metabolic integration
1/60
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
61 Terms
three major sources of metabolic fuel for the body
proteins, carbohydrates, and lipids
key pathways required to convert metabolic fuels into ATP
amino acid metabolism, carbohydrate metabolism, lipid metabolism, the citrate cycle, and oxidative phosphorylation
pancreas
secretes insulin in response to rising blood glucose levels
secretes digestive proteases that degrade dietary proteins in the small intestine
trypsin
chymotrypsin
elastase
intestines
small intestine
absorbs nutrients
secretes peptide hormones
large intestine
absorbs water and electrolytes
stomach
prepares food for small intestine by producing chyme
secretes peptide hormones that control eating behaviors through neuronal signaling in the brain
liver
central processing facility and metabolic hub
determines what dietary nutrients and metabolic fuels are sent to peripheral tissues
glucose regulator that removes excess glucose from blood when carb levels are high and releases glucose when carb levels are low
cntrolled by insulin and glucagon signaling
portal vein directly delivers nutrients from the small intestine and inactivates toxins from the diet
the fate of glucose-6-phosphate depends on
the metabolic needs of the liver and peripheral tissues
fates of glucose-6-p
converted into glucose-1-p for glycogen synthesis
dephosphorylated to be released into the blood to be used by other tissues (brain)
converted to 6-phosphogluconolactone in the PPP if NADPH is needed
converted to fructose-6-p for glycolysis to make pyruvate and then make acetyl-CoA for OXPHOS
skeletal muscle
uses fatty acids, glucose, or ketone bodies depending on physical movement required (rapid burst of activity or endurance)
can be used as an energy source for the body during times of starvation
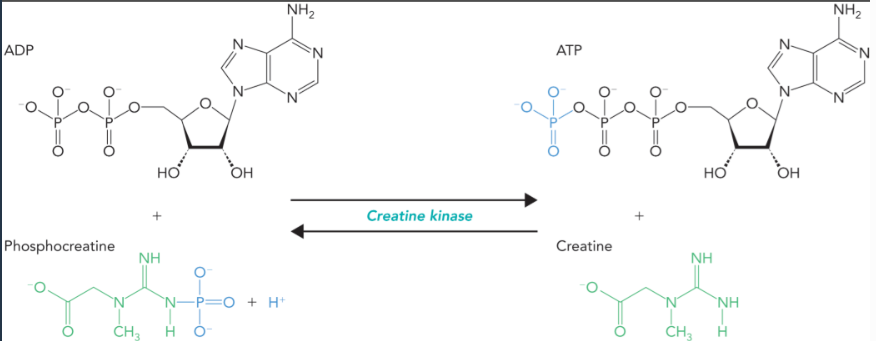
creatine kinase
catalyzes a reversible phosphoryl transfer reaction with ADP and phosphocreatine that generates ATP and creatine when a more sustained level of muscle activity is needed
resynthesizes phosphocreatine from ATP and creatine when ATP pools return to normal
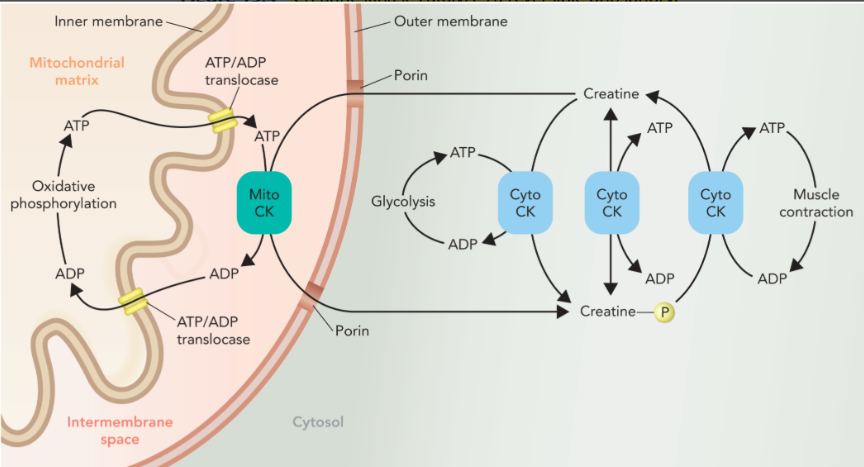
phosphocreatine shuttle
two creatine kinase isozymes
mito CK moves phosphate from the mitochondrial matrix to the cytosol through phosphocreatine
phosphate is transferred to ADP in the cytosol to generate ATP for muscle contraction through cyto CK
cyto CK also phosphorylates creatine to generate phosphocreatine
cardiac muscle
uses mostly fatty acids and ketone bodies to continuously sustain a steady heart beat for an entire lifetime
primary mode of energy conversion is aerobic respiration using acetyl-CoA provided by fatty acid oxidation and ketone bodies
adipose tissue
secretes peptide hormones (adipokines) to regulate metabolic homeostasis
makes up 15-25% of an individual’s mass
two basic types of adipose tissue
subcutaneous: just underneath the skin in the thighs, buttocks, arms, and face
visceral: located deep within the abdominal cavity and secretes adipokines
elevated levels of certain types of adipokines in the blood have been associated with
increased risk of cardiovascular disease
brain
control center of our bodies
20% of the oxygen consumed by the body is used for oxidative phosphorylation
depends mostly on glucose— 60% of the body’s glucose is required by the brain
astrocytes
glial cells that make multiple contacts with endothelial cells of microcapillaries to transport glucose metabolites to surrounding neurons for use in energy conversion reactions that generate ATP
kidneys
located on either side of the abdominal cavity
only other major organ that uses gluconeogenesis to export glucose to other tissues
remove concentrated levels of urea, ammonium, ketone bodies, and other soluble metabolites from the blood
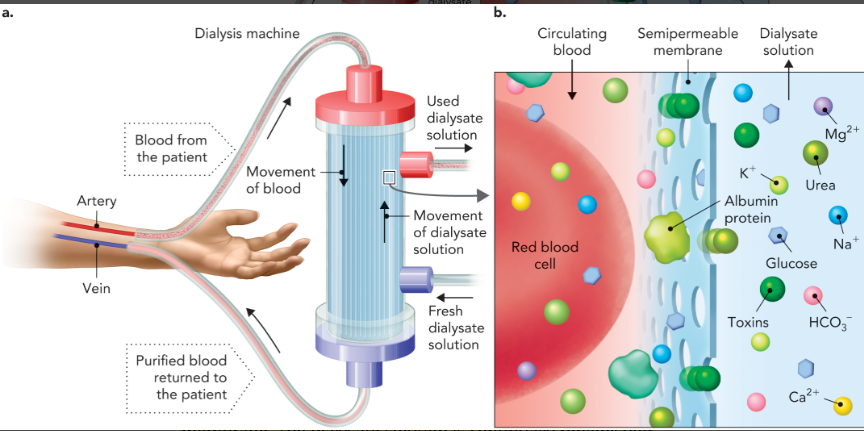
hemodialysis
procedure used by patients with kidney disease to remove waste products from the blood using high diffusion rates across a semi-permeable membrane
metabolic homeostasis
the process of maintaining optimal metabolite concentrations and managing chemical energy reserves within an organism
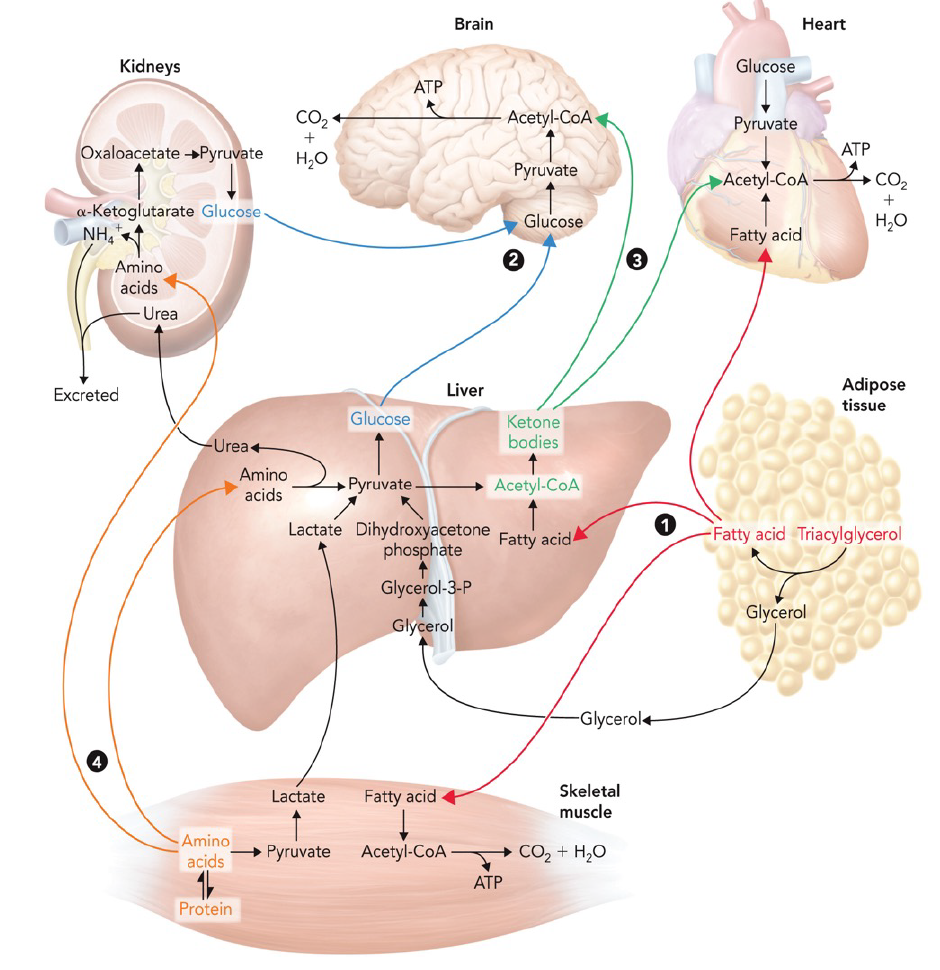
under normal homeostatic conditions the liver is the
control center and plays a crucial role in regulating metabolite flux among tissues and organs
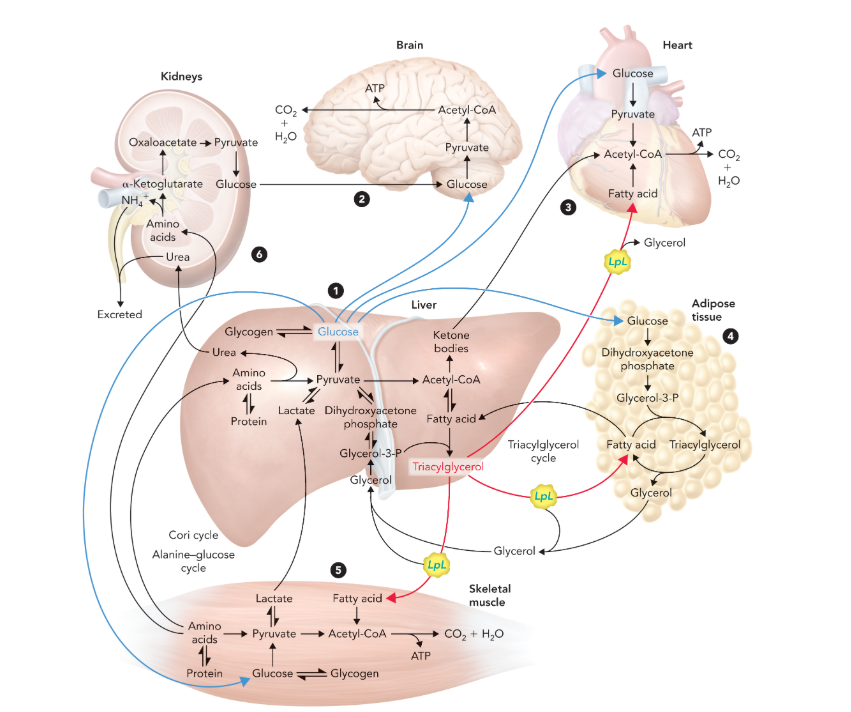
the six primary functions required to maintain homeostatic under normal conditions
liver exports glucose, ketone bodies, and triacylglycerols to the peripheral tissues for use as metabolic fuel
brain requires constant input of glucose
cardiac muscle uses fatty acids and ketone bodies for most energy needs but also uses small amounts of glucose
triacylglycerol cycle maintains circulation of high-energy fatty acids between liver and adipose tissue
skeletal muscle uses glucose and fatty acids from both the liver and dietary sources for ATP synthesis and also exports lactate back to the liver to complete the Cori cycle during prolonged physical exertion
amino acids glutamine and alanine transport excess nitrogen obtained from protein degradation in the muscle to the liver nd kidneys for excretion as urea
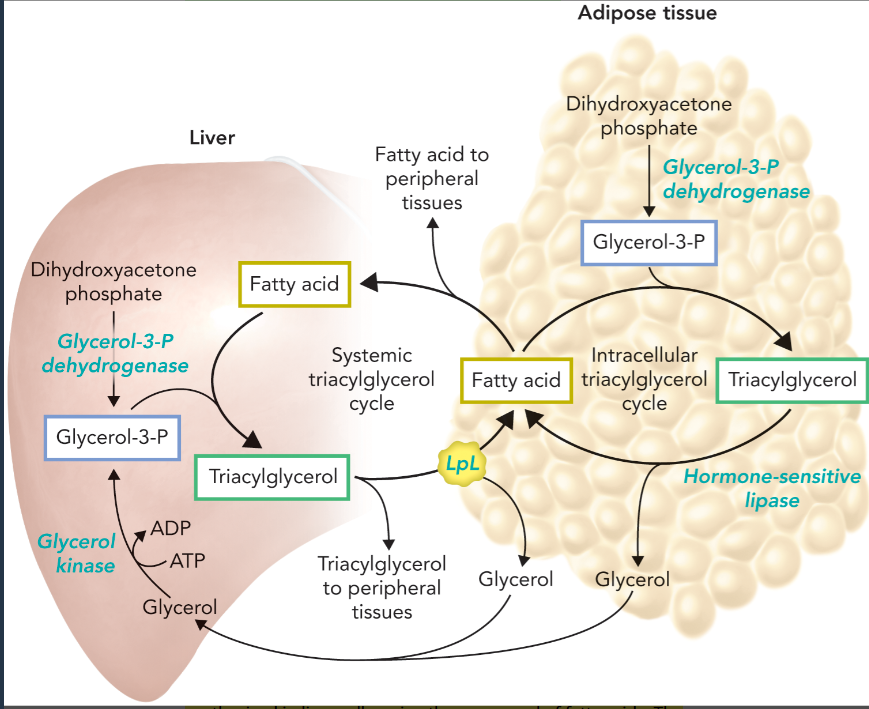
triacylglycerol cycle
an interorgan process that continually circulates fatty acids and triacylglycerols between adipose tissue and the liver to maintain circulation of energy-rich fatty acids that can be used by the peripheral tissues
the two components of the triacylglycerol cycle
systemic component cycles fatty acids between adipose tissue and the liver in the form of fatty acids bound to albumin and TAGs contained in lipoprotein particles
intracellular component in adipocytes cycles fatty acids in the form of cytosolic free fatty acids and TAGs stored in lipid droplets
dependent on sufficient level of glycerol-3-phosphate
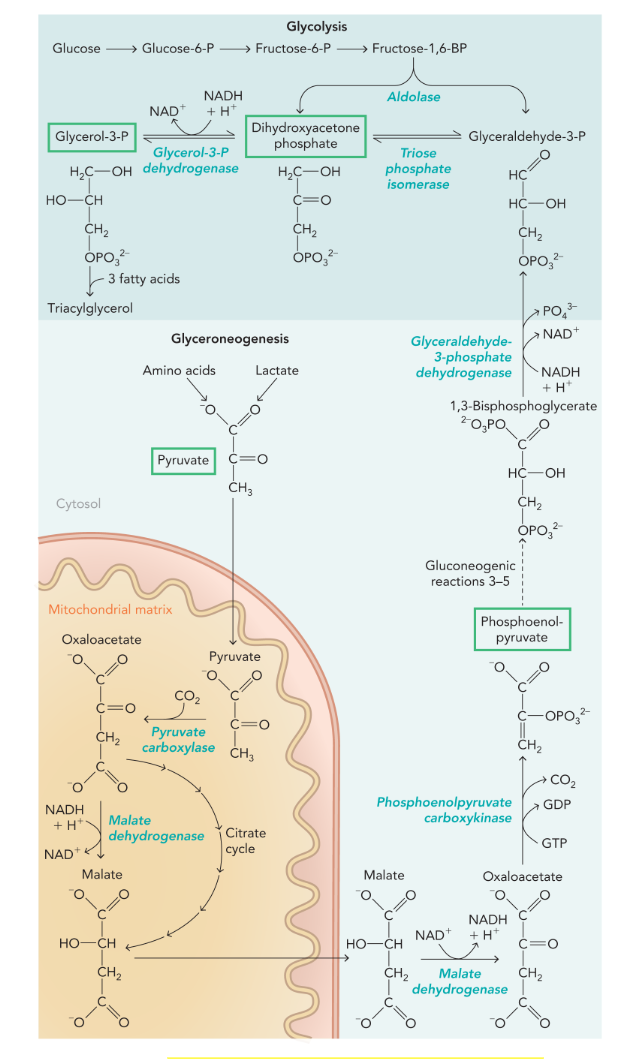
glyceroneogenesis
functions in adipocytes and hepatocytes to synthesize dihydroxyacetone phosphate when blood glucose levels are low (no glycolysis)
uses many gluconeogenic enzymes
does not make glucose
insulin
released by pancreatic beta cells when blood glucose levels are high
stimulates glucose uptake in the liver, skeletal muscle, and adipose tissue
activates glycogen synthesis and fatty acid synthesis
glucagon
released by pancreatic alpha cells
stimulates gluconeogenesis, glycogen degradation, and fatty acid export from adipose tissue when food is not available
glucose uptake in the liver cells is primarily due to (insulin signaling)
increased metabolic flux through glycolytic, glycogen synthesis, and triacylglycerol synthesis pathways
in skeletal muscle insulin stimulates glucose uptake by
increasing the level of GLUT4 protein on the cell surface and also by increasing the flux through glycolytic and glycogen synthesis pathways
in adipose tissue insulin stimulates glucose uptake by
increasing GLUT4, leading to increased fatty acid and glycerol synthesis; and by activating fatty acid uptake from lipoprotein particles to promote TAG storage
in the brain insulin signaling stimulates
neuronal signaling in the hypothalamus that leads to decreased eating and increased energy expenditure (anorexigenic neurons)
glucagon signaling in the liver tissue stimulates
glucose export as a result of increased rates of gluconeogenesis and glycogen degradation
in adipose tissue glucagon activates
TAG hydrolysis and fatty acid export
skeletal muscle and brain cells lack enough levels of
glucagon receptors (glucagon insensitive)
primary responses of insulin in liver (regulation of enzymes)
elevated expression of glucokinase to trap glucose into glycolysis or glycogen synthesis
activation of protein phosphatase 1 to dephosphorylate PFK-2/FBP-2 and activate PFK-1 through increased levels of fructose-2,6-bisphophate which inhibits fructose-1,6-bisphosphatase and gluconeogenesis
protein phosphatase 1 also stimulates glycogen synthase and inhibits phosphorylase kinase and glycogen phosphorylase
pyruvate dehydrogenase complex is activated to create more acetyl-CoA for fatty acid synthesis
protein phosphatase 2A is stimulated to activate acetyl-CoA carboxylase to synthesis malonyl-CoA from acetyl-CoA
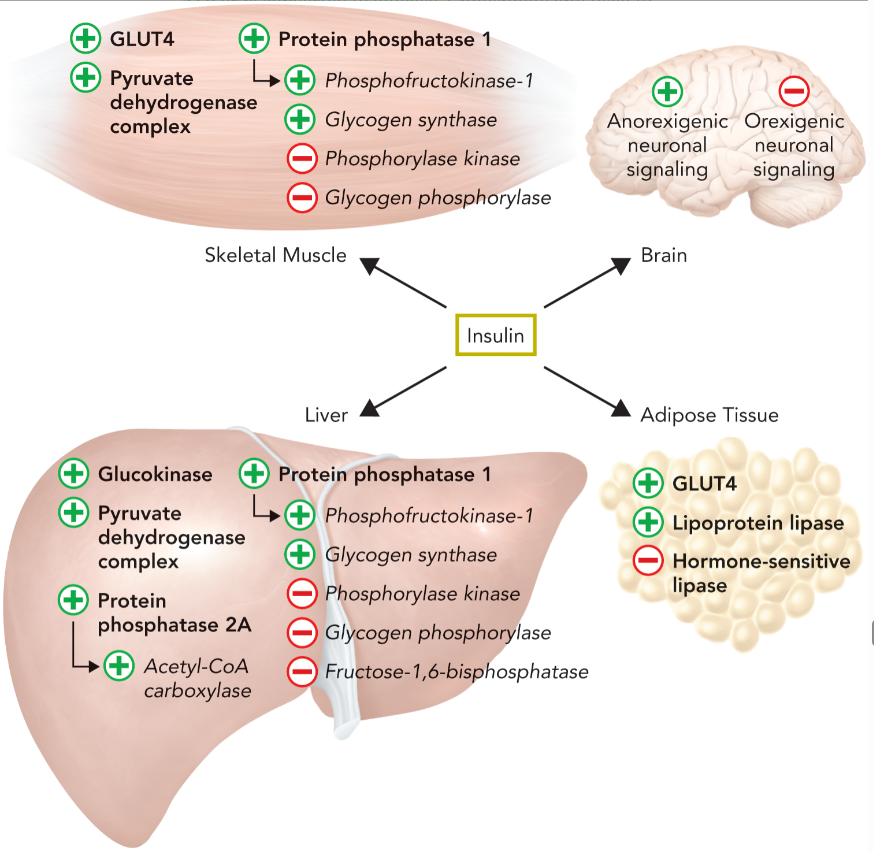
primary responses of insulin in skeletal muscle (regulation of enzymes)
activation of protein phosphatase 1 to dephosphorylate PFK-2/FBP-2 and activate PFK-1 through increased levels of fructose-2,6-bisphophate which inhibits fructose-1,6-bisphosphatase and gluconeogenesis
protein phosphatase 1 also stimulates glycogen synthase and inhibits phosphorylase kinase and glycogen phosphorylase
pyruvate dehydrogenase complex is activated (no fatty acid synthesis)
all glucose is used for metabolic fuel within the tissue; it cannot be exported or converted to fatty acids
primary response of insulin signaling in adipose tissue
lipoprotein lipase is stimulated to hydrolyze fatty acids from chylomicrons and VLDL particles to promote lipid uptake
hormone-sensitive lipase is inhibited to decrease fatty acid release from adipocytes
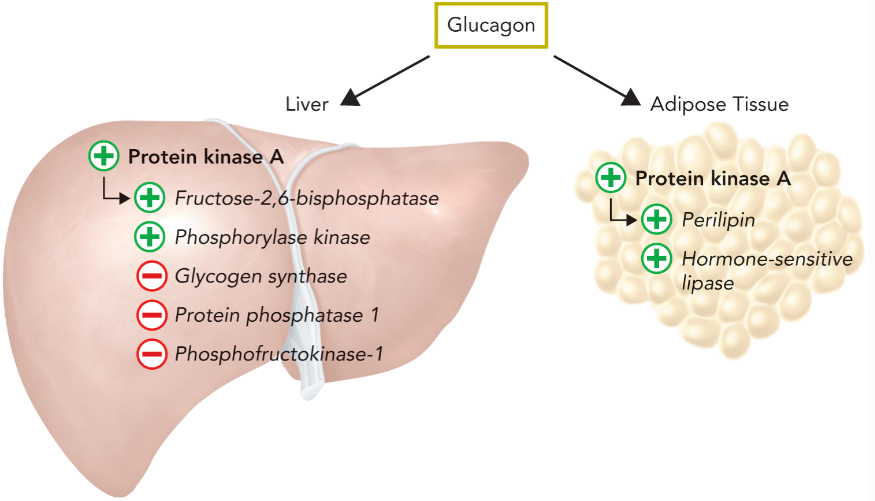
the glucagon receptor is a G-protein coupled receptor that stimulates
adenylate cyclase activity and production of cyclic AMP which activates PKA
primary response of glucagon signaling in the liver
PKA activates phosphorylase kinase to induce glycogen degradation (inactivates glycogen synthase and protein phosphatase 1)
PKA phosphorylates PFK-2/FBPase-2 which leads to decreased fructose-2,6-BP levels and increased flux through gluconeogenesis
primary response of glucagon signaling in adipose tissue
PKA phosphorylates perilipin and hormone-sensitive lipase to hydrolyze TAGs, they bind to serum albumin and and transported to tissues for use in energy conversion reactions; glycerol is metabolized by liver in glycolytic and gluconeogenic pathways
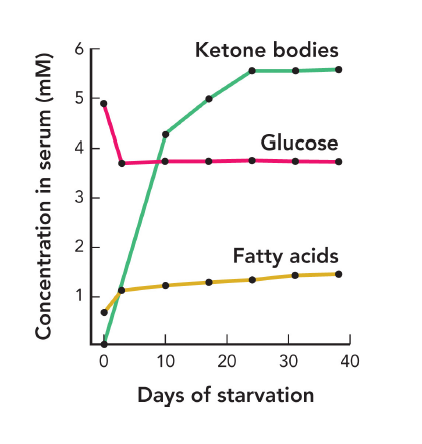
with the onset of starvation, blood glucose levels are initially maintained by
degradation of liver glycogen in response to glucagon signaling
during starvation metabolic flux is altered in two ways
gluconeogenesis is stimulated in the liver and kidneys to generate glucose for brain cells and erythrocytes
fatty acids are used as the primary metabolic fuel in almost all tissues
flux of metabolites between major tissues and organs during starvation
once glycogen stores are depleted, adipose and skeletal muscle are the primary sources of metabolic fuel during starvation
fatty acids released from TAG hydrolysis in adipose tissue are transported to skeletal muscle and the heart by serum album protein
fatty acids are used to generate acetyl-CoA for the citrate cycle and OXPHOS
acetyl-CoA produced from fatty acids in the liver is used for the production of ketone bodies, which are an important energy source for the heart and the brain during starvation
amino acids derived from protein degradation in skeletal muscle provide necessary carbon to make pyruvate or alpha-ketoglutarate (catabolism of skeletal muscle is delayed as long as possible)
alpha-ketoglutarate is used in the citrate cycle to make oxaloacetate which is then used in gluconeogenesis to make glucose
metabolic energy balance
calories consumed/ day (input) = calories expended/ day (output)
caloric input > output = positive energy imbalance
caloric input < output = negative energy imbalance
a balanced input/ output occurs at about ~1500-2000 calories/ day
metabolic syndrome
lifestyle of prolonged positive energy imbalance
abdominal obesity
insulin resistance
hypertension (high blood pressure)
hyperlipidemia (high LDL)
high risk for cardiovascular disease
type I diabetes
juvenile
insulin dependent
insufficient insulin production by pancreatic beta cells
type II diabetes
adult onset
insulin resistance
coupled to positive energy imbalance (weight gain)
desensitization of insulin receptor signaling
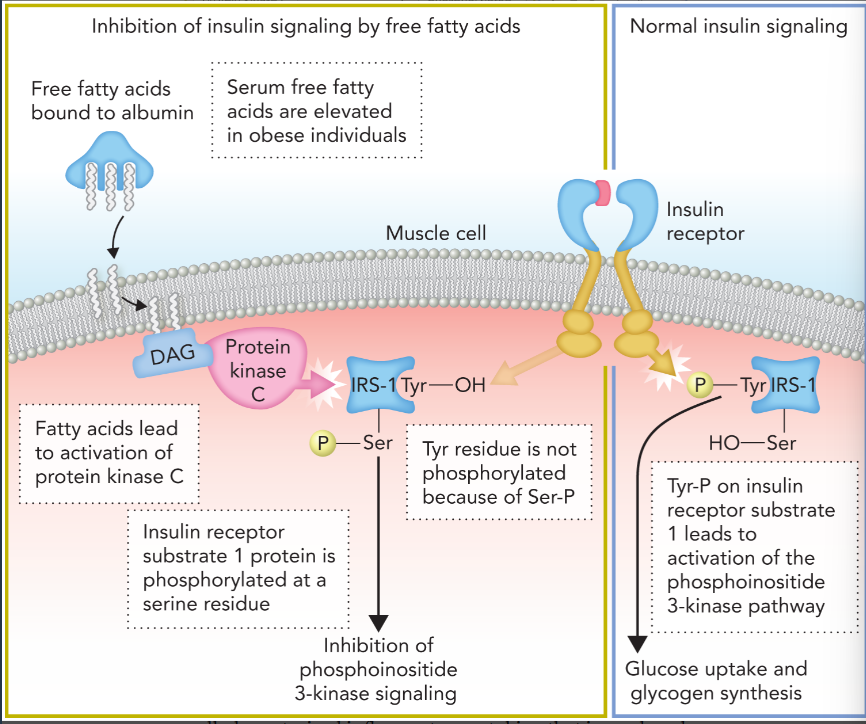
inhibition of insulin signaling by free fatty acids (insulin resistance)
increased levels of free fatty acids lead to the production of diacylglycerol (DAG) in the plasma membrane which stimulates protein kinase C
PKC phosphorylates insulin receptor substrate 1 (IRS-1) on serine residues, which inhibits the normal phosphorylation of IRS-1 tyrosine residues by the insulin receptor
results in lack of insulin signaling leading to high glucose levels in blood
tumor necrosis factor alpha is an
inflammatory cytokine that is produced at higher levels in adipocytes when lipid stores are high
autocrine signaling by TNF-alpha in adipocytes leads to
down regulation of genes involved in the uptake and storage of fatty acids which leads to increased levels of fatty acids in serum and interferes with insulin signaling in muscle
drugs that treat diabetes
alpha-glucosidase inhibitors (miglitol)
sulfonylurea drugs (glipizide), inhibit ATP-dependent K+ channel
drugs that stimulate AMPK (metformin)
ligand agonists of the nuclear receptor PPARgamma (thiazolidinediones)
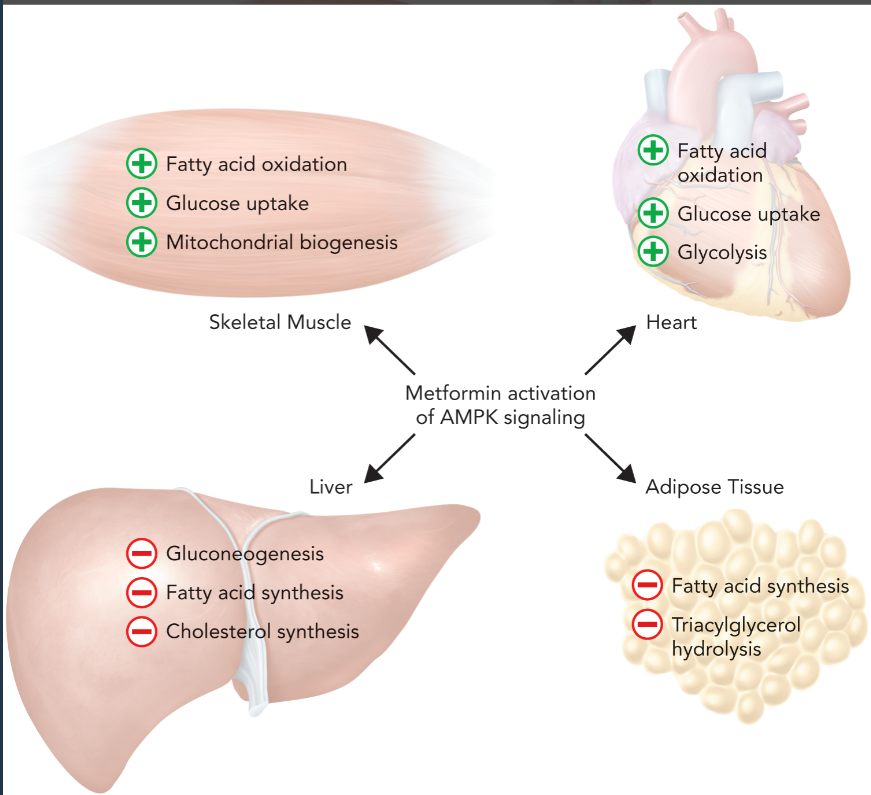
metformin mechanism
guanidine analog that improves sensitivity in multiple tissues by elevating AMP levels, which activates AMP-activated protein kinase (AMPK) signaling
increased levels of AMP in skeletal muscle leads to increased rates of fatty oxidation, glucose uptake, and mitochondrial biogenesis to raise ATP levels
in the heart, fatty acid oxidation, glucose uptake and metabolic flux through the glycolytic pathway is increased
inhibits metabolic flux through the gluconeogenic, fatty acid synthesis, and cholesterol synthesis pathways in the liver
inhibits the rates of fatty acid synthesis and TAG hydrolysis in adipocytes
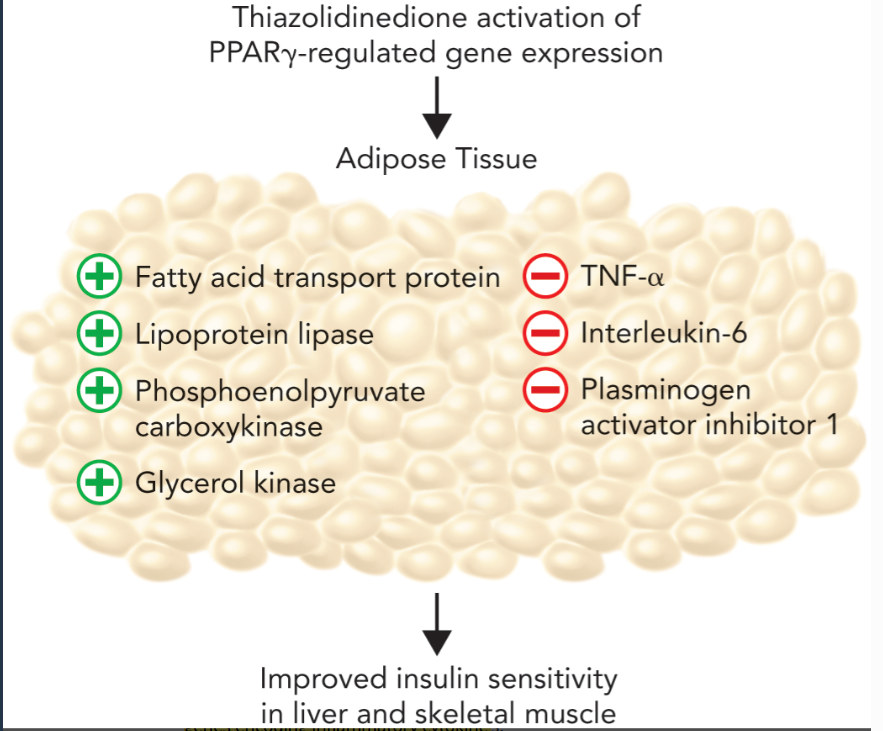
the insulin sensitizing activity of thiazolidinediones is due to
PPARgamma-mediated regulation of adipocyte gene expression which affects glucose and lipid metabolism in liver and muscle tissues
PPARgamma signaling
peroxisome proliferator-activated gamma (PPARgamma) nuclear receptors
PPARgamma induces expression of proteins (fatty acid transport protein and lipoprotein lipase) that inrcease the influx of fatty acids into adipocytes
increases expression of enzymes (phosphoenolpyruvate carboxykinase and glycerol kinase genes) that activate TAG synthesis through elevated levels of glycerol-3-phosphate
Thiazolidinedione-mediated activation of PPARgamma signaling in adipose tissue leads to down-regulation of inflammatory cytokines (TNF-alpha)
three primary factors influencing metabolic homeostasis
genetic inheritance
nutrition
exercise
glycemic index
a numerical value indicating how fast glucose is released into the blood after eating carb containing foods relative to the rise in blood glucose levels after drinking a solution of pure glucose
<55 healthy carbs
foods with a high glycemic index are associated with
a drop in blood glucose levels below the normal fasting state, which can leave one feeling lethargic and cause insulin spikes
running a 10 km race in an hour requires (endurance)
aerobic respiration using glucose derived from muscle glycogen and acetyl-CoA obtained from the degradation of fatty acids
a 200-meter sprint requires (short burst of activity)
phosphocreatine and anaerobic respiration of muscle glycogen as a source of ATP
ATP and phosphocreatine provide energy for muscle contraction for only
a few seconds
conversion of glucose to lactate can supply ATP for
several minutes