GEOL 316: Environmental Geochemistry Midterm
1/125
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
126 Terms
Course Theme
Asking the question “Is Lake Matoaka clean?” via many instrumental analyses
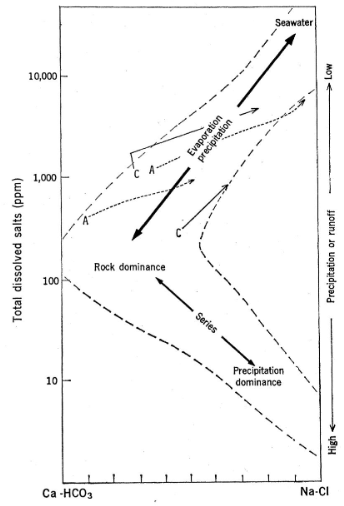
Three Major Controls on Water Chemistry
Rock-dominated
Precipitation-dominated
Evaporation-dominated
What is Lake Matoaka defined by?
A ROCK-DOMINATED system due to the weathering and dissolution of fossil mollusk shells in the sediments in Williamsburg (contributes Ca2+ and HCO3-; LARGE dissolved load of these ions > suspended load). Chemistry of the system = dependent on the materials eroding into the water body. Also ~220 mg/L (ppm), which plots in rock-dominated space.
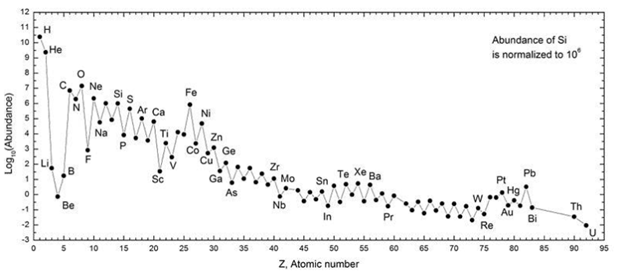
What are the main drivers for the diversity of elements in our universe?
The Big Bang + Supernova/Nebula Theory — explains 1) why certain elements are so rare, 2) why Earth’s composition is dominated by specific elements, and 3) why the universe is so high in C/N/He (but not Earth)
Relative Abundances of Elements in our Solar System
DECREASES with increasing atomic number
ALTERNATIONS between even/odd atomic numbers w/ even proton numbers = MORE abundant (because more stable electrically)
Abundance drastically DROPS OFF after Fe
H and He are by far the MOST abundant (why - first elements to form; these eventually formed the rest; also light and volatile - blown off terrestrial rocky planets to outer solar system, forming gas giants)
Big Bang
All matter concentrated in an infinitesimally small and dense singularity. This singularity exploded in the Big Bang, resulting in continual expansion of the universe since the explosion (red shift).
Formed H (75%) and He (25%) (big bang nucleosynthesis). These atoms began to exert gravity (have mass), pulling atoms closer and closer until they form nebulae (gas clouds). Eventually dense enough to COLLAPSE inward and form stars.
Nuclear Fusion/Stellar Nucleosynthesis
Occurs in every star = nuclei smash together to form larger elements that then collide with other elements to form even LARGER elements (ex. H + He = Li, He + He = Be). This can form elements up to Fe (atomic # = 26)…
Why is Fe the cutoff?
Iron has the strongest nucleus/is the strong force = strong nucleus prevents addition of more protons (NO MORE LARGER ELEMENTS).
Supernovae
As H/He are consumed, heavier/less reactive elements concentrate (e.g., accumulation of Fe). Fusion ceases with lack of fuel, and the star collapses. Protons and electrons unite, and the neutron core rebounds in a supernova. Releases enough energy to slam nuclei together and form elements > Fe (supernovae nucleogenesis).
Elements greater than Fe are rare B/C supernovae only sometimes occur with star death + long life cycle for stars.
Differentiation of Elements in our Solar System
Central star formed with atoms exerting enough gravity at high density. Lighter elements (H, He) blown away by solar winds, freezing to form gas giants. Heavier elements (Si, Fe, Mg) retained closer, forming terrestrial planets.
EIGHT MAJOR ELEMENTS OF THE LITHOSPHERE
O
Si
Al
Fe
Ca
Na
K
Mg
All formed via stellar nucleosynthesis (more common than supernovae nucleogenesis + NOT light enough to be blown away)
Elements of the Hydrosphere, Atmosphere, and Biosphere
Atmosphere = N, O, H, Ar, C
Hydrosphere = O, H, Ca, Na, Cl, Mg
Biosphere = H, C, O, N P, S
Ionization Energy
Amount of energy required to dislodge an electron from an atom
First Ionization Energy
Amount of energy required to dislodge the LOOSEST electron from an atom.
Controls on Ionization Energy
Number of protons/nuclear charge (increase protons = increase energy)
Distance of e- to nucleus (e.g., atomic radius) (increase = decrease energy)
Ionization Energy on the Periodic Table
Rows = INCREASE from left to right (adding protons)
Columns = DECREASE from top to bottom (ex. Ne -- Na; e- is farther from the nucleus with added electron shells)
How does low ionization impact charge of ions?
Group I/II atoms are likely to form cations (low ionization energy) = not a lot of energy to dislodge an e-. Group VI/VII atoms are likely to form anions (high ionization energy) = unlikely to dislodge e-.
Electronegativity
Tendency of an atom to attract an additional electron. Usually atoms with high ionization energy (top right of periodic table) = F, O, Cl, S, Br, I, and N.
Cations = low ionization energy + low electronegativity (easily lose e-
Anions = high ionization energy + high electronegativity
Cations and anions always ______ each other in waterways.
BALANCE (moles of cations = moles of anions)! Sum of + charge = sum of - charge!
Common Waterway Cations
Ca2+, Mg2+, Na+, K+, H+, Cs+, Rb+, Sr2+
Common Waterway Anions
Cl-, F-, Br-, I-, NO3-, SO42-, CO32-
How do complex polyatomic anions and neutral molecules form in water?
HYDROLYSIS and PARTIAL HYDROLYSIS!
Hydrolysis with Si4+
Si4+ (aq) + 4H2O (l) → H4SiO4 (aq) + 4H+ (aq) — strongly-charged molecules can break up water molecules AND generate acidity (release free H+). This reaction controls the common form of elements in waters (thus H4SiO4 and SO42- exist rather than S6+ and Si4+).
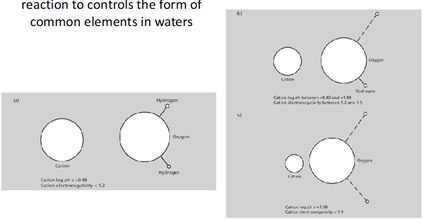
What determines whether an atom undergoes hydrolysis or not?
Cation with HIGH electronegativity + high charge density (e.g., small radius + high charge) = readily react with water, breaking off hydrogens from O. P6+ can also undergo hydrolysis. Al3+ and Fe3+ can undergo PARTIAL hydrolysis (loss of only one H).
Units of Concentration
Mass per liter (g/L, mg/L, micrograms/L)
Moles per liter (mol/L where mole = 6.022 × 10²³ atoms)
Mole per mass (mol/kg)
Parts per part (ppm, ppb)
Parts-per-million (ppm)
Same as mg/L (same value, can interconvert). Same as saying ___ g / 10^6 grams. Note that density of groundwater and surface waters in this class is 1 g = 1 mL (for conversions) (EXCLUDES oceans)
Parts-per-billion (ppb)
Same as micrograms/L (same value, can interconvert). Same as saying ___ g / 10^9 grams. Note that density of groundwater and surface waters in this class is 1 g = 1 mL (for conversions) (EXCLUDES ocean).
Concentration
How much of a substance is present physically
Activity
How much of a substance can be predicted to participate in a chemical reaction. Accounts for the electrostatic, interfering forces of other ions in solution! Mixture of multitude of ions = interfering electrostatic attraction between chemical species reduces the reactivity of reactions. Utilizes a correction factor…
Activity Equation
Activity = γimi where mi is the concentration (IN mol/L) and γi is the activity coefficient! Activity coefficients range from 0 to 1 depending on ionic strength. 0 = complete interference (no reaction of ion). 1 = no other ions present in solution results in no interference (activity = concentration).
Ionic Strength (I.S.)
Unitless measurement that characterizes the number of moles of charge per liter of an ENTIRE solution.
I.S. = ½ SUM (mizi2) where mi is concentration (mol/L) and zi is charge of each species. I.S. accounts for effect of multivalent ions.
As ionic strength increases, γi ________
DECREASES (and so does activity). More complex solution = more interference = lower activity!
Converting from mmol to M
1000 mmol (mM) = 1 M
Steps to Solving for Activities of Chemical Species in a Solution
Convert concentration values to mol/L
Calculate ionic strength of the solution.
Calculate the activity coefficients for EACH ion
Calculate the activities (Activity = miγi)
Three Forms of Calculating the Activity Coefficients
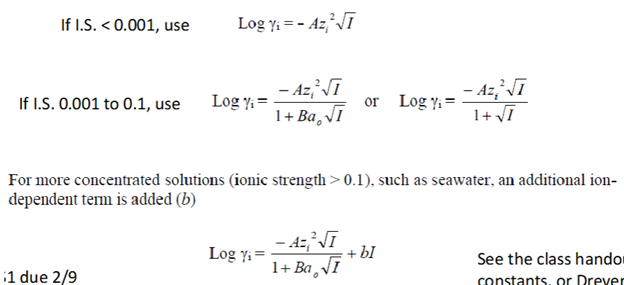
What is the activity coefficient dependent on?
Ionic strength, temperature (A), charge of the chemical species, size of the species, etc. In this class, the temperature of waterways is always assumed to be 25 degrees C (A = 0.5085).
Activity values should always be lower than ____
CONCENTRATION VALUES!!! Activity coefficient = a % of the concentration of a chemical species that can be predicted to react in a chemical equation.
Gibbs Free Energy Equation
ΔG° = ΔH° - TΔS°
ΔH°
Change in enthalpy (heat). Positive = increase in heat. Negative = decrease in heat (favored spontaneously).
ΔS°
Change in entropy (disorder). Positive = increase in disorder (favored spontaneously). Negative = decrease in disorder.
What conditions of H and S are favored spontaneously (e.g., a reaction happens naturally)?
Negative ΔH° and positive ΔS° (release heat = less E = more stable; more disorder = more stable). Note that the direction of a reaction can depend on T when both are the same sign!
When ΔG° is negative…
The reaction is SPONTANEOUS as read (from left to right). Likely a negative ΔH° and positive ΔS°.
When ΔG° is positive…
The reaction is REVERSE SPONTANEOUS as read (spontaneous right to left; e.g., opposite direction). Likely a positive ΔH° and negative ΔS°. Requires energy input from left to right.
Photosynthesis-Cellular Respiration Example
6CO2 (g) + 6H2O (l) ←→ C6H12O6 (s) 6O2 (g) — respiration proceeds right to left. Photosynthesis proceeds left to right. This equation happens to be SPONTANEOUS from right to left (e.g., respiration) due to an increase in entropy from right to left (and decrease in heat/enthalpy).
Photosynthesis, thus, REQUIRES energy (in the form of sunlight).
ΔG°f
Standard free energy of formation = free energy change when one mole of the species is formed from its constituent elements in standard state. Values on HANDOUT!!!
ΔG°Reaction = ΔG°f (products) - ΔG°f (reactants) where ΔG°R predicts spontaneity…
Negative ΔG°R
Spontaneous left to right!!
Positive ΔG°R
Spontaneous right to left
ΔG°R of 0
The reaction is in equilibrium (no tendency in either direction).
Equilibrium
A steady state where the relative amounts proceeding forward and backward in a reaction are equal (e.g., EQUAL FLUX). Often seen in geology → amount of continental landmass remains in a steady state (ex. processes that form continents (subduction = volcanoes) BALANCE processes that destroy continents (erosion)) and amount of seafloor remains in steady state (divergent creation and convergent subduction/recycling).
K
The equilibrium constant (e.g., the ratio of products to reactants AT equilibrium). Will remain constant.
For an equation aA + bB ←→ cC + dD… K = [C]c [D]d / [A]a [B]b where [] refers to the activity of chemical species.
What happens to products and reactants as a solution proceeds to equilibrium?
Whatever is in abundance (e.g., reactants) will decrease in concentration, and whatever is in deficit (e.g., products) will increase in concentration. Reactants and products do not have to equilibrate at the same concentration. Change in concentration just has to asymptote to a slope of 0.
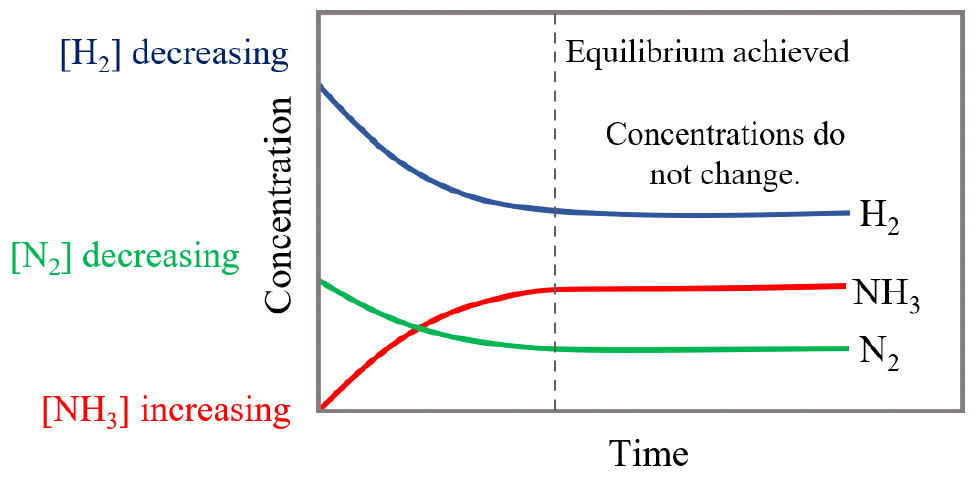
Dissolved Species in the K Equation
Use ACTIVITY = miγi where mi is in mol/L
Solids & Liquids in the K Equation
The equilibrium constant is always 1 (for instance, liquid H2O)
Gases in the K Equation
Express activity/concentration as PART-PER-PART! Examples = O2 (g) makes up 20.95% of Earth’s atmosphere (thus 20.95 / 100), CO2 (g) exists at 428 ppm in our atmosphere (thus 428 / 10^6), and SO2 (g) exists at 14 ppb over coal-fired power plants (thus 14 / 10^9)
How can you solve for K?
log K = - ΔG°Reaction / 5.708 kJ/mol (relates standard Gibbs free energy of a reaction to its K). K is a unitless constant.
K > 1
More products than reactants (products favored)
K < 1
More reactants than products (reactants favored)
K = 1
Neither the products nor reactants are favored (equal concentrations/activities)
Once you solve for K, how can you use it?
You can plug K into the equilibrium constant equation (with products over reactants) and solve for the activities of specific chemical species (ex. H2CO3 in the CO2 raindrop chemistry example) by plugging in other known activities (ex. H2O (l) = 1, CO2 (g) = 428/10^6, etc.). Activities are in mol/L!
In non-polluted conditions, how does the activity of H+ relate to the activity of HCO3- in raindrops?
They must BALANCE/EQUAL! So that there is CHARGE BALANCE in the raindrop. Other ions are not important/no effect on charge of raindrop (due to such small concentrations).
So… [H+] = [HCO3-] → can write as [H+]2 if both on same side of equation. Used to solve for pH (concentration of H+).
pH
pH = -log [H+]
Rainwater is _______ due to the dissolution of CO2 from the atmosphere in rainwater.
ACIDIC! Generates H2CO3 that then dissociates into H+ (generating acidity) and HCO3-.
What makes SO2 (g) from coal-fired power plants such a dangerous pollutant?
The equation SO2 (g) + H2O (l) ←→ H2SO3 (aq) has a MUCH LARGER K (1.66) than the CO2 dissolution example (products favored MUCH more). Thus, SO2 more readily dissolves in water droplets, and H2SO3 then dissociates, forming larger concentrations of H+ (more sulfuric acid). Larger production of acidity = stronger acid rain (ex. 0.017 / 10^-6.366 → H2SO3 dissociates by a factor of 40,000 more readily).
How can you calculate comparative solubility in water?
Divide the K’s of each DISSOLVING equation (ex. 1.66 / 0.0342 = 48x more soluble for SO2).
How is SO2 quantitatively much more dangerous as a pollutant than CO2?
At just a concentration of 14 ppb, there is a pH of 4.7 in raindrops with SO2 (g) (due to high K’s of dissolving and dissociation). CO2 = 428 ppm and pH of 5.6! Thus, SO2 has been the more targeted pollutant for legislation.
K is a _____ value
PREDICTIVE — can differ from the actual concentration due to various occurrences (ex. pollution = more input of CH4 into the atmosphere faster than the reaction that breaks down CH4 (slow)).
Calculating [H+] Using pH
pH = 10^-pH
Ratio of Activities of Chemical Species
You can also solve for the RATIO of activities (one over the other) to see how much more common one species is over the other.
[OH-]
Can used pH to solve for [OH-] because [OH-] = 10^-14 / [H+]…
When working with mineral dissolution and precipitation, write the reaction as dissolving from ______
LEFT TO RIGHT
If K predicts dissolution, how do you sometimes get precipitation of a mineral?
Le Chatelier’s Principle (Q does not equal K)
Le Chatelier’s Principle
If equilibrium is disturbed by changing conditions, the position of equilibrium shifts to counteract the change and reestablish equilibrium. Example = with water evaporation, there is a decrease in volume and thus increase in concentration of dissolved species. Thus, the reaction will proceed right to left (precipitation to remove dissolved species).
If we have known activities (NOT theoretical molar proportions), we can use ____
ΔGReaction (not standard)
ΔGReaction
ΔGReaction = R * T * ln(Q / K) where R is the gas constant and T = temperature
Q
Ion activity product! Q is the same ratio of products over reactants as K BUT it is in current conditions (not at equilibrium). E.g., this uses GIVEN moles.
Saturation Index
Q / K
If Q < K, ΔGR is neg → left to right → dissolution favored
If Q > K, ΔGR is pos → right to left → precipitation/formation favored
Steps for Mineral Dissolution/Formation Problems
Write out mineral equation
Find ΔG°Reaction (molar proportions)
Find K (using ΔG°Reaction)
Find Q (by solving for activities of ions of interest FROM mg/L)
Calculate ΔGReaction (Q/K predicts direction)
Minerals
Naturally-occurring crystalline solids that have a definable chemical formula. Volcanic glass = not a mineral b/c amorphous (random structure).
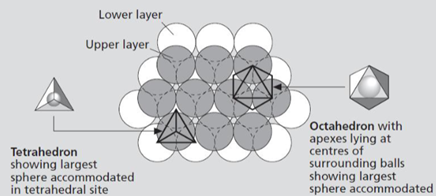
How are elements ordered and packed together in minerals?
Anions = large spheres that stack in closest possible arrangement.
Two Types of Voids
Tetrahedral coordination (4 anions)
Octahedral coordination (6 anions)
Si4+ Tetrahedra
This cation is very small and fits into TETRAHEDRAL coordination sites of O2- (octahedral = larger cations like Al3+). Covalently-bonded tetrahedra… thus, these O2- anions have 7/8 valence e-, making them prone to bonding (form mineral structures).
What predicts the coordination number (# of surrounding anions) of a cation?
Ratio of the radius of the cation to the radius of the anion. In silicates, this focuses on O2- as the anion. Si4+ = coordination number of 4 in silicates based on atomic radius differences.
Silicate Cations w/ Coordination Number 4
Si4+, Al3+, Be2+
Silicate Cations w/ Coordination Number 6
Al3+, Fe2+, Fe3+, Mg2+, Li+, Na+, Ca2+
Silicate Cations w/ Coordination Number 8
Na+, Ca2+, Sr2+, K+, Ba2+
Silicate Cations w/ Coordination Number 12
K+, Ba2+, Cs+
Cations that are close in size can ___________
SUBSTITUTE for one another in crystal structures (ex. Si4+ and Al3+, Mg2+ and Fe2+, etc.).
Mafic Magmas
Relatively large proportions of Mg2+/Fe2+ (can form mineral networks with these cations connecting different Si-tetrahedra)
Felsic Magmas
Relatively low proportions of Mg2+/Fe2+ (thus mostly interconnected Si-tetrahedra)
How can oxygen atoms within the Si-O tetrahedrons fill their valence shell?
Ionic Bonds (monomer silicates)
Covalent Bonds (other silicates)
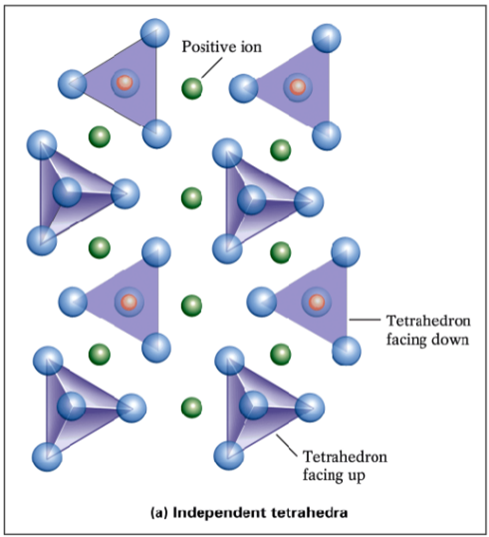
Monomer Silicates
Ionic bonds between a Group I/II cation (ex. Fe2+, Mg2+) and oxygen atoms of tetrahedra (ionic bridges). Thus, tetrahedra are isolated (no O-sharing). The Si:O ratio is 1:4. Includes olivine. NOT resistant to weathering (very weak).
Covalent Bonds
Electron-sharing between oxygens (O-sharing) in each tetrahedra allows polymerization of tetrahedra. Includes single-chain, double-chain, sheet, and framework silicates.
Single Chain Silicates
Each Si-tetrahedron shares 2 corner O with other tetrahedra. Forms single chains. Linked to other chains via large cations (weak ionic bonds). Si:O is 3! Includes pyroxenes
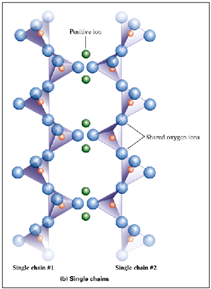
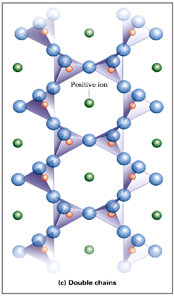
Double Chain Silicates
Each Si-tetrahedron shares 2 OR 3 corner O with other tetrahedra. Interspersed cations = additional bonding between chains. Si:O is 1:2.75! Includes amphiboles
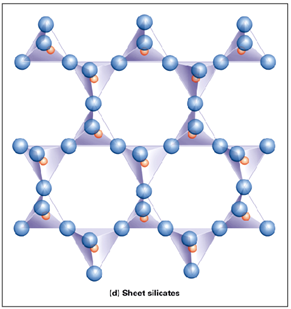
Sheet Silicates
Each Si-tetrahedron shares all 3 basal oxygens with other tetrahedra. Forms flat sheets. “Apical” oxygens bond to positive ions and link sheets. Si:O is 1:2.5! Includes micas and clays.
Framework Silicates
All oxygens are shared between adjacent tetrahedra. Si:O is 1:2! Includes quartz and feldspars. Feldspars are framework silicates with Al3+ substitutions for Si4+ → creates a (-1) charge that is balanced by the addition of K+ (alkali feldspars) OR Na+/Ca2+ (plag feldspars). E.g., substitution = mineral families!
As you move from isolated to single chain to … to framework, what occurs?
Increasing polymerization (connectivity)
Increasing O-sharing
Increasing covalent bond %
Increasing hardness
Decreasing solubility
Increasing % silica (Si)
More resistant to weathering
Carbonates
Common primary group of minerals! Important anion is CO32-! Very soluble! Calcite and dolomite ((Ca,Mg)CO3) precipitate out in oceans and are common in sed (limestones, dolostones) and metamorphic (marble) rocks.
Chemical Weathering
Results in a change in composition! Primary minerals are often unstable and undergo chemical rxns to form secondary minerals.
Two Main Types of Chemical Weathering Reactions
Congruent Rxns
Incongruent Rxns