L5 - Trace Element Geochemistry
1/74
Earn XP
Description and Tags
Covers Partition Coefficients (KD) and compatibility, types of trace element variation diagrams, rare earth elements, high field strength elements, and large ion lithophile elements.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
75 Terms
What is a trace element?
They substitute for major or minor elements since they are too dilute to form their own phase.
What are the concentrations of a trace element?
<0.1 wt.%
How are trace elements reported?
often reported as ‘ppm‘ or more properly micro-grams/gram
What is the partition coefficient, D/KD?
the concentration of the element in the solid / the concentration of the element in the melt
D = [element]solid / [element] melt
What does the partition coefficient represent?
The compatibility of an element
When D>1 is it compatible or incompatible?
Compatible - easily incorporated into the crystal lattice
When D<1 is it compatible or incompatible?
Incompatible - hard to incorporate into the crystal lattice.
What is the Bulk partition coefficient (DB)?
The weighted average of individual partition coefficients for each mineral phase

How to determine a mineral-melt partition coefficient?
directly measure in natural systems
directly measure in synthetic systems (make your own rocks with control)
calculate based on thermodynamics
What is an example of directly measuring in a natural system?
Measuring the strontium ratio in the rim of a plagioclase and the matrix with an Electron Microprobe or laser. Divide the rim composition by the Matrix/glass composition to get the partition coefficient.
What is an example of measuring in a synthetic system?
Squeeze a rock powder with a piston to make it melt and quench to measure the solid and liquid stages.
What are some essential variables needed to calculate the D-value of an element?
Essential Variables:
Pressure
Temperature
Bulk Composition
Elastic Properties of a cationic site
What is strain energy?
Generated when substituting a cation with a different IR to the major cation.
Proportional to a measure of the stiffness (Young’s modulus, E = stress/strain) of the cation-oxygen bonds in that site.
How does cation size relate to the strain exerted?
A larger cation exerts a positive strain
A smaller cation exerts a negative strain
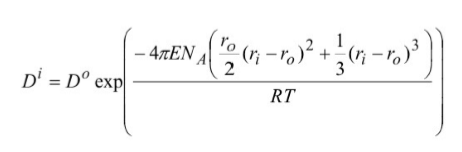
What do the following terms mean:
E
NA
r0
ri
R
T
D0
Di
E - Youngs modulus (stiffness of site)
NA - Avogadro’s number
r0 - Radius of site
ri - radius of substituting cation
R - Gas constant (free roaming)
T - Temperature (reflects earth conditions)
D0 - Partition Coefficient of the major cation site
Di - the partition coefficient of an element into a given site
When is the minimum strain energy reached?
When ri = r0
When the cation has an identical radius to the site radius.
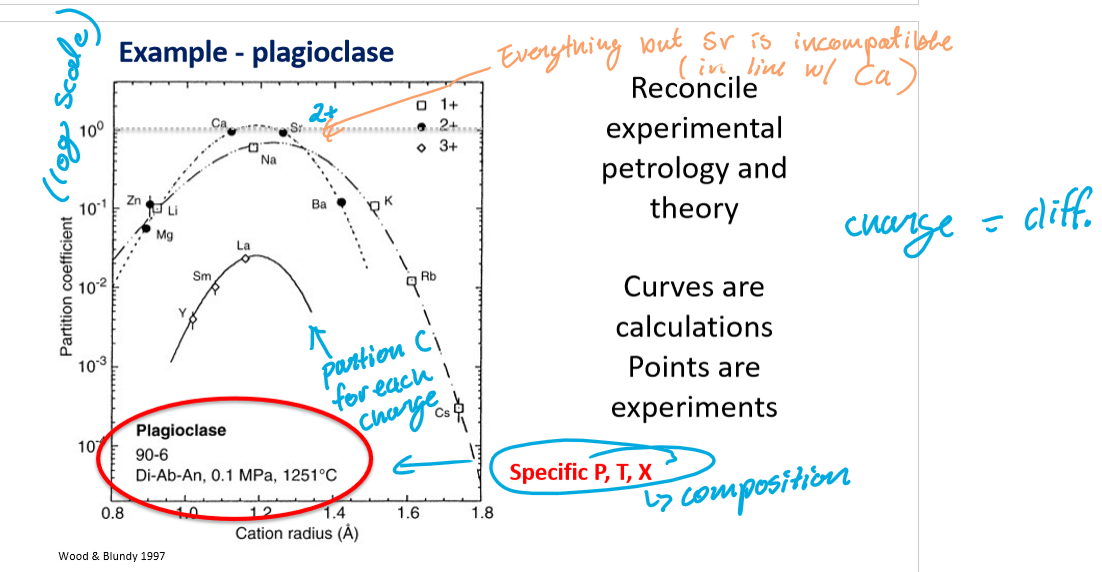
What do the curves represent?
The calculated partition coefficients for each charge at a specific temperature, pressure, and composition.
What are the major elements?
Na, Mg, Ca, Fe, Al, Si, O, Ti, K, P, and Mn
What element groups make up the Large Ion Lithophile Elements (LILE)?
Alkali and Alkaline Earth Elements
Specifically K, Rb, Cs, Sr, and Ba
Do the LILEs have a high or low electronegativity?
Low electronegativity (<1.5); therefore, want to get rid of electrons
What is the valance for
Alkalis
Alkali Earths?
+1
+2
Do they form strong or weak Ionic bonds?
strong (except for Be)
How does water affect the bonding of LILEs? What is the result?
Water disrupts the bonding of LILEs and could cause and explosion
Are LILEs soluble or non-soluble in aqueous solutions? How does this relate to mobility in metamorphism and weathering?
LILEs are relatively soluble in aqueous solutions and therefore quite mobile during metamorphism and weathering.
What factor controls the behavior of LILEs in igneous rocks?
Ionic radius and charge (remember valence)
What is the range of ionic radii from the LILEs?
From 118 picometers (pm) for Sr to 167 pm for Cs
What type of sites are required to incorporate LILEs?
Needs 12-fold sites (between sheets in phyllosilicates)
Hard to fit into 4 and 6-fold sites resulting in distortion of the lattice (highly incompatible in most minerals)
What are the main cationic lattice sites in basaltic and ultramafic rocks?
tetrahedral (4-fold)
octahedral (6-fold)
What cations occupy the tetrahedral (4-fold) and octahedral (6-fold) sites?
Tetrahedral (4-fold) - occupied by Si and Al (and less often by Fe3+ and Ti4+)
Octahedral (6-fold) - usually occupied by Ca, Mg, or Fe (less rarely by Na)
Where do the LILEs concentrate/accumulate during differentiation?
Concentrate in the melt phase, since they are incompatible.
accumulate in late products of differentiation
What groups of elements make up the Rare Earth Elements (REE)?
The Lanthanides and Actinides (two rows of elements at the bottom of the periodic table)
only includes 2 actinides (U and Th), since they are the only ones stable enough to survive over the history of the Earth
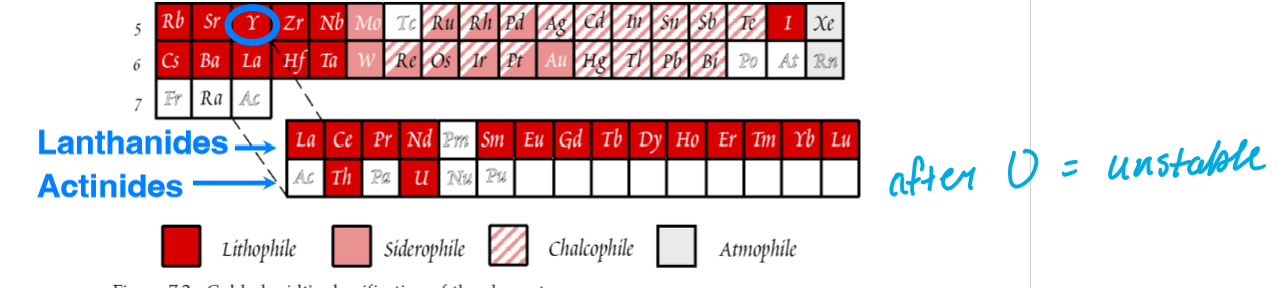
Why is Y included in the REEs?
Y shares the same chemical properties, including charge and ionic radius, as the Heavier rare earths, and as a result behaves much like them.
What is the valence of lanthanides?
3+ over a wide range of oxygen fugacities
What are some exceptions to the valance state? (Hint: describes 4 different elements)
Ce4+ and Eu2+ exist in addition to Ce3+ and Eu3+ (two valance states).
Ce4+ and Eu2+ = high oxygen levels
Ce3+ and Eu3+ = low oxygen levels
Th is always 4+ valence state
U may be 4+ or 6+, depending on oxygen levels
Are REEs soluble or insoluble in aqueous solutions? Why?
Relatively insoluble in aqueous solutions.
due to their higher charges and high ionic potential
What is one exception to REE insolubility?
U can be soluble in its oxidized form (U6+), to form a soluble oxyanion complex UO22-.
What is the order of orbital shells filled?
1st = S orbitals
2nd = P orbitals
3rd = D orbitals
4th = F orbitals
What causes a similar chemical behavior in the lanthanides?
The 4f orbitals are being filled, so the configuration of the valence electrons is similar in all the REEs.
What groups fill the following orbitals?
S orbitals
P orbitals
D orbitals
F orbitals
S orbitals: alkali metals (group 1) and alkaline earth metals (group 2)
P orbitals: Groups 13-18, include the boron group, carbon group, nitrogen group, chalcogens, halogens, and noble gases (excluding Helium)
D orbitals: Groups 3-12, the transition metals
F orbitals: Filled by lanthanides and actinides
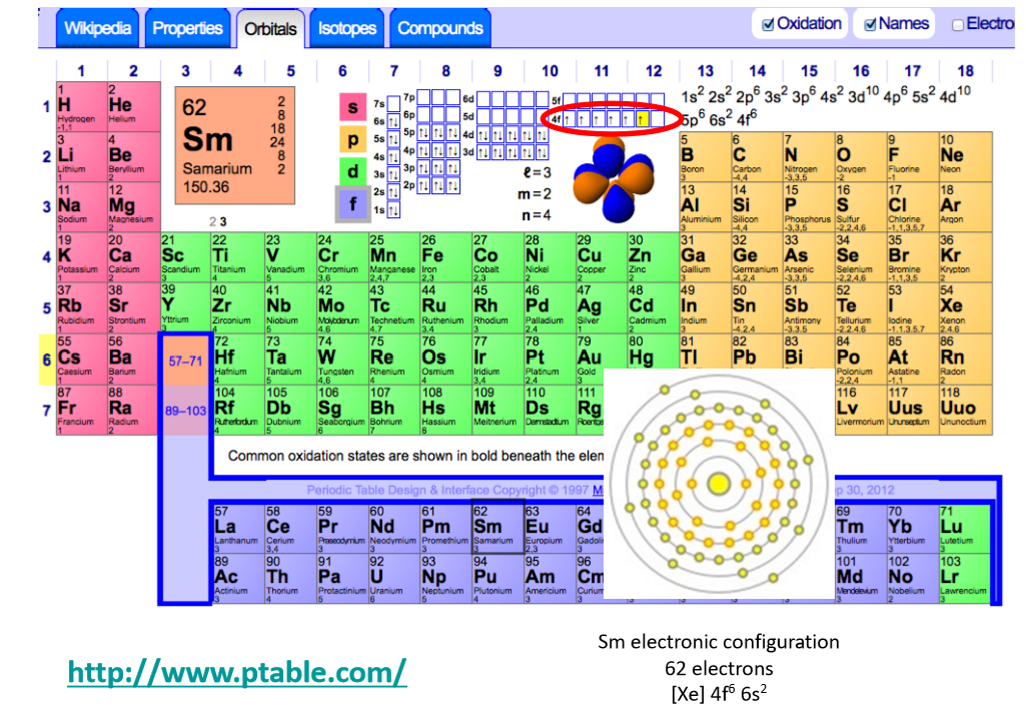
How does the ionic radius of the REEs change as you move from left to right across the rows?
Ionic radius decreases progressively from La3+ to Lu3+ by 25%
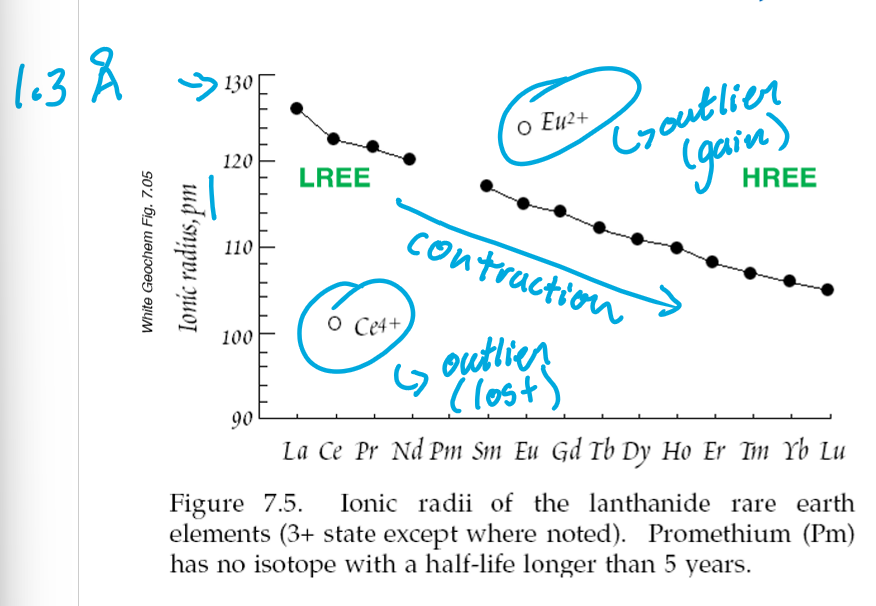
Based on the calculated D/KD of REEs in Cpx, are the REEs compatible or incompatible into Cpx?
HREEs are compatible and are incorporated into the crystal lattice
LREEs are incompatible and stay in the melt
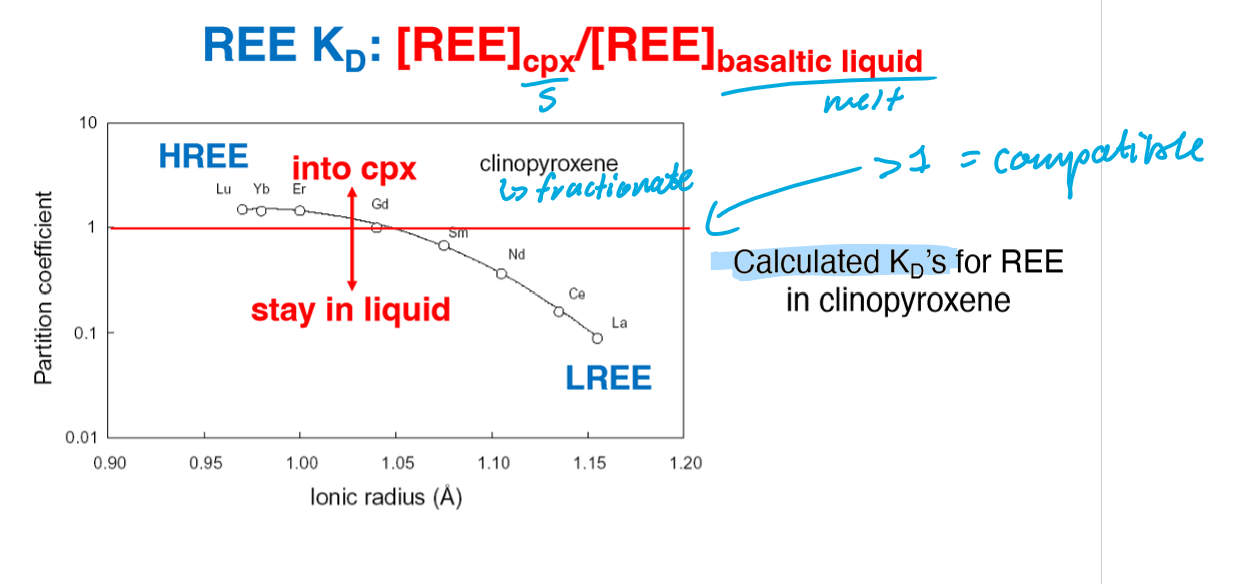
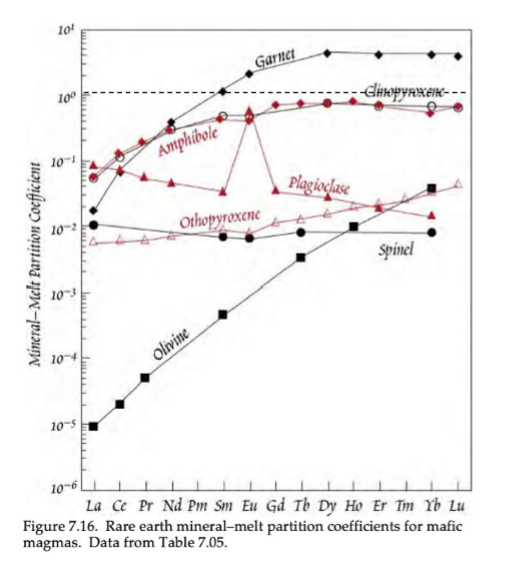
Based on this image of trace element D-Values, what elements show compatibility of REEs?
Garnet is the only mineral that shows compatibility of HREEs.
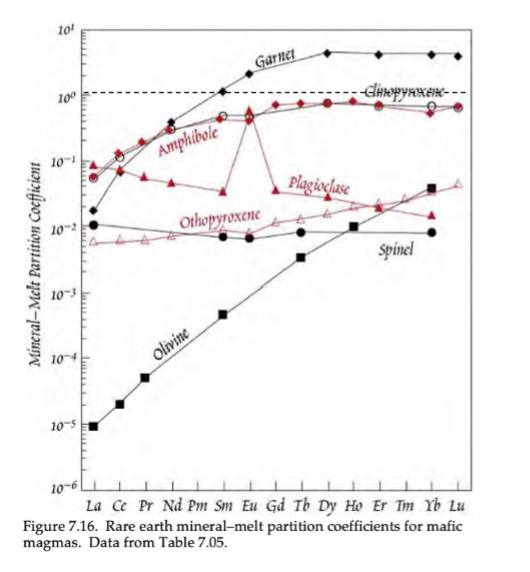
Based on this image of trace element D-Values, what causes the general inclination and declination trends?
Inclination trend = more compatibility of HREEs compared to LREEs
Declination trend = more compatibility of LREEs compared to HREEs
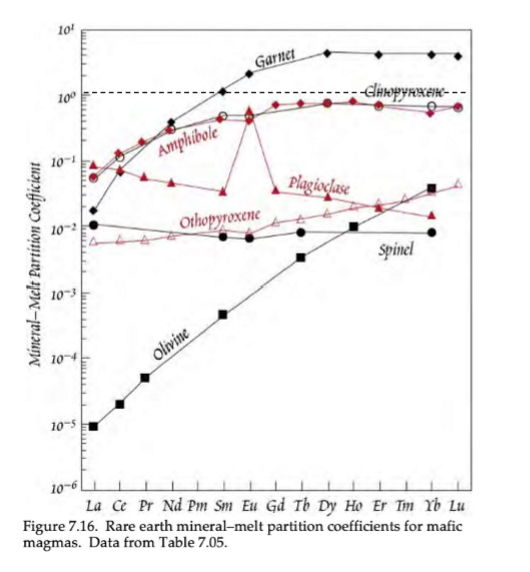
Based on this image of trace element D-Values, what is the cause for the outliers (Eu anomaly in plagioclase)?
Outlier (Eu anomaly in plagioclase) is caused by the two different valance states in Eu (Eu2+ and Eu3+), meaning it can be incorporated into two different fold sites.
What creates more flexibility in the sites of Cpx?
slightly inclined
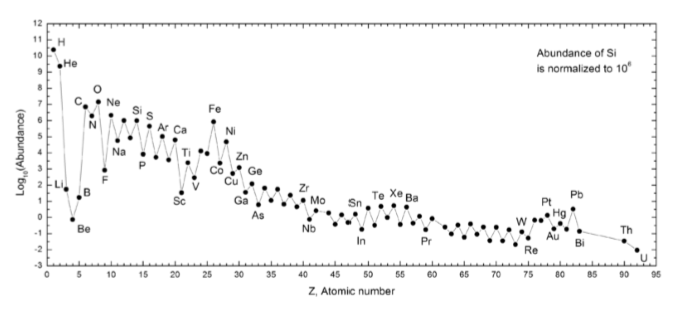
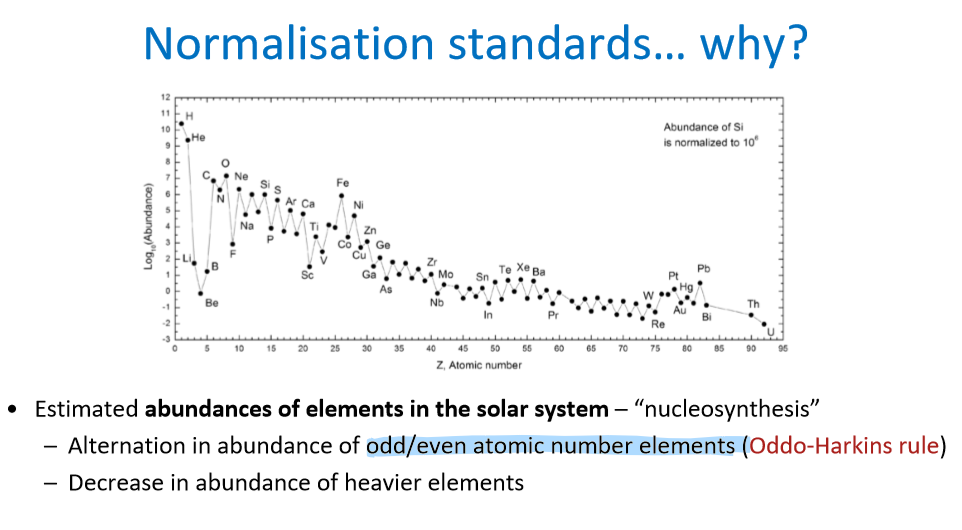
What is the name for the zigzag pattern observed in this graph? What causes it? How do we remove it?
Zigzag pattern is called the Oddo-Harkins Rule/Effect
Caused by the alternation of odd/even atomic number elements
Removed by normalizing with (dividing by) a chondrite or others
What is a spider diagram?
An incompatible element normalized variation diagram
The X-axis goes from most to least incompatible
y-axis is sample/normalization (in log scale)
What are Spider Diagrams used for?
used to visualize and compare the trace element or REE compositions in rocks
how enriched/depleted etc.
What elements make up the High Field Strength (HFS) Elements?
Zr, Hf, Ta, Nb + U and Th
What are these elements considered HFS?
Due to their high ionic charge: Zr4+, Hf4+, Ta5+, and Nb5+
Th and U are sometimes included in this group because of their multiple valence states (Th4+ and U6+ or U4+)
What is a consequence of the HFS high charge?
High charge results in all of the elements being relatively small cations
What is the ionic radii range in HFS?
64 pm for Nb5+ and Ta5+
72 pm for Zr4+
76 pm for Hf4+
U4+ and Th4+ are larger
What makes the HFS elements incompatible?
size may be appropriate for many cation sites in common minerals, but their charge is too large and requires one or more coupled substitutions to maintain charge balance (energetically unfavorable)
What is the difference in compatibility within the HFS elements?
Hf and Zr = moderately incompatible elements
Nb and Ta = highly incompatible elements
Are HFS soluble or insoluble in aqueous solutions? Why?
Very insoluble
Their ionic charge/ionic radius ratio (high electrostatic potential)
Are HFS mobile or immobile during weathering and metamorphism?
very immobile
How is immobility displayed on a graph?
through the presence of many different trends
How many valance states do the Transition Metals have?
2 or more (since in D block)
Do the transition metals have higher or lower electronegativity than alkali and alkaline earths? What type of bonding is expected?
Higher electronegativity; therefore, more covalent bonding
What elements do transition metals bond to to create:
ionic bonds
covalent bonds?
ionic bonds - bond with oxygen in oxides and silicates
covalent bonds - bond with other non-metals (ie. sulphur)
What is the level of solubility in transition metals?
Solubility is variable
moderately Incompatible = Ti, Cu, and Zn
Very compatible = Cr, Ni, Co
How is mobility differences found using a graph?
compare altered and fresh samples on the same plot
Mobile = change in concentration (lower or higher when comparing)
immobile = no change in concentration (remains the same in both samples)
Where do semi-volatile elements partition into?
into a fluid or gas phase (Cl, Br)
or form compounds that are volatile (C → CO2, S → SO2)
generally in trace concentrations
What determines whether the semi-volatile elements partition into a fluid or gas phase?
the measure of oxygen fugacity (O levels)
low O = fluid
High O = gas
What does the solubility of CO2 in silicate magmas determined by?
Is a function of pressure
low [CO2] = CO2-H2O gas phase exsolved from magmas
High [CO2] and CO2/H2O ratios = immiscible carbonatite liquid (CaCO3 is a dominant component)
What happens to the remaining Semi-Volatile elements?
Always present in trace concentrations
never reach saturation in magmas
never exsolve as independent gas or fluid phase
partition into gas phase formed by exsolution of CO2 and H2O
Do Noble gases chemically bond in rocks and minerals?
Never bond
Do noble gases have a large or small radii? How does this effect their incorporation into minerals?
Large radii
cannot be easily accommodated into cationic or anionic sites; therefore, have very low concentrations
What minerals are part of the noble gases?
He,Ne, Ar, Kr, Xe
Where are noble gases commonly found?
Present in inclusions of garnet
What is the most abundant REE? Can it form it’s own phase?
Ce is generally the most abundant REE and forms its own phase in rare instances
What element can the heaviest REE substitute for? What mineral?
Heaviest REEs sub for Al3+ in Garnet
What is the partition coefficient range of Lu in Garnet? What does it depend on?
4-10
depends on the composition of magma and garnet
In mafic minerals, what is more incompatible, HREEs or LREEs? how about Plagioclase?
Mafic = LREEs are more incompatible than HREEs
Plag = HREEs are more incompatible then LREEs (D does not exceed 0.1)
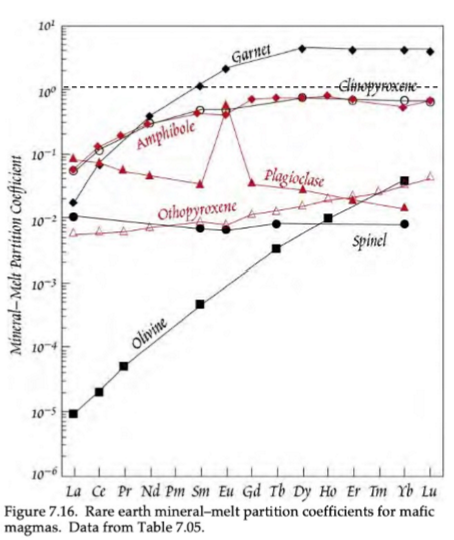
What does Eu substitute for in Plagioclase feldspar? What results form this?
Eu (when in 2+ valance) subs for Ca2+
Plag is often rich in Eu
minerals in equilibrium with plag become relatively Eu-depleted