BIO CHEM CH 1
1/34
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
35 Terms
What are the distinctive properties of living systems
organisms are complication and highly organized into organelles that differentiated
biological structures serve functional purposes
living systems are actively engaged in energy transformations
living systems have a remarkable capacity for self replication via DNA replication
living systems depend on…
energy transformation
ultimate source of energy
light (solar light flows to photosynthetic organisms and then herbivores and carnivores)
ATP
most energized molecule; broken down to produce energy for different pathways; nitrogenous base, ribose sugar, and 3 phosphate group
NADPH (nicotinamide adenine dinucleotide phosphate) / NADH (nicotinamide adenine dinucleotide)
electron carriers used for oxidation reactions for different pathways; energy rich molecules; one used for synthesis and the other for breaking down
what kinds of molecules are biomolecule
macromolecules (protein, nucleotides, fatty acids, carbohydrates)
double stranded DNA
nucleic acids; is unwound for replication
four main elements involved in formation of macromolecules / in living systems; why?
H, C, N, and O; can form strong covalent bonds by e- pair sharing
what 2 elements most makeup the human body
H and O
what property unites H, O, C, and N and renders these atoms so appropriate to the chemistry of life?
their ability to form covalent bonds by sharing electron pairs
C-C bonds are the main building block to form..
complex structures of fatty acids and cholestorol molecules
long hydrocarbon chains cause the molecule to be..
very non-polar / very hydrophobic (fatty acids / oils can not be dissolved in oil due to their long hydrocarbon chains)
what is the structural organization of complex biomolecules
inorganic precursor → metabolites → building blocks → macromolecules → supramolecular complexes → organelles
four classes of macromolecules
carbohydrates, nucleic acid, proteins, lipids (review monomer and polymers in the image)
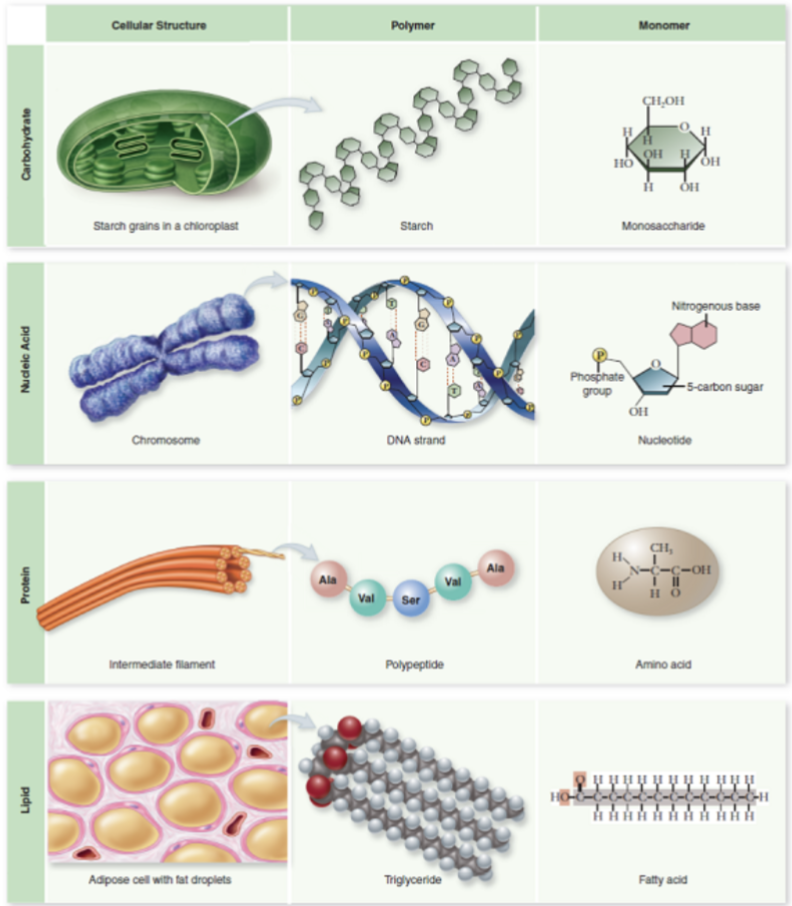
how do the properties of biomolecules reflect their fitness to the living condition
amino acids are the building block of…
proteins
when two amino acids bind and loose a water a —- is formed
peptide bond
polysaccharides are formed when — are joined together. what type of bond is formed
sugar; glycosidic bond
nucleic acids form what type of molecule? what bond is made?
DNA and RNA; diester bonds
example of covalent bonds
diester bonds, peptide bonds, glycosidic bonds
weak forces
van der waals forces, hydrogen bonds, ionic interaction, hydrophobic interaction
rank the weak forces from the weakest to strongest
van der waal < hydrogen bonds < ionic interaction < hydrophobic interaction
van der waals
occurs due to closeness and angles of bonds (0.4 radius (nm)) (the smaller radius = stronger); easily broken down / weakest of all the weak bonds
hydrogen bonding
occurs between H, N, O, S; will have an acceptor and a donor
ionic interaction
between negative and positive molecules; contribute to the stability of proteins
hydrophobic interaction
between hydrophobic and hydrophilic molecule (i.e. fatty acids)
biomolecular recognition is mediated by…
weak chemical forces; i.e. when an antibody is connecting to an antigen to kill a virus due to the weak forces between them; i.e. the connection of a substrate to an enzyme’s active site
weak forces restrict organisms to a…
narrow range of environmental condition;
what is meant by the idea that weak forces restrict organisms to a narrow range of environmental conditions
weak forces are unable to withstand change; i.e. when a protein is denatured ( due to temp change, PH change, etc) the weak forces are broken down and only covalent, peptide bonds remain
Glycolysis occurs when there is no O2 in cytosol (anaerobic) → will provide enough energy for other pathways by breaking glucose into pyruvate; for glycolysis to occur, the enzyme used in each step use weak forces to connect
what is the organization and structure of cells
prokaryotic (single membrane, no nucleus / organelle) and eukaryotic cells (larger / organelles / complicated / nucleus)
prokaryotic cell
cell wall give support, shape, and protection
cell membrane acts as barrier that controls the entry of most substances into and exiting the cell
nuclear area has the genetic information
ribosomes are the site of protein synthesis
storage granules are used for metabolic fuel
cytosol is the site of intermediate metabolism
eukaryotic cell
ECM is for cell recognition and communication
cell membrane is selectively permeable
nucleus holds DNA
chloroplasts (plant) are the site of photosynthesis
mitochondria is where ATP production occurs
vacuoles transport and store nutrients
ER - protein synthesis (rough/ribosome), lipid synthesis (smooth)
golgi - modifies, sorts, and packages lipids
lysosomes / peroxisomes - break down waste
cytoskeleton -structural support
what are viruses
enter host cell for survival; have outer protein-coating
viral protein coat / spike protein
recognizes same protein structure in the host cell to enter the host; spike protein binds to receptor of host cell surface that allows the virus to enter the host cell and replicate
peptidase enzyme / protease enzyme
responsible for breaking down protein coats of virus to release the DNA molecule of the virus into the host cell (inhibition of protease / peptidase can prevent infection of the virus)