Final Complete
1/335
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
336 Terms
How do scientists use ice cores to determine past temperatures?
If the ice has a higher concentration of the isotope 18O it was formed in a cooler climate; if it has a lower concentration of 18O it was formed in a warmer climate. This is similar to the use of foraminifera fossils that was described in chapter 2, box 2.1. We can also measure the concentration of greenhouse gases trapped in air bubbles in the ice.
What is albedo? Give an example of an object with (a) high albedo and (b) low albedo.
Albedo is the percentage of radiation reflected from a surface. Snow has a high albedo; soot has a low albedo
In what way do greenhouse gases warm the planet?
Greenhouse gases are transparent to sunlight but they absorb longer-wavelength radiation coming from the ground. So the heat is trapped in the Earth’s atmosphere
Which of the following is the correct order of the layers of the atmosphere from the ground up?
a. stratosphere, mesosphere, troposphere, thermosphere
b. thermosphere, troposphere, stratosphere, mesosphere
c. troposphere, stratosphere, mesosphere, thermosphere
d. stratosphere, troposphere, mesosphere, thermosphere
c. troposphere, stratosphere, mesosphere, thermosphere
What would the temperature of the Earth be if there were no atmosphere?
15°C
545°C
0°C
−19°C
−19°C
Which of the following is a greenhouse gas?
nitrogen
oxygen
methane
argon
methane
The Maunder Minimum or “Little Ice Age” of the late seventeenth century is believed to have been caused by
a large volcanic eruption.
a period of very low sunspot activity.
a decrease in the concentration of greenhouse gases in the atmosphere.
greater global albedo due to heavy cloud cover in the Northern Hemisphere.
a period of very low sunspot activity.
The “Year Without a Summer” in the 1800s was caused by
a large volcanic eruption.
a period of very low sunspot activity.
a decrease in the concentration of greenhouse gases in the atmosphere.
greater global albedo due to heavy cloud cover in the Northern Hemisphere.
a large volcanic eruption.
Which of the following energy sources does not directly produce carbon dioxide?
coal
nuclear power
natural gas
petroleum
nuclear power
The amount of water vapor in the atmosphere ranges from 0% to 4%. You experience this as relative humidity, which can range from 0% to 100%. What is the difference between the absolute amount of water in the atmosphere and relative humidity?
The relative humidity is the water vapor in the atmosphere as a percentage of the amount that would cause rain or snow. So if the relative humidity is 100% then it’s going to start raining. The relative humidity increases as the temperature goes down, even if the actual amount of water vapor stays the same. This is because cold air holds less water than warm air.
Why is ozone at the surface considered harmful and ozone in the stratosphere considered beneficial?
Ozone is a more powerful oxidant than ordinary oxygen. If you inhale it can cause coughing and lung congestion. On the other hand, ozone in the upper atmosphere protects us from ultraviolet radiation
Contrast the geologic conditions responsible for the formation of coal, oil, and natural gas.
Coal is formed from decaying plant matter, mostly from coastal swamps. The plant absorbs carbon dioxide and solar energy. The energy is stored in hydrocarbons. When the plant dies its energy is usually released back into the atmosphere, but if it falls into stagnant, oxygen-depleted water and becomes buried by sediment then the energy remains with the plant material. As more sediment accumulates above the plant material the pressure increases and it gradually turns into coal. There are several stages in the formation of coal, as illustrated in table 22.1, page 536. Peat, lignite, subbituminous coal, bituminous coal and anthracite have progressively higher concentrations of energy.
Oil is formed under somewhat different conditions. It originates as marine lifeforms in shallow seawater or tropical lagoons. When the lifeforms die they usually oxidize and release their energy, but if they are rapidly buried in sand and mud then they turn into a hydrocarbon-rich sediment called “sapropel”. When the sapropel is buried it heats up, at a rate of 25°C for every thousand meters. At 50°C to 100°C it forms oil. Between 100°C and 200°C it forms natural gas. Above 200°C the hydrocarbons break down completely
Which is not a type of coal?
lignite
bituminous
sulfite
anthracite
sulfite
Coal forms
by crystal settling.
through hydrothermal processes.
by compaction of plant material.
on the ocean floor.
by compaction of plant material.
If driven 12,000 miles per year, how many more gallons of gasoline per year does a sport utility vehicle or pickup truck rated at 12 miles per gallon use than a minicompact car rated at 52 mpg? Over five years, how much more does it cost to buy gasoline at $4 per gallon for the low-mileage car? At $8 per gallon (the price in many European countries)?
Utility vehicle: 12,000 miles per year divided by 12 miles per gallon is 1000 gallons per year.
Minicompact car: 12,000 miles per year divided by 52 miles per gallon is 231 gallons per year.
The difference is 769 gallons per year.
At $4 per gallon this is 769×4 = $3076 per year, or $15,400 over five years.
At $8 per gallon it’s twice this, or $30,800
What is the approximate shape of the solar system?
The solar system is approximately disc-shaped. The planet whose orbit has the greatest inclination is Mercury, which has an orbital inclination of about 7°. Asteroid orbits are also in the same plane, but comets can have orbits that are inclined at large angles. Table 23.2 on p. 570 has the orbital properties of all the planets.
Why is the surface of Venus hotter than the surface of Mercury, despite Mercury being closer to the Sun?
Venus has a much thicker atmosphere than Mercury, which creates a large greenhouse effect, as described in chapter 21. On Mercury there is almost no atmosphere, so the side that faces the sun is very hot (about 430°C) and the side away from the sun is very cold (about -173°). Venus’ surface temperature is about 480°C
Why is the Earth much less cratered than the Moon?
As explained on page 574, most of the craters on Earth have been obliterated by weathering, erosion, volcanism and plate tectonics. There are a few places where we may be able to see the remains of ancient craters. For example, the southeast part of Hudson Bay forms a round structure that was once believed to have been formed by a meteor impact:
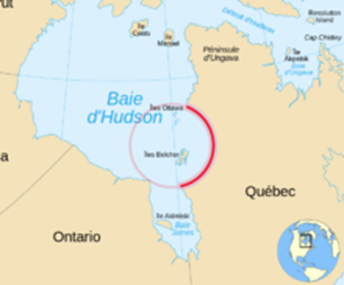
Nastapoka arc
(from Wikipedia)
Detailed studies have shown no evidence of shatter cones, radial fractures or shock metamorphism, so most geologists now believe that this circular arc was formed by tectonic activity. However, some believe that this tectonic activity conformed to an earlier crater that dates back to the Archean eon. Likewise the Silurian dolomite basin, a round structure that underlies lower Michigan and parts of surrounding states, including southeastern Wisconsin, was once believed by some to be the result of an ancient asteroid impact, but no evidence has been found to support this. Craters that have been positively identified on Earth include Meteor Crater in Arizona, which was formed about 50,000 years ago, the Sudbury astrobleme, which goes back 1.8 billion years, the Vredefort crater in South Africa, which is 2 billion years old, and the 2.4 billion year old Suavjärvi crater in Russia.
The craters of the Moon that we can see through a telescope were formed over four billion years ago, when bombardment by large meteors was much more frequent. The “maria” or “seas” are basaltic plains that were formed after the main bombardment ended. That’s why there are relatively few craters within the maria.
The planet Mars has a relatively large number of craters, which indicates that geologic activity on Mars ceased billions of years ago.
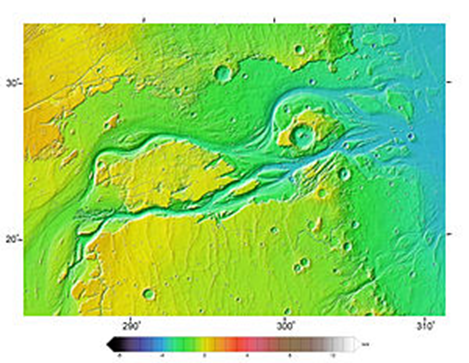
Why do scientists believe there was once liquid water on the surface of Mars?
There are channels on Mars that look as though they were formed by flowing water:
Kasei Valles, seen in MOLA elevation data. Flow was from bottom left to right. North is up. Image is approx. 1,600 km (990 mi) across. The channel system extends another 1,200 km (750 mi) south of this image to Echus Chasma.
But the main evidence consists of layered rocks recovered by Mars rovers. These resemble the sedimentary rock layers that we have here on Earth. Today there isn’t any liquid water on the Martian surface because the atmospheric pressure is so low that it evaporates instantly.
Why do Mercury and the Moon lack an atmosphere?
They formed after all the gas had been used up.
They are so cold that all their gases have frozen into deposits below their surface.
They formed before the solar nebula had captured any gas.
They are so small that their gravity is too weak to retain an atmosphere.
They are so small that their gravity is too weak to retain an atmosphere.
Why would it be difficult to land a spacecraft on Jupiter?
Jupiter has no solid surface.
Jupiter’s immense gravity would squash it.
Jupiter’s intense magnetic field would destroy it.
The clouds are so thick it would be hard to navigate to a safe spot.
Jupiter has no solid surface.
Mercury’s inclined and elliptical orbit is believed to be the result of
its proximity to the Sun.
collision with an enormous planetesimal.
the gravitational pull of Jupiter.
its very slow rotation.
collision with an enormous planetesimal.
An astronomical unit (AU) is equivalent to approximately
50 million miles.
150 million kilometers.
100 billion kilometers.
250 million miles.
150 million kilometers.
Why do mafic magmas tend to reach the surface much more often than felsic magmas?
Felsic magma (which is higher in silica content) is more viscous than mafic magma.

How would you distinguish, on the basis of minerals present, among granite, gabbro, and diorite
Granite has a wide range of compositions, but it usually contains quartz or potassium feldspar, in addition to other minerals. Diorite and gabbro both contain ferromagnesians and plagioclase feldspar, but diorite has more amphibole and gabbro has more pyroxene.
Why is a higher temperature required to form magma at the oceanic ridges than in the continental crust?
According to the section “Explaining Igneous Activity by Plate Tectonics”, subsection “Igneous Processes at Convergent Boundaries”, paragraph titled “The Origin of Granite”, the melting point of silicic rocks is lower than that of mafic rock, especially if water is present. (If water is present it’s called “flux melting”.)
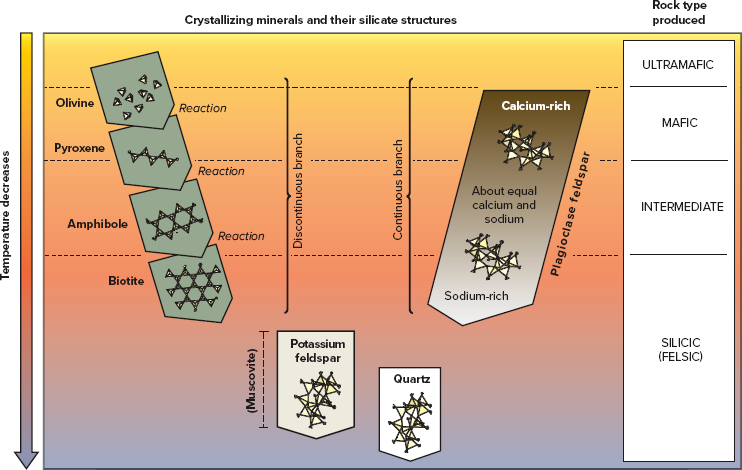
Describe the differences between the continuous and the discontinuous branches of Bowen’s reaction series.
The discontinuous branch is on the left; the continuous branch is on the right. Quartz and potassium feldspar belong to both branches.
The major difference between intrusive igneous rocks and extrusive igneous rocks is
where they solidify.
chemical composition.
type of minerals.
all of the preceding
where they solidify
Which is not an intrusive igneous rock?
gabbro
diorite
rhyolite
peridotite
rhyolite
Flux melting occurs when
water is added to the asthenosphere.
a mantle plume increases the temperature of the asthenosphere.
mantle material undergoes depression.
water is added to the asthenosphere
The continuous branch of Bowen’s reaction series contains the mineral
pyroxene.
plagioclase.
amphibole.
quartz
plagioclase
The discontinuous branch of Bowen’s reaction series contains the mineral
pyroxene.
amphibole.
olivine.
all of the above
all of the above
By definition, stocks differ from batholiths in
shape.
size.
chemical composition.
age.
size
A discordant shallow intrusive structure is called a
stock.
dike.
sill.
laccolith.
dike
The most common igneous rock of the continents is
basalt.
granite.
rhyolite.
peridotite.
granite
Mafic magma is generated at divergent boundaries because of
decompression melting
What would happen, according to Bowen’s reaction series, under the following circumstances: olivine crystals form and only the surface of each crystal reacts with the melt to form a coating of pyroxene that prevents the interior of olivine from reacting with the melt?
This is basically what happened with the mineral sample that I showed you in class: a layer of olivine remains at the bottom, and then the other layers form above it. Most of the sample consists of quartz, which is the last mineral formed.

Examine the photo of the Canadian Rockies on the first page of this chapter. Which of the four Earth systems can you see? How are they interacting with each other?
Biosphere, geosphere, hydrosphere, atmosphere. All four Earth systems are visible in this photograph. The clouds are part of the atmosphere; the glacier and lake are part of the hydrosphere; the grass and trees are part of the biosphere; and the rocks are part of the geosphere.
Draw a cross section of the Earth and label each of the compositional layers and mechanical layers.
Igneous rock, metamorphic rocks, Sedimentary rocks
What are the relationships among the mantle, the crust, the asthenosphere, and the lithosphere?
The crust is the very top layer with oceanic and continental crust. The mantle is the second layer. The lithosphere is the mantle and the crust. While the asthenosphere is below the lithospher
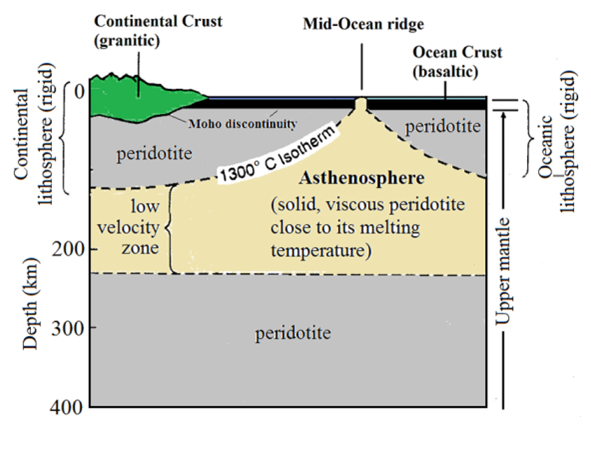
What tectonic plate are you currently on? Where is the nearest plate boundary, and what kind of boundary is it?
We are on the North American Plate. The nearest plate boundary is the Mid-Atlantic Ridge. It is a divergent boundary
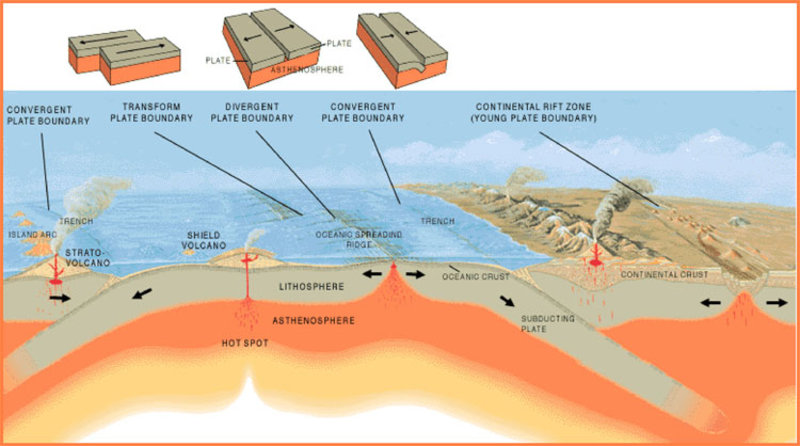
Draw a sketch of each of the major types of plate boundaries. Show the direction of plate motion and label important features.
Convergent boundary, transform boundary, divergent boundary
Explain why prehistoric cave dwellers never saw a dinosaur.
dinosaurs became extinct 70 million years ago, and hominids didn’t appear until 5 million years ago.
Plate tectonics is a result of Earth’s internal heat engine, powered by (choose all that apply)
the magnetic field.
the Sun.
gravity.
heat flowing from Earth’s interior outward
gravity and heat flowing from the earths interior outward
A typical rate of plate motion is
3–4 meters per year.
1 kilometer per year.
1–10 centimeters per year.
1,000 kilometers per year.
1-10 centimeter per year
Oceanic and continental crust differ in
composition.
density.
thickness.
all of the preceding.
all of the preceding.
The lithosphere is
the same as the crust.
the layer beneath the crust.
the crust and uppermost mantle.
only part of the mantle.
the crust and uppermost mantle
Why are some parts of the lower mantle hotter than other parts?
Because there are regions that have a greater concentration of radioactive materials, and these generate a greater amount of heat
According to plate tectonic theory, where are crustal rocks created? Why doesn’t Earth keep getting larger if rock is continually created?
Crustal rocks are created at the spreading centers (divergent plate boundaries) but they melt back into the mantle ant convergent plate boundaries.
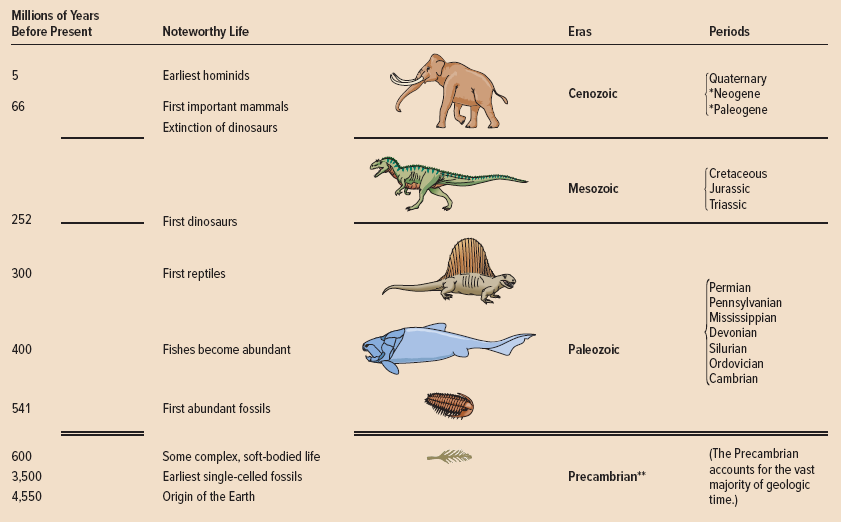
What percentage of geologic time is accounted for by the last century?
(100 years)/(4.5 billion years)×(100%) = 2.2×10–6 %
What are some of the technical difficulties you would expect to encounter if you tried to drill a hole to the center of the Earth?
Extreme pressure and Extremely high temperature
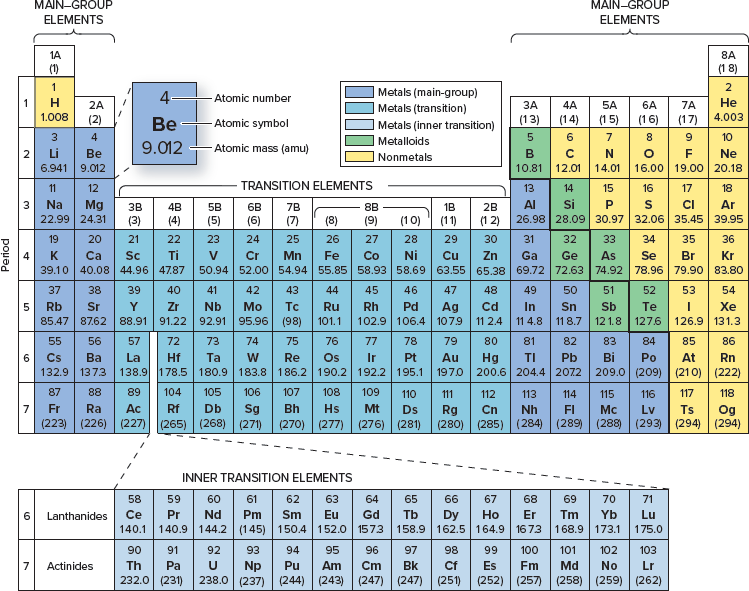
Which element has an atomic number of 24?
Chromium
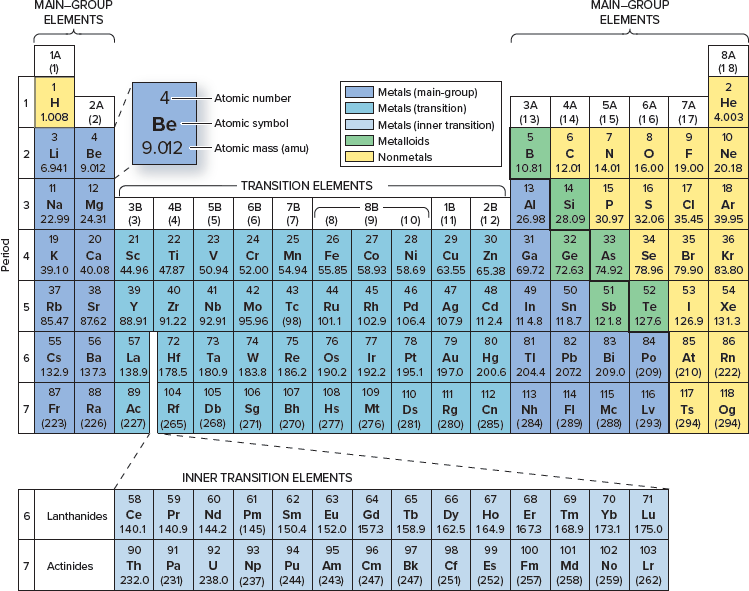
How many neutrons does it have?
28
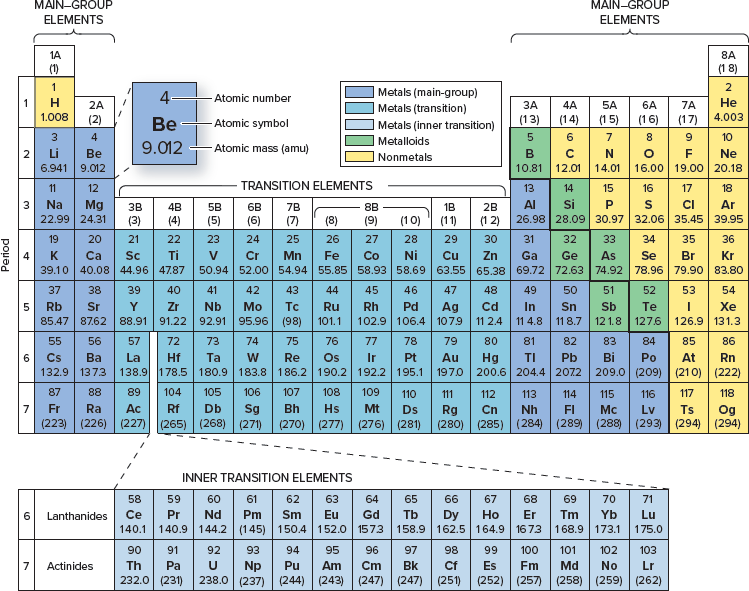
How many electrons does a neutral atom of this element have?
24
The mineral fluorite (CaF2) is composed of calcium (Ca2+) and fluorine (F−). What kind of bonding holds them together? Explain how you know this.
It’s an ionic bond because calcium has only two electrons in its outermost shell and fluorine has seven. So when calcium and fluorine bond together the outermost calcium electrons spend most of their time going around the fluorine atoms, creating a positive ion and a negative ion.
Using triangles to represent silica tetrahedra, start with a single triangle (to represent isolated silicate structure) and, by drawing more triangles, build on the triangle to show a single-chain silicate structure. By adding more triangles, convert that to a double-chain structure. Turn your double-chain structure into a sheet silicate structure.
Isolated: single triangle
Single chain: one row
Double chain: two linked rows
Sheet: many rows forming a flat layer
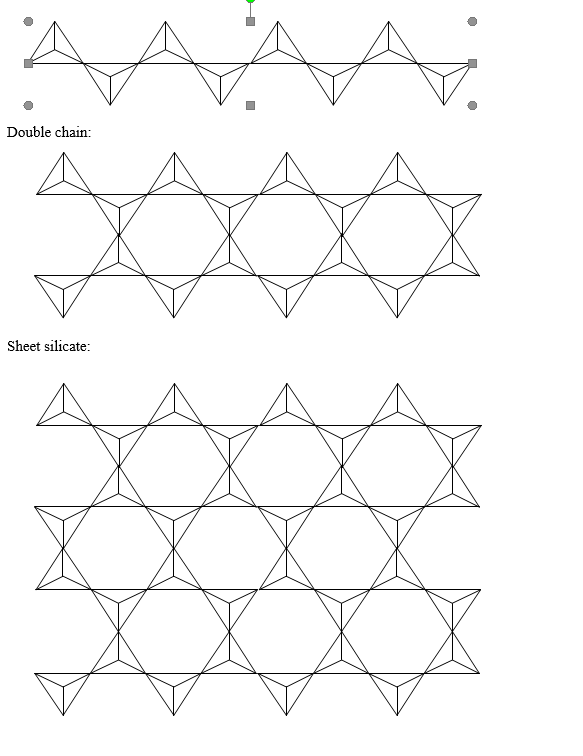
How do the crystal structures of pyroxenes and amphiboles differ from one another? Which physical property is used to distinguish between them?
Pyroxenes and amphiboles differ in how their silicate tetrahedra are linked, and this structural difference directly controls their cleavage,
Pyroxenes have a single-chain silicate structure. Each SiO₄ tetrahedron shares two oxygen atoms, forming long, straight chains. have two directions of cleavage that intersect at nearly 90° (about 87° and 93°).
Amphiboles have a double-chain silicate structure. This produces two cleavage directions that intersect at oblique angles, approximately 56° and 124°.
Write out the definitions for the terms element, mineral, and rock; then for each of the following substances, determine whether it is an element, a mineral, or a rock: Iron
Element: A pure substance made of only one type of atom. An element cannot be broken down into simpler substances by chemical means.
Mineral: A naturally occurring, inorganic solid with a definite chemical composition and an ordered internal (crystalline) structure.
Rock: A naturally occurring solid made up of one or more minerals (or mineraloids). Rocks do not have a fixed chemical composition.
Classification: its an element
Write out the definitions for the terms element, mineral, and rock; then for each of the following substances, determine whether it is an element, a mineral, or a rock: Quartz
Classification: is a mineral.
Write out the definitions for the terms element, mineral, and rock; then for each of the following substances, determine whether it is an element, a mineral, or a rock: Carbon
Classification: is an element.
Write out the definitions for the terms element, mineral, and rock; then for each of the following substances, determine whether it is an element, a mineral, or a rock: Diamond
Classification: is a mineral
Write out the definitions for the terms element, mineral, and rock; then for each of the following substances, determine whether it is an element, a mineral, or a rock: Obsidian
Classification: is a rock.
Write out the definitions for the terms element, mineral, and rock; then for each of the following substances, determine whether it is an element, a mineral, or a rock: Calcite
Classification: is a mineral.
Write out the definitions for the terms element, mineral, and rock; then for each of the following substances, determine whether it is an element, a mineral, or a rock: calcium
Classification: is an element.
Write out the definitions for the terms element, mineral, and rock; then for each of the following substances, determine whether it is an element, a mineral, or a rock: Limestone
Classification: is a rock.
Write out the definitions for the terms element, mineral, and rock; then for each of the following substances, determine whether it is an element, a mineral, or a rock: Granite
Classification: is a rock
How would you distinguish between the following pairs of minerals on the basis of physical properties? olivine/pyroxene
Olivine has poor or no cleavage
Pyroxene has two distinct cleavage directions that intersect at nearly 90°
How would you distinguish between the following pairs of minerals on the basis of physical properties? calcite/quartz
Calcite has three directions of cleavage that do not meet at right angles.
Quartz has no cleavage
How would you distinguish between the following pairs of minerals on the basis of physical properties? mica/halite
Mica has perfect cleavage in one direction, allowing it to split into very thin, flexible sheets.
Halite has three directions of cleavage that intersect at 90 degrees,
How would you distinguish between the following pairs of minerals on the basis of physical properties? amphibole/hematite
Amphibole typically appears dark green to black and has two directions of cleavage
Hematite can appear distinctive reddish-brown streak.
Name the nonsilicate mineral groups and describe their defining chemical characteristics
Carbonates contain the carbonate ion (CO₃²⁻).
Sulfates contain the sulfate ion (SO₄²⁻).
Sulfides contain sulfur (S²⁻) but no oxygen.
Oxides consist of oxygen bonded to a metal, but the oxygen is not bonded to silicon, carbon, or sulfur.
chlorides contain Cl but no O
Native elements consist of only one element.
A substance that cannot be broken down into other substances by ordinary chemical methods is a(n)
crystal.
element.
molecule.
acid.
element
Which of these is not part of the definition of a mineral?
Organic
Crystalline
Specific chemical composition
Naturally occurring
Organic
The subatomic particle that contributes a single negative electrical charge is the
proton.
neutron.
electron.
electron
Atoms of an element containing different numbers of neutrons but the same number of protons are called
ions.
covalent.
isotopes.
neutral.
isotopes
Atoms with either a positive or a negative charge are called
compounds.
ions.
elements.
isotopes.
ions
The bonding between Cl and Na in halite is
ionic.
covalent.
metallic
Ionic
Which is not true of a single silica tetrahedron?
The atoms of the tetrahedron are strongly bonded together.
It has a net negative charge.
The formula is SiO4.
It has four silicon atoms.
four silicon atom
In a single chain silicate, how many of the oxygen atoms in each silica tetrahedron are shared with neighboring silica tetrahedra?
None
One
Two
Three
Four
Two
Which of these common minerals is not a silicate?
Quartz
Feldspar
Gypsum
Mica
Gypsum
The ability of a mineral to break along preferred directions is called
fracture.
crystal form.
hardness.
cleavage.
cleavage
Crystalline substances are always
ionically bonded.
minerals.
made of repeating patterns of atoms.
made of glass.
made of repeating patterns of atoms
Which of these minerals commonly forms as an evaporite (precipitate from evaporating water)?
Quartz
Hematite
Halite
Feldspar
Halite
How does oxygen in the atmosphere differ from oxygen in rocks and minerals?
Oxygen atoms in the atmosphere are bonded with other oxygen atoms (O2 or O3) or with carbon atoms (CO2).
What happens to the atoms in water when it freezes? Is ice a mineral? Is a glacier a rock?
The hydrogen atoms in a water molecule have a slight positive charge, while the oxygen atom has a slight negative charge. When water freezes the molecules bond together in a hexagonal lattice, with the hydrogen atoms of each molecule being attracted to the oxygen atoms of neighboring molecules. Ice is a mineral and a glacier is a rock.
How would you expect the appearance of a rock high in iron and magnesium to differ from a rock with very little iron and magnesium?
A rock high in iron and magnesium will be darker in color. In class I showed you a sample of biotite, which is a type of mica that is high in iron and magnesium, and a sample of muscovite, which is not high in iron and magnesium. These are both varieties of mica, but the biotite is darker in color.
What might explain the remarkable alignment of the Cascade volcanoes?
The volcanoes are the result of the collision of the Juan de Fuca plate with the North American plate. The lava erupts when the subducted plate reaches a certain depth
Why are volcanic eruptions at convergent plate boundaries typically more explosive than those at divergent plate boundaries?
Convergent plate boundaries give rise to felsic volcanoes, which have viscous lava, while divergent boundaries produce mafic lava
Why are there no active volcanoes in the eastern parts of the United States and Canada?
There are no active volcanoes in the eastern United States because the nearest plate boundary is thousands of miles away and there are no “hot spots”, or mantle plumes, nearby.
What do pillow lavas indicate about the environment of volcanism?
eruption took place underwater.

Consider the eruption of Mount Merapi, described at the beginning of this chapter and shown in figure 4.1. What kind of volcano is Merapi? What evidence did you use to determine this? Based upon your answer, what do you think the plate-tectonic setting is of Mount Merapi? What composition of lava do you think it mostly erupts?
is a composite volcano, as described in section “Types of Volcanoes”, subsection “Composite Volcanoes”. We know this because it produced pyroclastic flows. Most composite volcanoes are located near convergent plate boundaries, where one plate dives underneath the other. And in fact its near the boundary between the Eurasian plate and the Indian-Australian plate. Composite volcanoes erupt mostly felsic lava, with a high proportion of silicates.
Which of the following is an example of a shield volcano?
Mount St. Helens, Washington State
Mount Merapi, Indonesia
Mauna Loa, Hawaii
Cerro Negro, Nicaragua
Mauna Loa, Hawaii
The gas most commonly released during a volcanic eruption is
water vapor.
carbon dioxide.
sulfur dioxide.
hydrogen sulfide.
oxygen.
water vapor
_____ is a rock composed of frothy volcanic glass
Obsidian
Basalt
Tuff
Pumice
Volcanic breccia
Pumice
A lava flow with a ropy or billowy surface is called
pahoehoe
a′a
pillow lava
lahar
lava tube
pahoehoe
Which of these is not a type of pyroclastic material?
ash
dust
lapilli
a′a
bomb
a’a
Which of these is not a major type of volcano?
shield
cinder cone
composite
stratovolcano
spatter cone
spatter cone
An example of a composite volcano is
Mount Rainier.
Fujiyama.
Mount Vesuvius.
all of the preceding.
all of the preceding
Which volcano is not usually made of basalt?
shield
composite cone
spatter cone
cinder cone
composite cone
What factors control whether a rock behaves as a brittle material or a ductile material?
According to figure 15.4, all rocks behave elastically when the stress is relatively low. But when the stress exceeds a certain level, some rocks exhibit brittle behavior, which means they break, and others exhibit ductile behavior, which means they bend. The difference between the two depends on the type of rock and also on the temperature and pressure, as explained in chapter 7, section “Metamorphism”, paragraph 8