Diffusion and Membrane Potential
1/33
Earn XP
Description and Tags
ch.3 pg 63-65, 77-84 Action and Graded potential ch.4 87-102
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
34 Terms
Explain equilibrium and resting membrane potentials
What is the concept of diffusion
Identify the differences between leak channel and Na+/K+ ATPase
What is diffusion?
the process of movement of molecules under a concentration gradient
Net movement in diffusion
is the overall flow of particles from a region of higher concentration to a region of lower concentration driven random molecular motion until equilibrium is reached
What factors affect the rate of net diffusion
Magnitude (of concentration gradient) - as increase concentration gradient, increase rate of diffusion
Permeability (of the membrane) - increase of permeability, increase rate of diffusion
Surface area (of the membrane) - increase surface area, increase rate of diffusion
Molecular weight (of the substance) - increase molecular weight, decrease rate of diffusion
Distance (over which diffusion takes place -thickness) - increase distance, decrease rate of diffusion
What is electrochemical gradient?
diffusion down a concentration (chemical) gradient —> high to low concentration
movement along an electrical gradient
what are neurons?
nerve cells thay specialized for electrical signaling over long distances
What is membrane potential (Vm)?
Membrane potential is a spearation of opposite charges across the plasma membrane
How does the cell create charge separation for K+ and Na+?
Equilibrium membrane potential for K+
Equilibrium membrane potential for Na+
How does the cell create charge separation?
Establishes and maintains concentration gradients for key ions (Na+, K+)
Ions diffuse through the membrane down their concentration gradients
Diffusion through the membrane results in charge separation, creating a membrane potential (electrical gradient
Net diffusion continues until the force exerted by the electrical gradient exactly balances the force exerted by the concentration gradient
This potential that would exist at this equilibrium is “equilibrium potential”
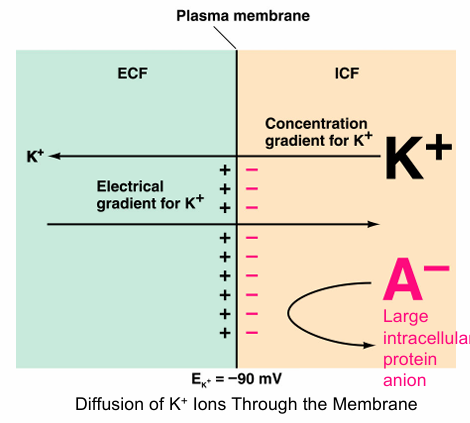
Process of Equilibrium potential for K+
K+ tends to move out of the cell
Outside of the cell becomes more positive
Electrical gradient tends to move K+ into the cell
Electrical gradient counterbalances gradient
No further net movement of K+ occurs
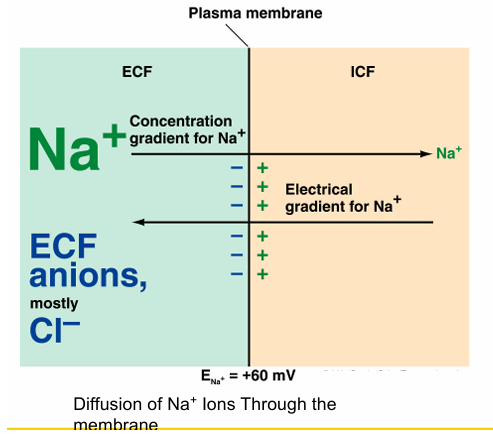
Process of Equilibrium potential for Na+
Na+ tends to move into the cell
Inside of the cell becomes more positive
Electrical gradient tends to move Na+ out the cell
Electrical gradient counterbalances concentration gradient
No further net movement of Na+ occurs
Nerst Equation
equation describing the equilibrium potential for a particular ion (i)
where R is the gas constant, T is the temperature in degrees Kelvin, z is the valence of the ionic species, & F is the Faraday constant
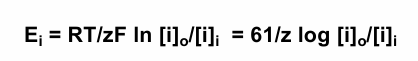
Resting Membrane Potential
K+ high in the ICF and Na+ high in the ECF
K+ drives equilibrium potential for K+
N+ drives equilibrium potential for Na+
How do K+ and Na+ penetrate the cell membrane?
Leak channels - permit ions to flow down concentration gradients
Why is Na+ higher outside of the cell and K+ higher inside the cell?
Na/K ATP ase - establishes and maintains concentradtion gradients
Concentration Gradient
Na+/K+ ATPase establishes the unequal distribution of Na+ and K+ ions inside and outside of the cell
Action Graded Potential
Understand the process involved with depolarization and hypolarization
Describe the differences between voltage-gated Na+ and K+ channels
Understand the roles of refractory periods
Describe the differences between action potential and graded potential
Depolarization
change in membrane polarization to more positive values than resting potential
Hyperpolarization
change in membrane polarization to more negative values than resting membrane potential
Action potential
Brief all-or-nothing reversal in membrane, potentialm (spike), lasting on the order of 1 millisecond that is brought about by rapid changes in membrane permeability, to Na+ and K+ ions