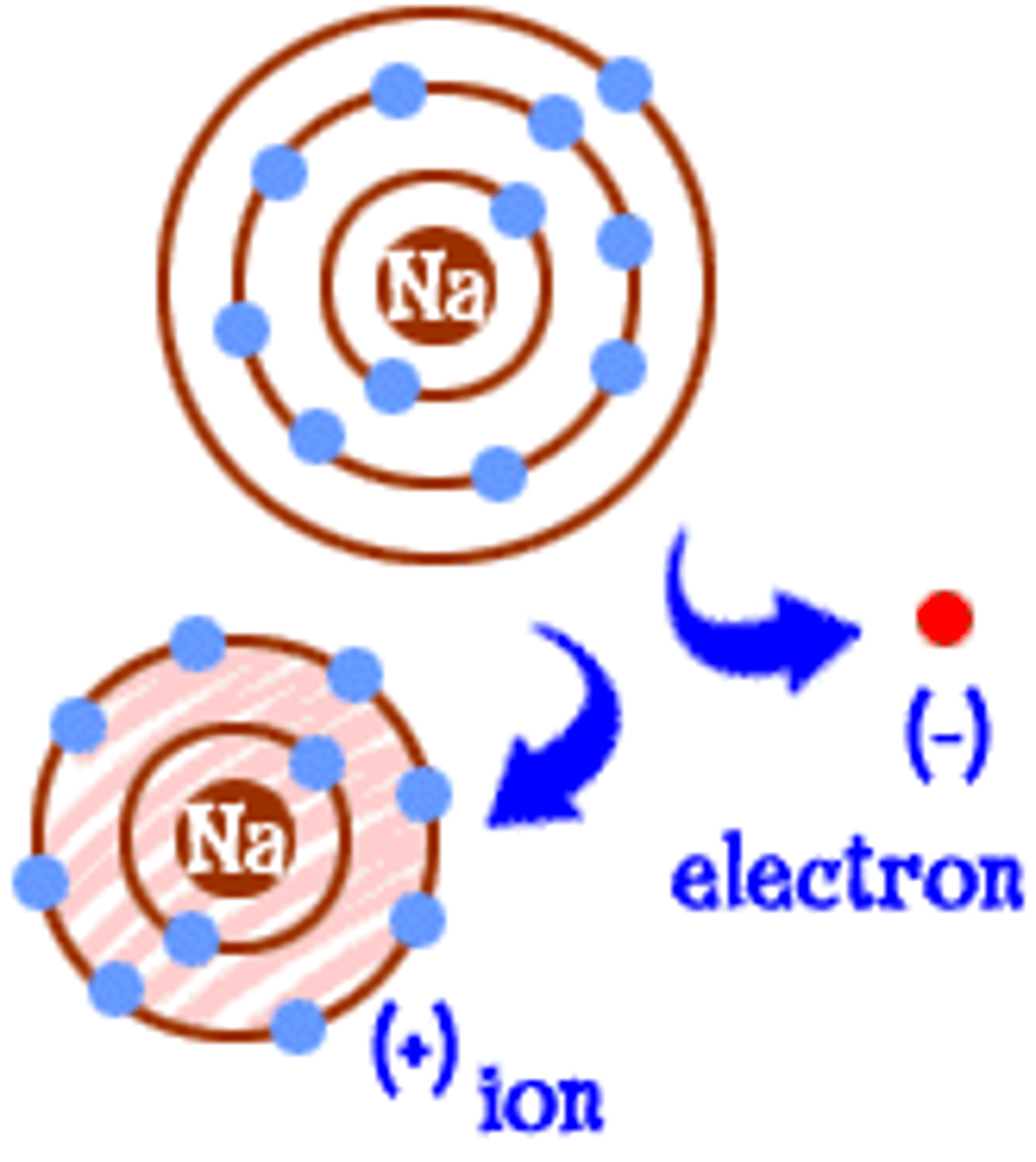
ionization
the chemical dissolving process in which neutral substances become charged ions
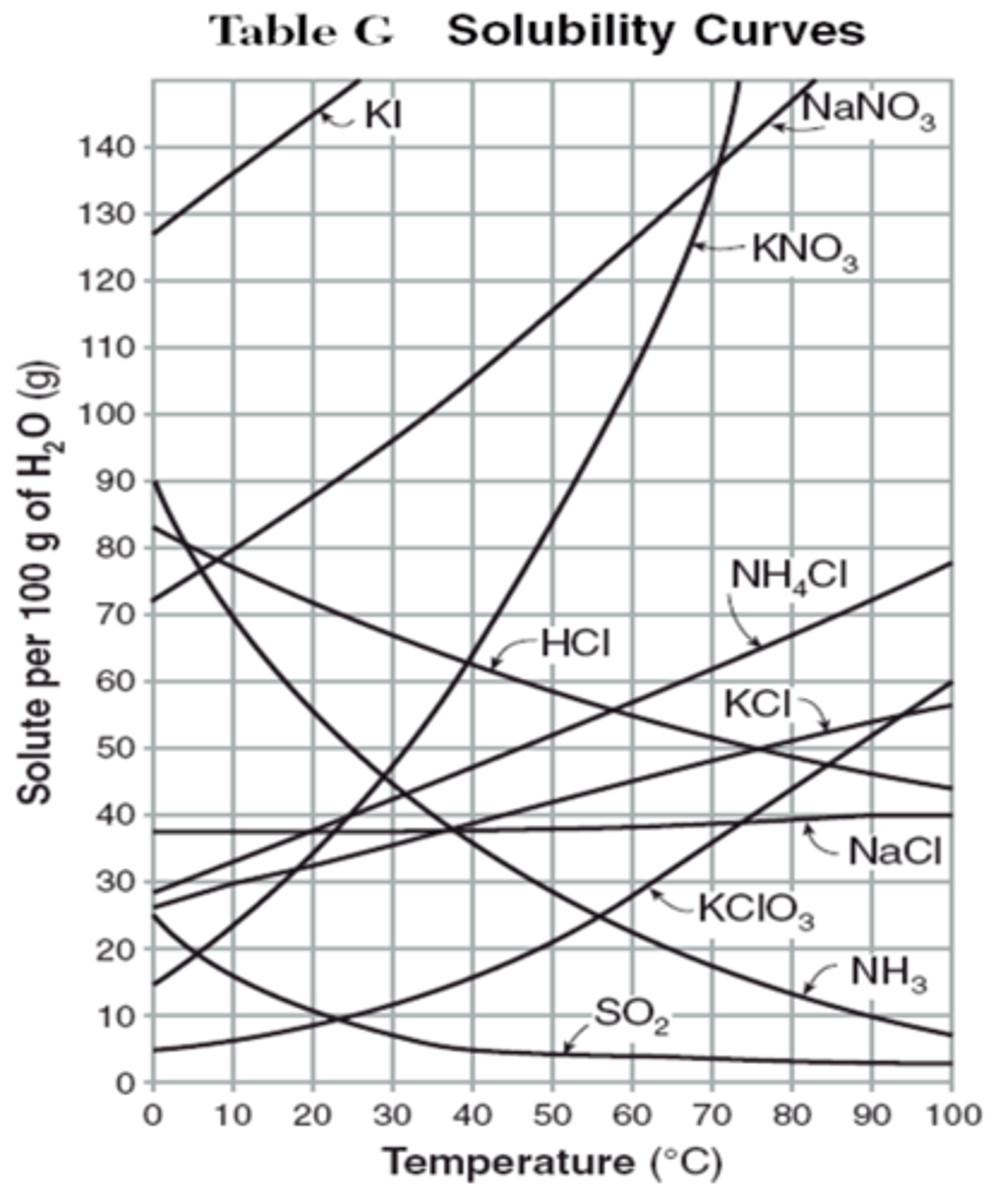
solubility
the maximum amount of solute that can dissolve in a certain amount of solvent at a particular temperature
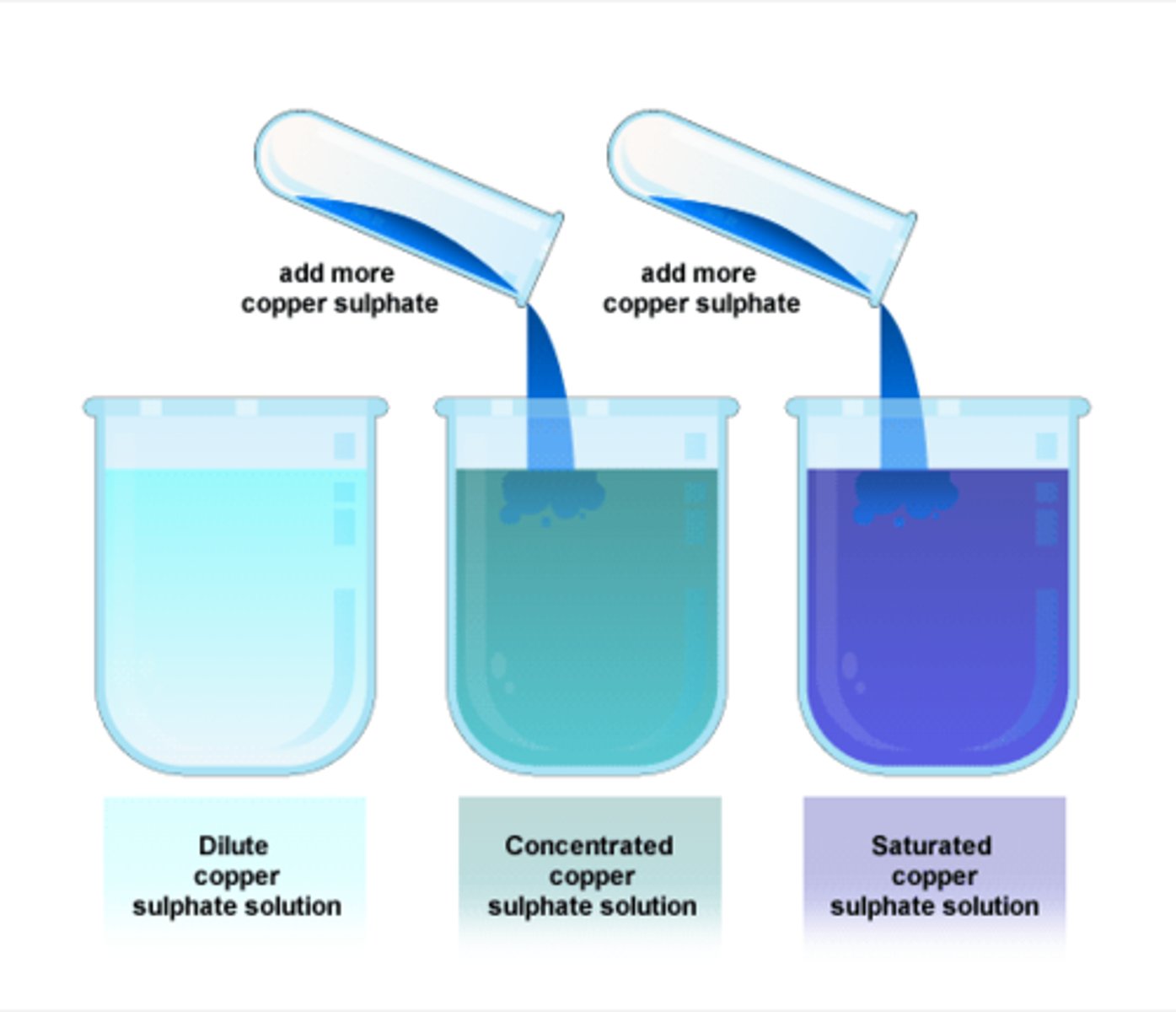
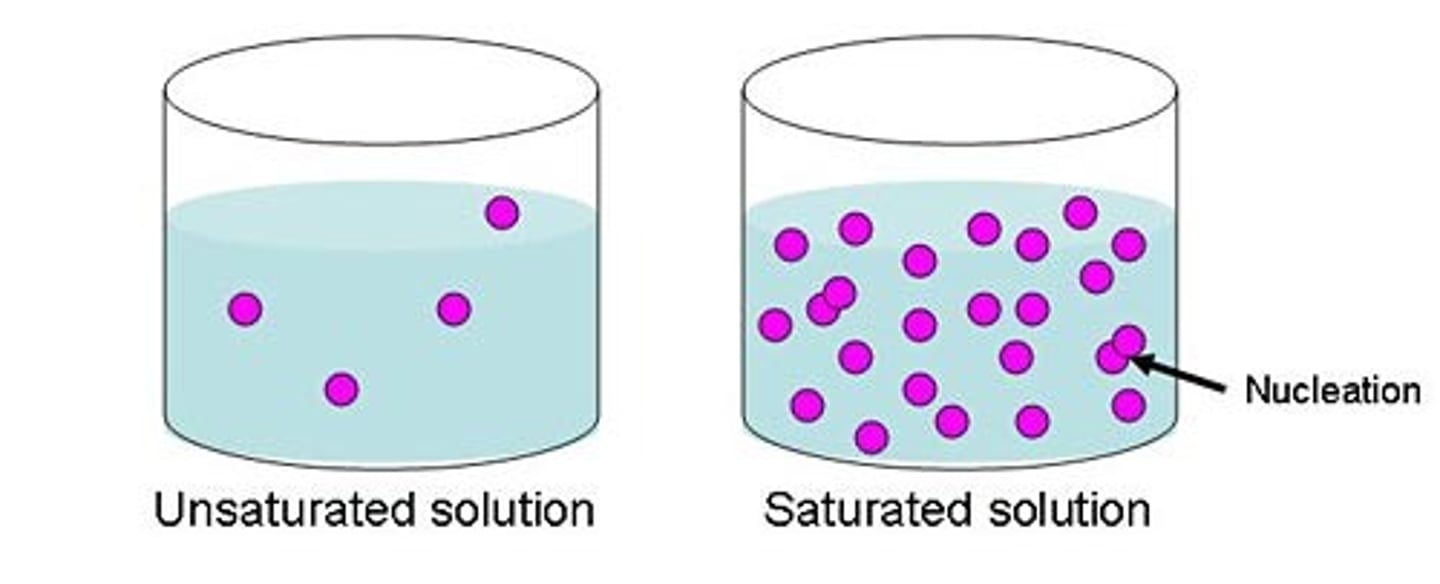
saturated solution
a solution in which no more solute can dissolve at a particular volume and temperature
unsaturated solution
a solution in which more solute can still dissolve at that volume and temperature
supersaturated solution
a solution in which more solute has dissolved than normally expected at that volume and temperature; usually achieved by careful heating and cooling

concentration
the amount of solute in a given amount of solvent; can be reported molarity (M)

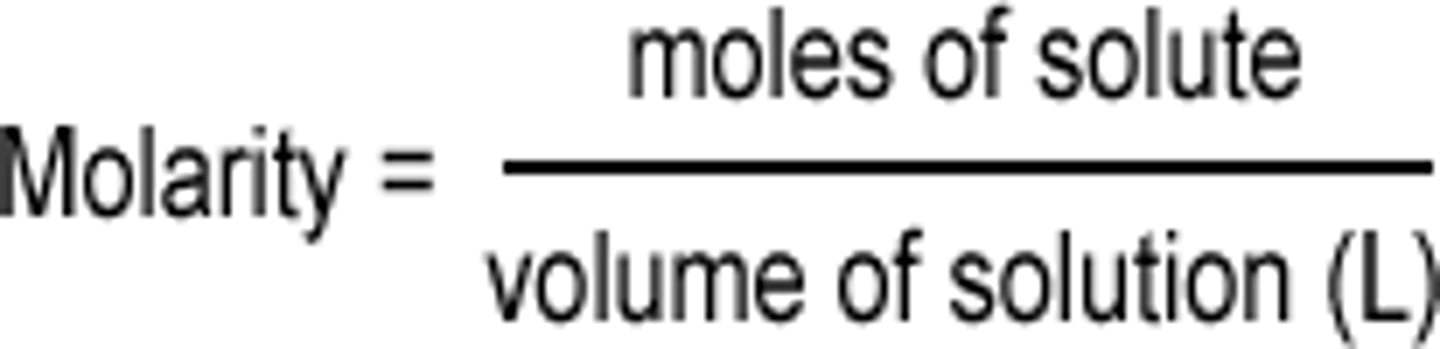
molarity (M)
concentration reported as the number of moles of solute per liters of solution
Acid
Has a pH less than 7
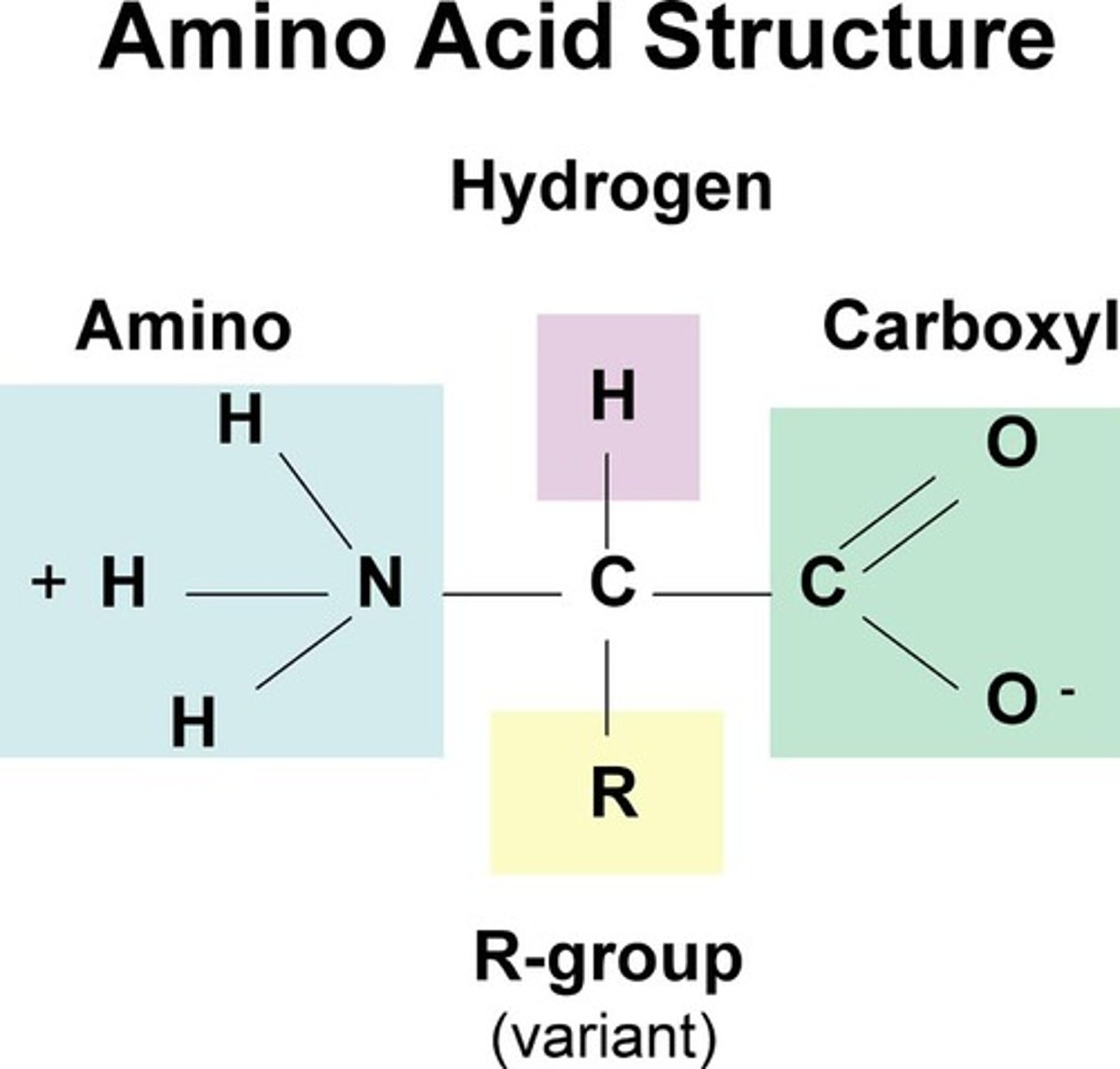
Base
Has a pH greater than 7
Salt
Formed when acids & bases react

pH scale
Measures how acidic or basic a substance is (Potential or power of Hydrogen ion)

Solution
Mixture Of Things In Two Different Phases.

A substance that produces H+ ions is a solutions is called a(n)
Arrhenius acid
Describe three properties of a solution that has a pH of 2.
sour, corrosive, ions completely dissociate, form hydronium ions, turns litmus paper red), proton donor, strong acid
Explain what an indicator, such as litmus paper, helps determine.
determines if it is an acid or base
A substance that produces hydroxide (OH-) ions in a solution is a(n)
Arrhenius base
When mixing a saltwater solution the water is the _________ and the salt is the __________.
solvent, solute
Explain what it means to have a concentrated solution.
a concentrated solution has a large amount of solute in the solvent
Phenolphthalein, litmus paper, and pH paper are all examples of ___________.
indicators
______ is the chemical reaction between an acid and a base which results in the formation of salt and water.
neutralization
What is the difference between saturated and an unsaturated solution?
saturated-contains all the solute it can hold at a given temperature while a unsaturated solution is able to dissolve more solute
Describe two ways to speed up the rate of dissolving sugar in water.
stirring, increase surface area, increasing the temperature
Bananas have a pH of 5.2. Explain what this means.
it is a weak acid
A supersaturated solution is ____________ compared to a saturated solution. (more stable, equally stable, dilute, unstable)
unstable
Water has a pH of
7-neutral
Lemons and pickles taste sour, this property indicates that the are _____________.
acids
Explain how a weak base is different from a strong base.
strong bases dissociate completely while a weak base does not, strong bases can be corrosive, strong bases can conduct more electricity than weak ones, stronger bases have higher pH reading
List three properties of bases?
slippery, bitter taste, strong bases are corrosive, many are crystalline solids in undissolved state, form hydroxide ions, proton acceptors, has a pH of above 7
Ammonia has a pH of 11.5--what does this mean?
strong base
What does the value of 7 on the pH scale indicate?
neutral
Soap is an example of a(n) __________. (acid, base, salt, ion)
base
Water is a ______ molecule.
polar
List three possible ways that phases of matter could combine to form a solution.
liquid/liquid, solid/liquid, gas/liquid, gas/gas, solid/solid
The ________ of the surface area of a solid, the faster it will dissolve.
larger
Surface area of a solid can be increased by
crushing it
Determine the type of solution you have if at 45 degrees, you add more solute and it dissolves.
unsaturated
In a polar covalent bond, what particle is shared unequally between the atoms?
electrons
Surface tension is a result of the __________ of water molecules
cohesion
Polarity of solutes and solvents determine
ability to form a solution
Like polarity in the solvent and solute forms a
solution.
Unlike polarity in the solvent and solute causes
immiscibility.
Bronsted-Lowry acid
proton donor (H+ donor)
Bronsted-Lowry base
proton acceptor (accepts H+)
On a graph of solubility for solutions the lines curve up for
solids, which are more soluble at higher temperatures.
On a graph of solubility for solutions the lines curve down for
gases, which are less soluble at higher temperatures.