Chemistry In Bio Study Guide for 12/18 & 12/19
1/28
Earn XP
Description and Tags
First time making one of these so I may be cooked!!
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
29 Terms
What are the three major subatomic particles of an atom?
Protons, Neutrons, Electrons
What is a proton’s charge?
+1
What is an electron’s charge?
-1
What is an element’s atomic mass number symbolize?
the sum of the protons and the neutrons in the nucleus
What does an element’s atomic number symbolize?
the amount of protons in the nucleus
what is a valence shell?
the outer-most shell of an atom
What does a group of elements on the periodic table represent? (The groups he made us label 1-8)
elements with the same number of valence electrons, period have the same number of electron shells
What is an isotope? (in biological context)
atoms of the same element that differ in the number of neutrons
What is the octet rule?
the rule that applies that most elements in the periodic table are most stable when there are eight electrons in their valence shell.
which one is a Bohr model?
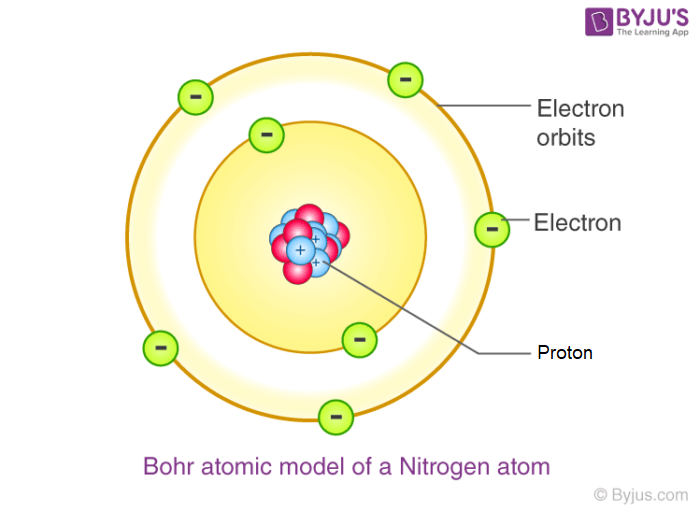
What is the difference between a molecule and a compound?
Molecule: two or more atoms bound together Compound: two or more elements bound together
What is an ion?
a charged particle
What is an ionic bond?
an attraction between negatively and positively charged atoms
What is a covalent bond?
when two atoms share electrons in such a way that each atom has an octet of electrons in the outer shell
What is electronegativity?
when one atom is able to attract electrons to a bigger degree than the other
What is a non-polar covalent bond?
when the sharing of electrons between two atoms is equal
What is a polar covalent bond?
when the sharing of atoms isn’t equal
How do you calculate the number of neutrons in an atom?
subtract the atomic number from the atomic mass number
How do you figure out the number of electrons in an atom? (neutral atoms)
it’s the same as the atomic number silly!
What is the duet rule?
the rule that states that light elements achieve stability with only two electrons in their valence shell
Where the non-metals attttt
WE ON THE RIGHTTTT
Where the metals attt
WE IN THE MIDDLE AND ON THE LEFT WOOOOOO
Al+3 + 3Cl-1 =
AlCl3
Be+2 + S-2 =
BeS
What charge does a cation have?
Positive
What charge does an anion have?
Negative
Why do atoms form ions?
to gain stability
Which elements are most likely to lose electrons to form positive ions, and which are most likely to gain electrons to form negative ions?
Metals would be more cations, nonmetals would be more anions
Which particles in an atom change during ion formation?
Protons and Electrons