GCSE AQA chemistry- Energy changes
1/15
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
16 Terms
What is an exothermic reaction?
A reaction that causes a rise in temperature to surroundings- it releases energy
What is an endothermic reaction?
A reaction that causes a decrease in temperature in the surroundings-it takes in energy
If a reversible reaction if exothermic in one direction what will it be in the reverse direction?
Endothermic
What must happen for bonds to be broken in a chemical reaction?
Reacting particles must collide with sufficient energy
What happens in a chemical reaction?
Atoms are rearranged as old bonds are broken and new bonds are formed
What is the activation energy?
The minimum amount of energy that particles must have to take place
What is the energy level diagram (reaction profile) for an exothermic reaction?
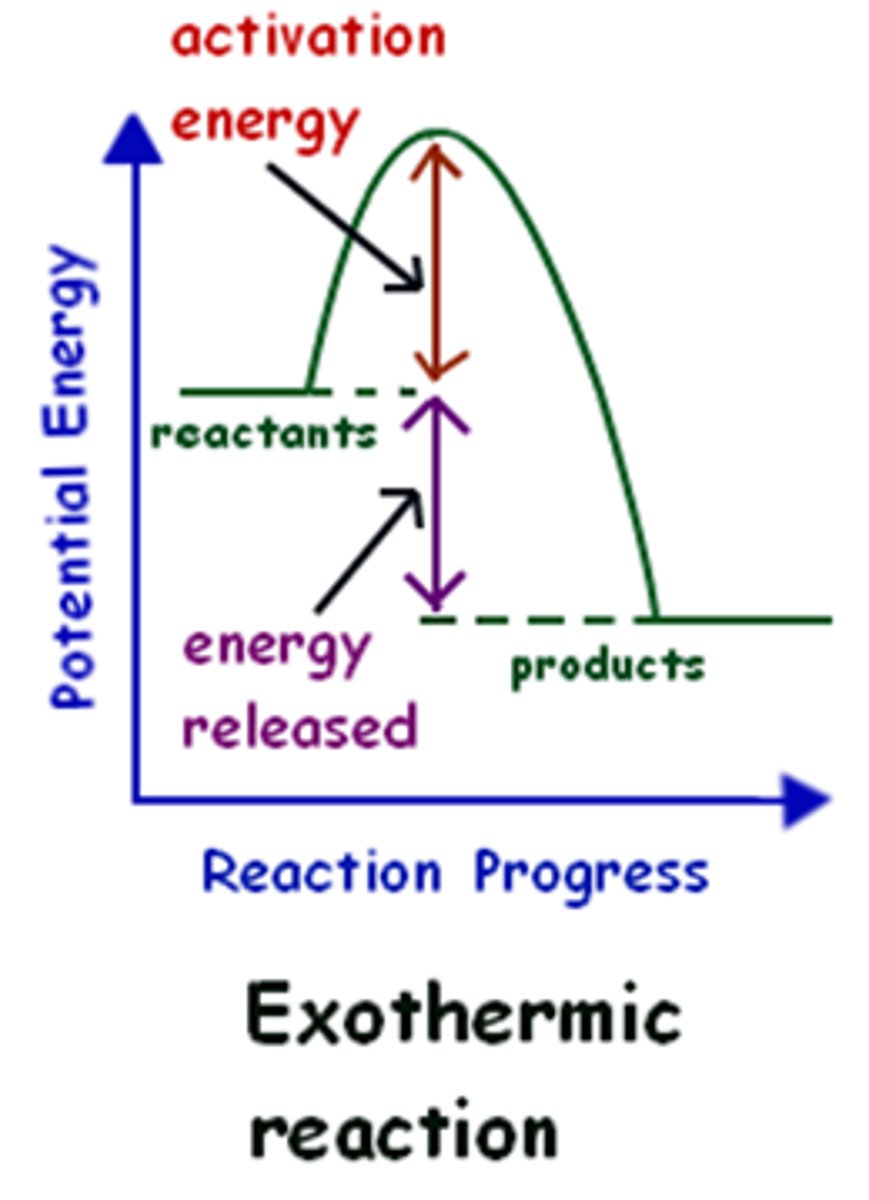
Why does an exothermic reaction graph have that shape?
Because energy is given out to the surroundings and the products have less energy than the reactants
What is the energy level diagram (reaction profile) for an endothermic reaction?
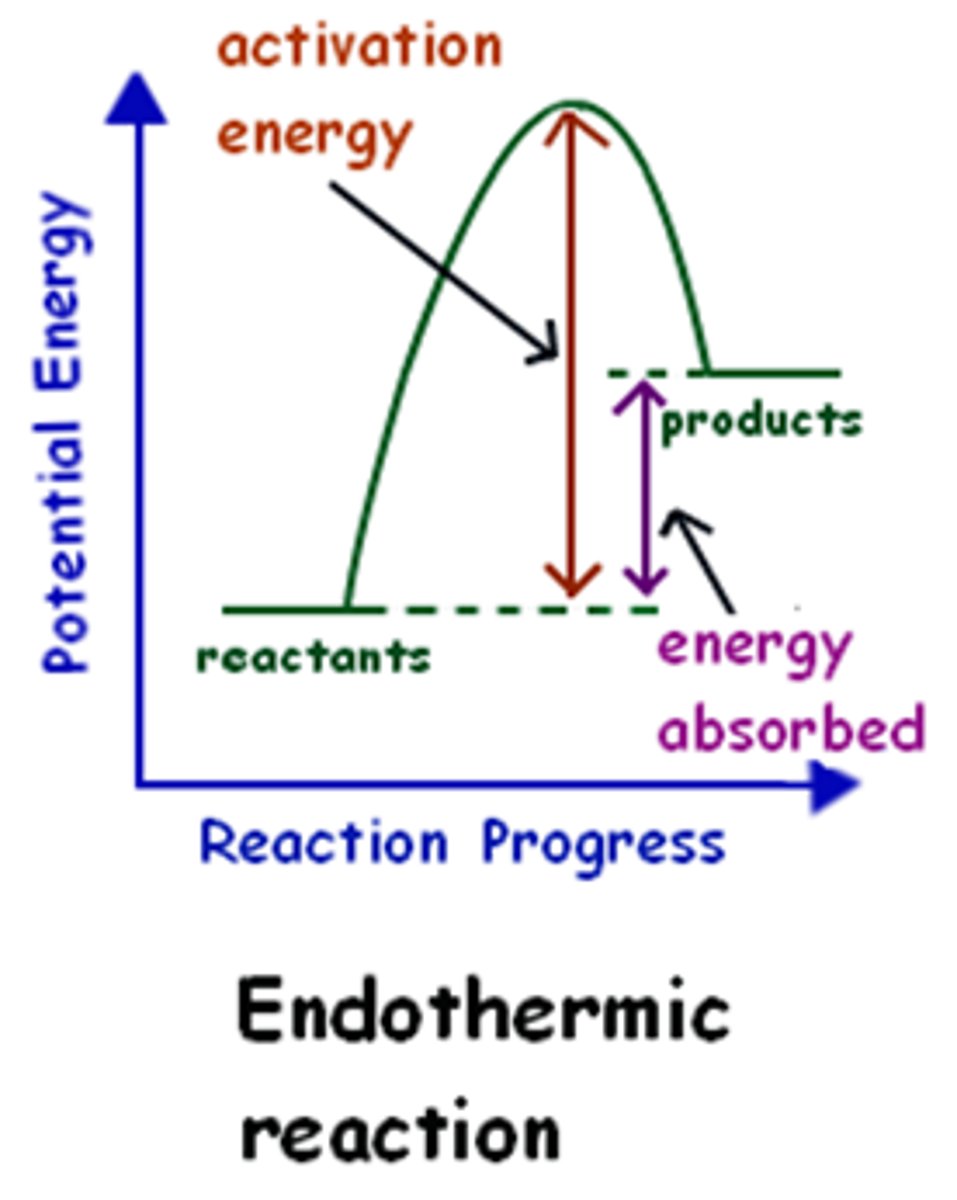
Why does an endothermic reaction graph has that shape?
Because energy is taken in from the surroundings and the products have more energy than the surroundings
Is breaking a bond in a chemical reaction an endothermic or exothermic process?
Endothermic
Is forming a bond in a chemical reaction an endothermic or exothermic process?
Exothermic
What determines if a reaction is exothermic?
If the energy released when bonds are formed is greater than the energy needed to break bonds
What determines if a reaction is endothermic?
If the energy needed to break bonds is more than the energy released as bonds formed
How can we measure the amount of energy produced in a chemical reaction in solution?
By mixing the reactants in an insulated container. This enables heat that would be lost to surrounding to be measured.
What sort of reactions is the insulated reaction method suitable for measuring energy produced?
Neutralisation reactions and reaction involving solids