Development
1/49
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
50 Terms
Gastrulation
outer layer
middle layer
inner layer
2. Gastrulation defines:
•Midline
•Anterior-Posterior axes
•Dorsal-Ventral axes
Initiation of neural development is dependent on ________
______-2 local invagination of a subset of cells in the very early
embryo; once complete the embryo consists of 3 layers of cells called
germ layers
• Ectoderm –
• Mesoderm –
• Endoderm –
______-3 defines:
______
_____ axes
_____axes
Notochord
primitive streak
Neuroectoderm
________forms at the midline of the gastrulating embryo; this is a central event for the development of the nervous system
generated by the _______
Define the embyonic midline and the axis of symmetry for the entire body
________- lies immediately above the notochord; gives rise to the entire nervous systems
________2- send inductive signals to the overlying ectoderm to cause a subset of cells to differentiate into neuroectodermal precursor cells
neuroectodermal precursor cells
Notochord sends inductive signals to the overlying ectoderm to cause a
subset of cells to differentiate into _________
Neurulation
neural plate
neural tube
• _____forms as a result of thickening of the neural ectoderm
• _____forms as a result of an infolding of the neural plate
• gives rise to the brain, spinal cord and most of the PNS
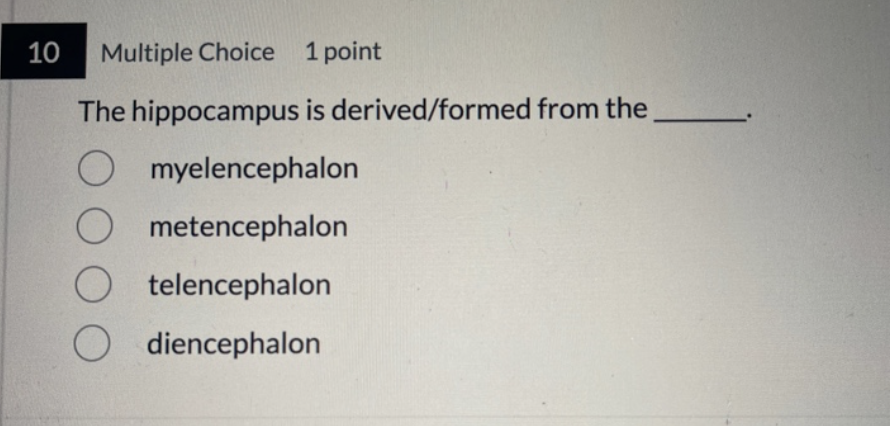
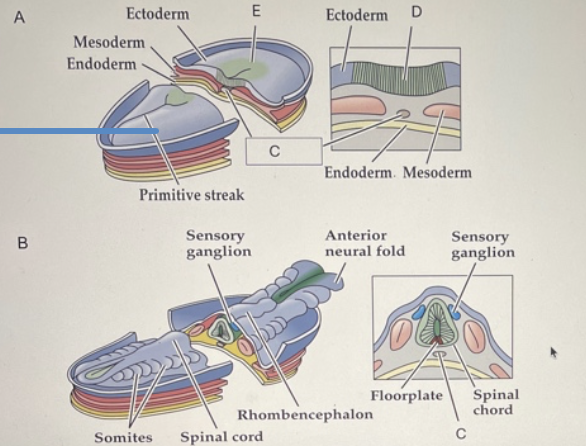
(a) late gastrulation and early neuralation, the notochord forms by Invagination of the mesoderm in the region of the primitive streak. The ectoderm overlying the notochord becomes defined as the neural plate.
Nuerlation in the mammalian embryo part 1
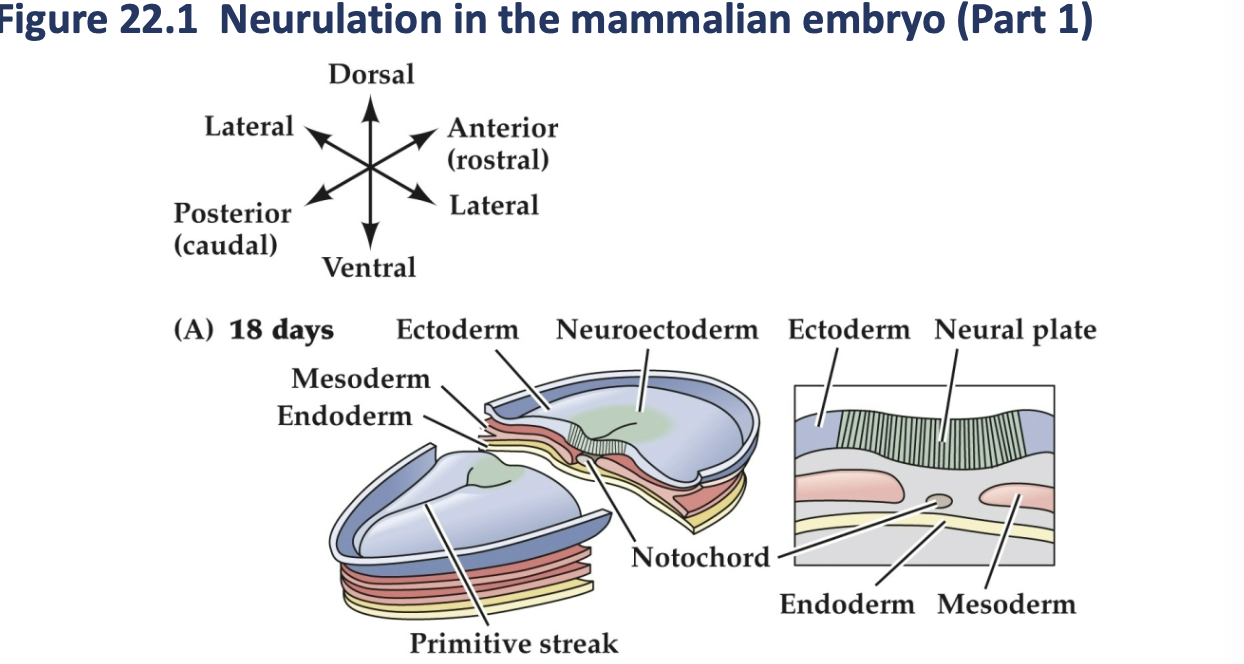
as neuralation proceed, the neural plate begins to fold at the midline (adjacent to the notochord (forming the neural groove and ultimately, the neural tube. The neural plate immediately above the notochord differentiates into the floor plate, whereas the neural crest emerges at the lateral margins of the neural plate (farthest firm the notochord).
Neuralation in the mammalian embryo (part 2)
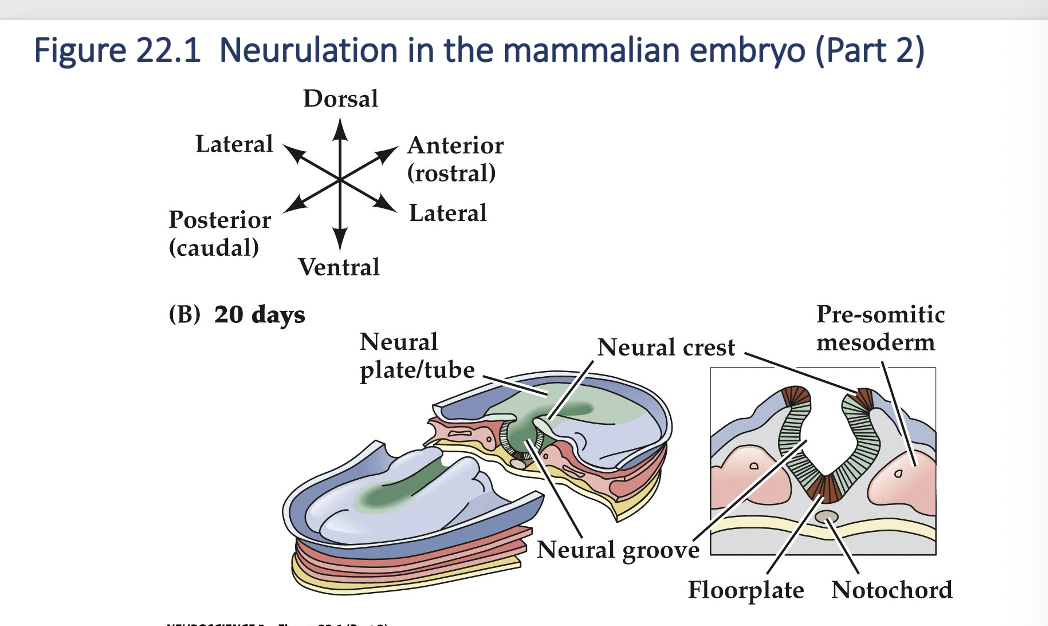
Once the edges of the neural plate meet in the midline, neural tube is complete. The mesoderm adjacent tot the tube them thickens and subdivides into structures called somites - the precursors of the axial musculature and skeleton.
Neurulation in the mammalian embryo (Part 3)
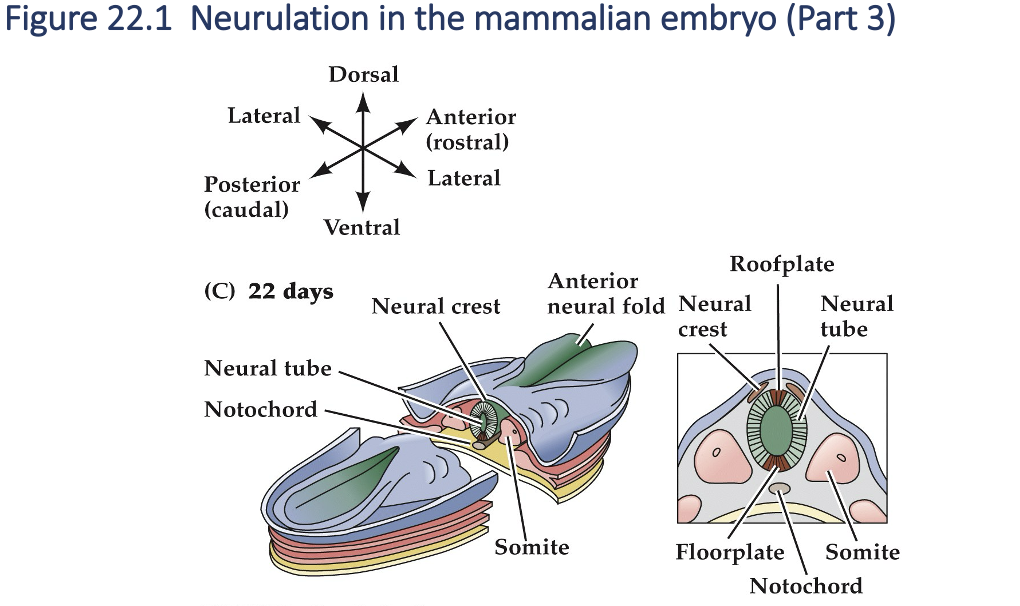
As development continues, the neural tube adjacent to the somites becomes the rudimentary spinal cord, and the neural crest gives rise to sensory and autonomic ganglia( the majors elements of the peripheral nervous systems). Finally, the anterior end of the neural plate (anterior neural folds) grow together at the midline and continue to expand, eventually giving rise to the brain.
Neurulation in the mammalian embryo (Part 4)
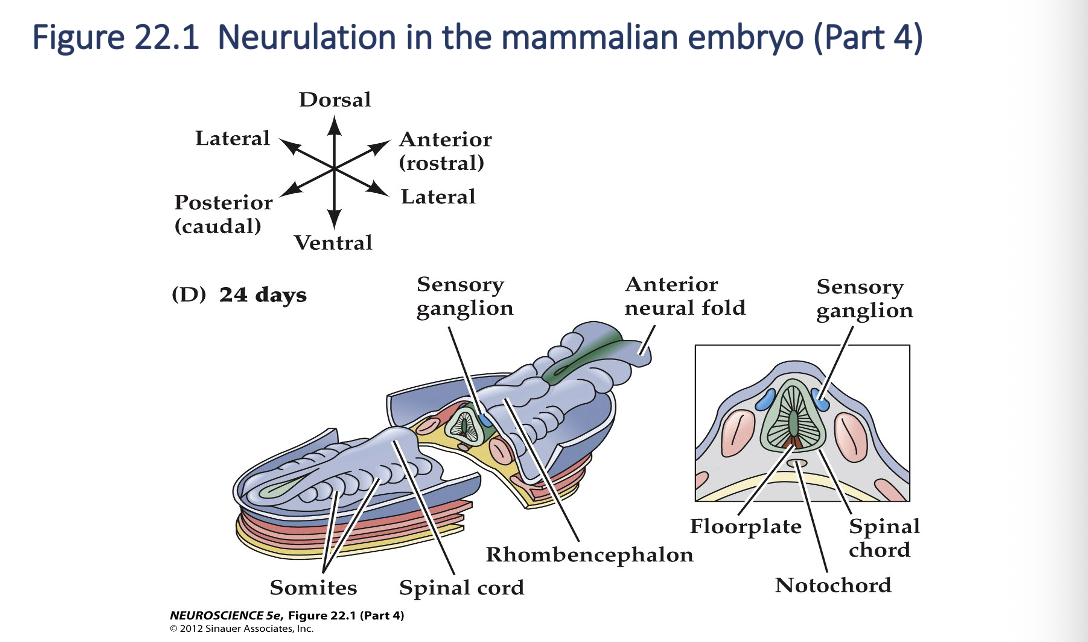
Neurulation
neural stem cells
floor plate
roof plate
notochord, floorplate, and roofplate
Nueroectodermal precursor cells = _______
____2- divide to produce more precursor cells
have the capacity to differentiate into any type of cell found in nervous tissue i.e Astrocytes, microglia, neurons etc.
some cells of the neural tube differentiate into specialized cells that form the _________.
molecular signals from the _____2 and the notochord specify the position and fate of the spinal cord and hindbrain.
precursors cells further away form the ventral midline rise to neurons in more dorsal regions of the spinal cord and hindbrain; facilitated by the _______.
the _______, floor plate and roof plate are transient structures that provide signals to the developing neural tube but disappear once initial nervous system development is complete.
Neural crest cells
Arise from the lateral edge of the neural plate as the neural tube forms.
migrate away form the neural tube through an extracellular matrix of mesenchymal cells.
Give rise to a variety of progeny (not CNS: portions of the PNS)
neurons and glia of the sensory and autonomic ganglia
neuronsecretory cells of the adrenal gland
neurons of the enteric nervous system
non- neuronal structures - pigment cells beneath the epidermis, cartilage and bone (partoculary of the face and skull)
Neural crest cells populations
Cranial -
Vagal-
Trunk -
Sacral -
____- ____ sensory ganglion, non neuronal cells that become the ___skeleton, thyroid gland, and teeth.
____- Enteric nervous system
____- Dorsal root ganglion
____- Additional neurons of the enteric nervous system and posterior ganglion of the sympathetic chain
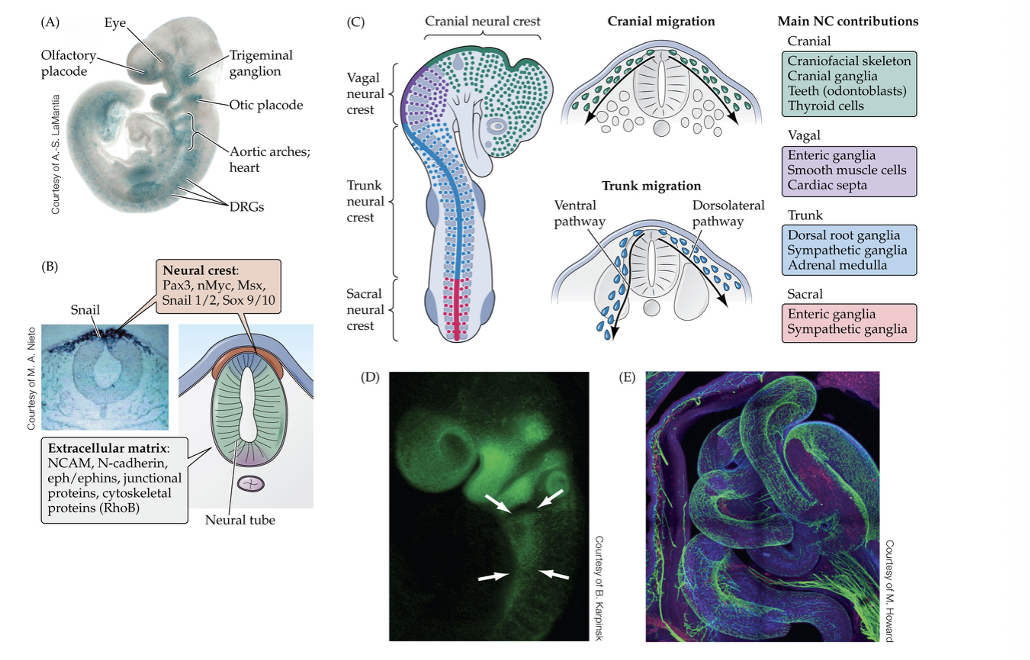
(a) this mid-gestation mouse embryo carries a reporter trangene that visualizes the migrating neural crest cell (blue) as they begin to accumulate in or near regions where senroy structures or major cardiac blood vessels will differentiate.
(b) the location of origin of the neural crest at the dorsal midline of the neural tube. This region is distinguished by a set of unique molecular determinates, including the transcription of the adhesion molecules listed at the bottom.
( c) Anterior posterior organization of four population of neural crest cells based on their location in the neural tube, and the gives rise to after the nerual creat cell have migrated to cranial, vagal, trunk, and sacral regions of the embryo.
(d) mid gestation mouse embryo carrying a reporter transgene that visualizes the mrigrating vagak neural crest.
(e) the vagal neural crest forms the enteric nervous system, which consists of a network of neurons and small ganglia
Neural crest in developing embryo
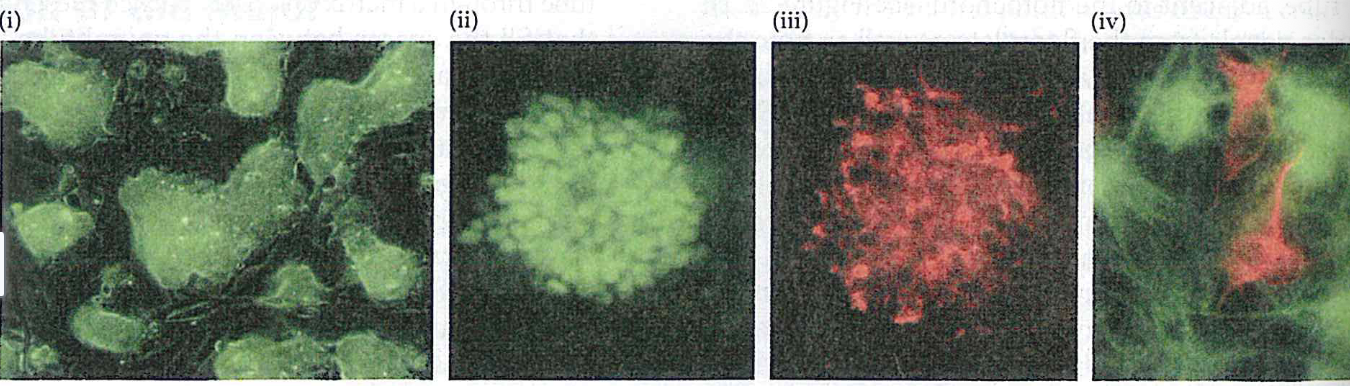
(a) - (i) colonies prior differentiation
(ii,III) after exposure to neutralizing signals SOx(green) nestin(red)
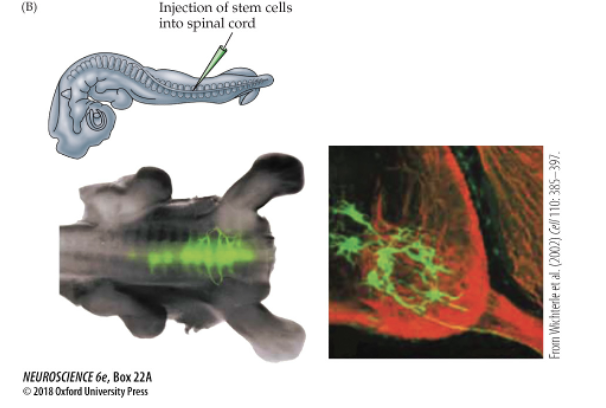
(iv) after sveral days, both nuerons generate ES cells
(b) Injection of fluorescently labeled embryonic stem cells into the spinal cord of a host chicken embryo shows that ES cells integrate into the host spinal cord and apparently extend axons.
embryonic stem cells stem types
Neural stem cell
Neural progenitor cell
Embryonic S cell (embryonic stem cell)
IPSC
difference between an embryonic stem cell and a neual stem cell
embryonic stem; Neural stem cells
Why neural stem cell transplants into the adult brain are often unsuccessful
environment
___- self renewing cell that can produce neurons, astrocytes, and oligodendrocytes.
___- a more limited cell that can only produce certain neural cell types and cannot self-renew continuously
___- A pluripotent stem cell form embryos that can become almost any cell type of the body
_____ cells can become nearly any cell type in the body, while ______ are limited to producing cells of the nervous system.
transplanted neural stem cells often fail because adult brain does not provide the proper signals and _____ needed for the cell to survive and differentiate correctly.
prosencephalon
mesencephalon
rhombencephalon
Following the formation of the neural tube, beginning of the major brain regions start to appear
bending, folding, and constriction of the neural tube
Initial subdivision of the neural tube- primitive brain regions
__- forebrain
__- midbrain
__- hindbrain
Early in gestation the neural tube becomes subdivided into the prosencephalon, mesencephalon, rhombencephalon. Initial bending of the neural tube at its anterior end leads to a cane shape showing major brain regions,
Figure 22.3 Regional specification of the developing brain (Part 1)
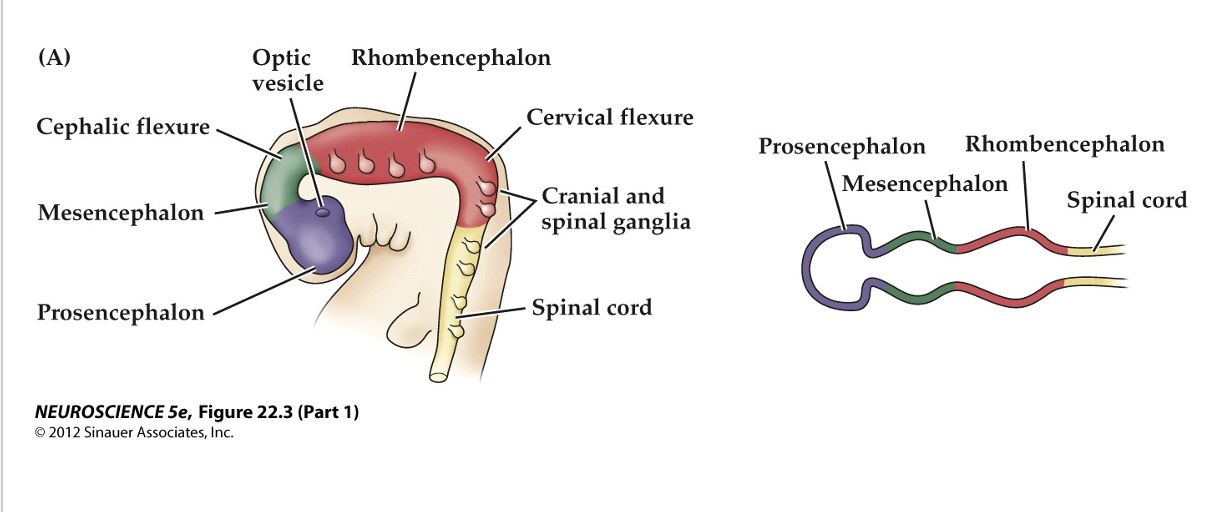
Further development distinguished the telencephalon and dicenpahlon from the prosencephalon; two other subdivisions the mesencephalon and myelencephalon
prosencephalon
telencephalon- cerebral cortex, hippocampus and basal ganglia
diencephalon - thalamus anf hypothalamus; optic cups -retina
Mesencephalon - superior and inferior colliculi and midbrain regmentum (ventrak portion, substantia nigria)
Rhombencephalon
metencephalon -cellbellum and pon
mylencephalon- medulla
Figure 22.3 Regional specification of the developing brain (Part 2)
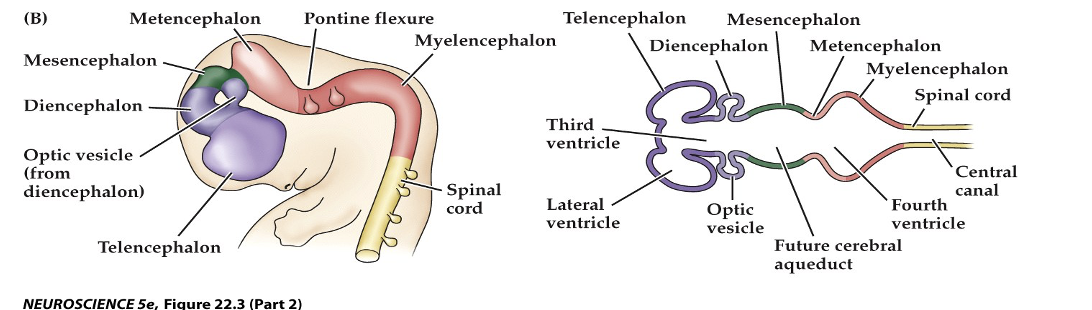
The fetal brain and spinal cord are clearly differentiated by the end of the second trimester. Several major subdivisions, including the cerebral cortex and cerebellum, are clearly seen from the lateral surfaces. forebrain at the level indicated showing the nascent sulci and gyri fo cerebral cortex.
Figure 22.3 Regional specification of the developing brain (Part 3)
•How do neural stem cells know what cell type to differentiate into?
early experiments
Neural induction
•precursor cells moved to a different regions, results differ depending on timing and location;Acquire the identity of the new region (receive instructions based on new location);Retain an identity that reflects their original position; Cells that are removed are compensated by local cell proliferation – little disruption to development; Absence of cells disrupts subsequent development’ Relocation of cells causes a complete change in the local developmental program’ Suggested that neural induction relies on signals provided by adjacent cells or tissues – not proven until the 1990’s
molecular signals are secretes by one embryonic cells class and tissue and then diffuse and act on adjacent cell class or tissue (roofplate, floor plate)
graded effects - based on distance from source of signal
more specific effects- at boundaries between distinct cell populations
_______ - gene expression, shape and motility are changed in target cell safter these inductive signals bind to their receptors
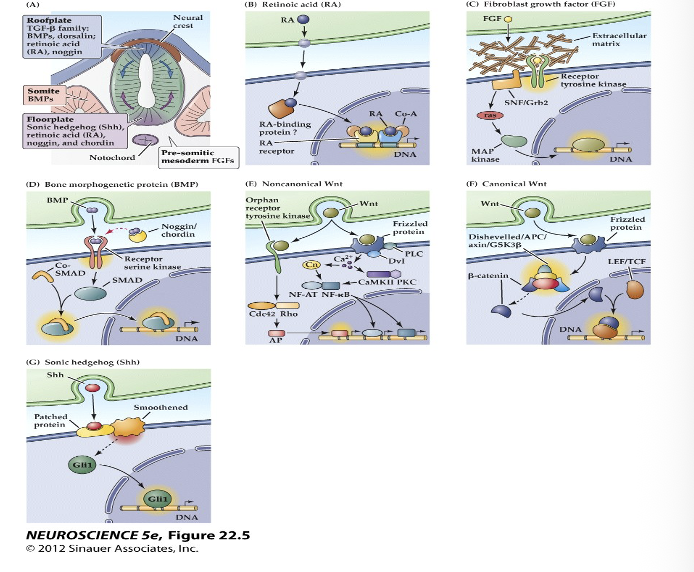
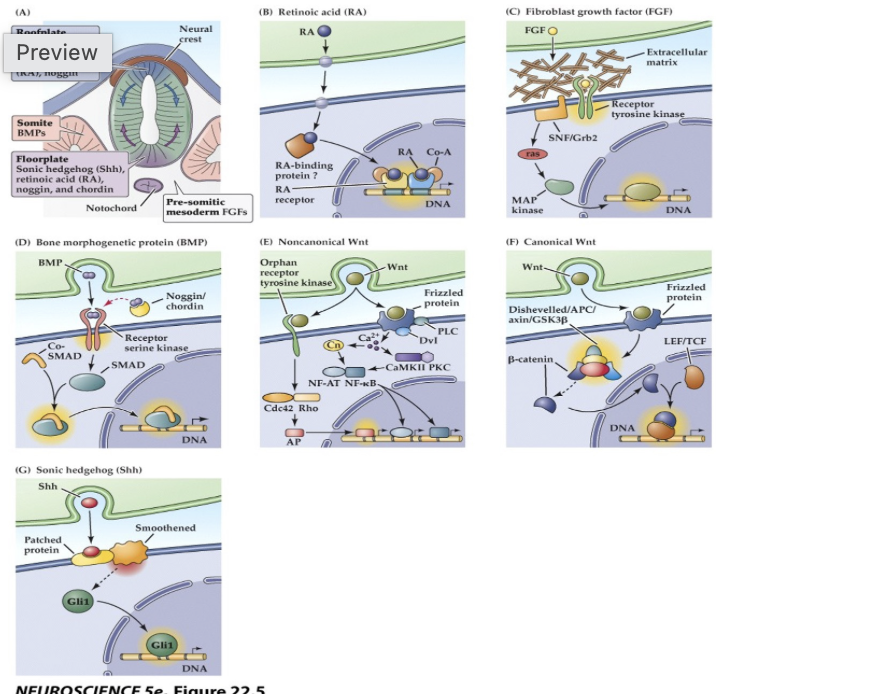
(A) The embryonic notochord, floorplate, and neural ectoderm, as well as adjacent tissues such as somites, produce the molecular signals that induce cell and tissue differentiation in the vertebrate embryo.
(B–G) Schematics of ligands, receptors, and primary intracellular signaling molecules for retinoic acid (RA); members of the FGF and TGF-â (BMP) superfamilies of peptide hormones; the Wnt family of signals; and Sonic hedgehog (Shh). Each of these pathways contributes to the initial establishment of the neural ectoderm, as well as to the subsequent differentiation of distinct classes of neurons and glia throughout the brain.
Major inductive signaling pathways in vertebrate embryos
Neurogenesis - production of new neurons
mature braon contain 100 billion neurons and many more glial cells
______- begins after the initial patterning of the brain is complete
precursor cells are located in the ventricular zone (innermost cell layer surrounding the lumen of the neural tube
All neurons are produced before brith, after birth precursors will disappear and no new neurons will be produced to replace those lost due to age, injury or disease
exception is adult neurogenesis
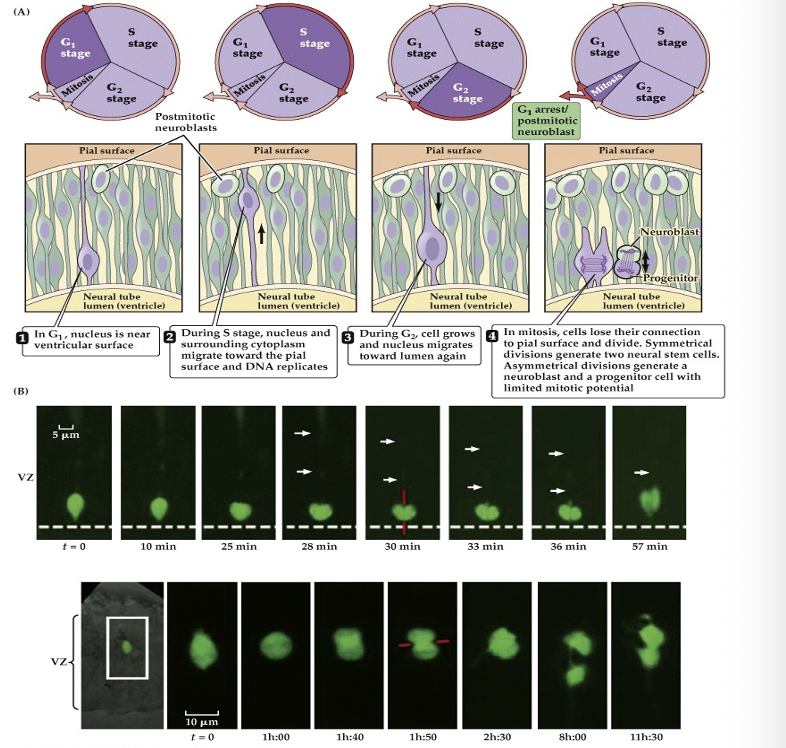
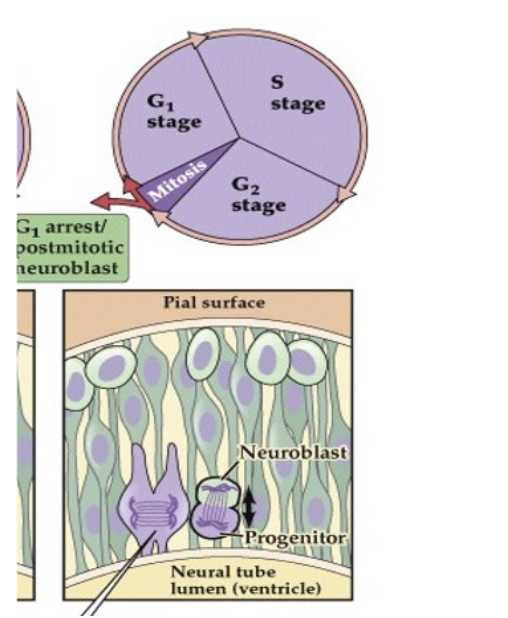
(A) Precursor cells in the vertebrate neuroepithelium are attached both to the pial (outside) surface of the neural tube and to its ventricular (lumenal) surface. The nucleus of the cell translocates between these limits within a narrow cylinder of cytoplasm (the ventricular zone, VZ). When cells are closest to the outer surface of the tube, they enter the DNA synthesis phase (S stage) of the cell cycle. Once the nucleus moves back to the ventricular surface (G2 stage), the precursor cells lose their connection to the outer surface and enter mitosis. When mitosis is complete, the two daughter cells extend processes back to the outer surface of the neural tube, and the new precursor cells enter a resting (G1) phase of the cell cycle. At some point a precursor cell generates either another progenitor cell that will go on dividing and a daughter cell—a neuroblast—that will not divide further, or two postmitotic daughter cells.
(B) Time-lapse microscopy permits visualization of symmetrical, vertically oriented division (red line) of a single radial glial stem cell in the cortex. The cell body is seen at the ventricular surface (dashed line); the arrows indicate the radially oriented process of the cell, which is mostly out of the focal plane necessary for visualizing the cell body. The radial processes are retained once the cell has divided.
symmetrical division- Produces two new neural stem cells to expand the precursor pool.
asymmetric - produces one neuroblast (which will become a neuron) and one progenitor cell with limited mitotic potential.
22.8 Neural precursor cells undergo mitosis in the ventricular zone.
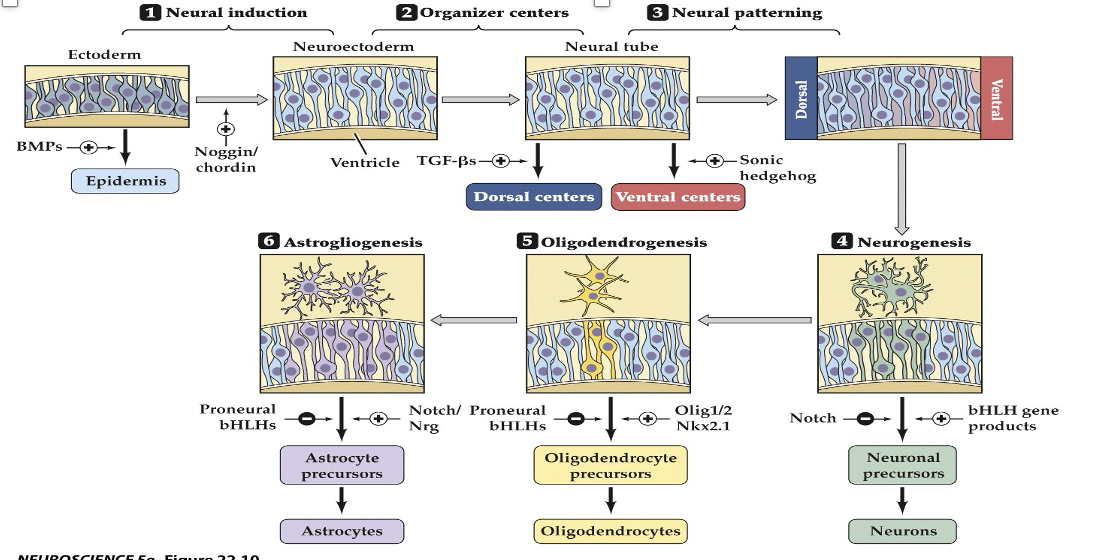
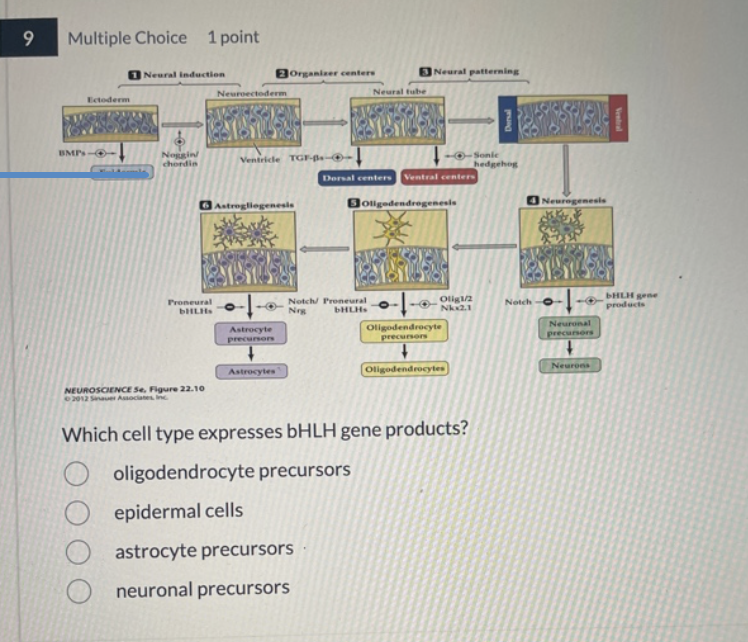
1–3) The steps by which ectoderm acquires its identity as neural ectoderm. Generation of neural precursors, or stem cells, relies first on the balance of BMP and its endogenous antagonists (e.g., Noggin and Chordin) in the developing embryo. Next, local sources of inductive signals, including TFG-â family members and Sonic hedgehog, establish gradients that influence subsequent neural precursor identities, as well as identifying local “organizers” (such as the floorplate and roofplate) that define the cellular identity of the inductive signaling centers. (4–6) Steps thought to define neurons, oligodendroglia, and astrocytes from multipotent neuronal precursors. Balanced signaling activity of Notch and transcriptional control of the bHLH proneural genes (named based on their ability to bias neural progenitor cells toward a differentiated neural fate) influence neurogenesis. Similarly, antagonistic transcriptional regulation via either the products of the bHLH genes or three additional transcription factors—Olig1, Olig2, and Nkx2.1—influences the generation of oligodendroglia. Continued antagonism between bHLH proteins, Notch signaling proteins, and the signaling molecule neuregulin (Nrg) is thought to influence the generation of mature astrocytes. Finally, in the adult brain, cells adjacent to the ventricles (which apparently have avoided becoming differentiated) remain as ependymal cells
22.11 Molecular and cellular mechanisms that guide neuronal and glial differentiation
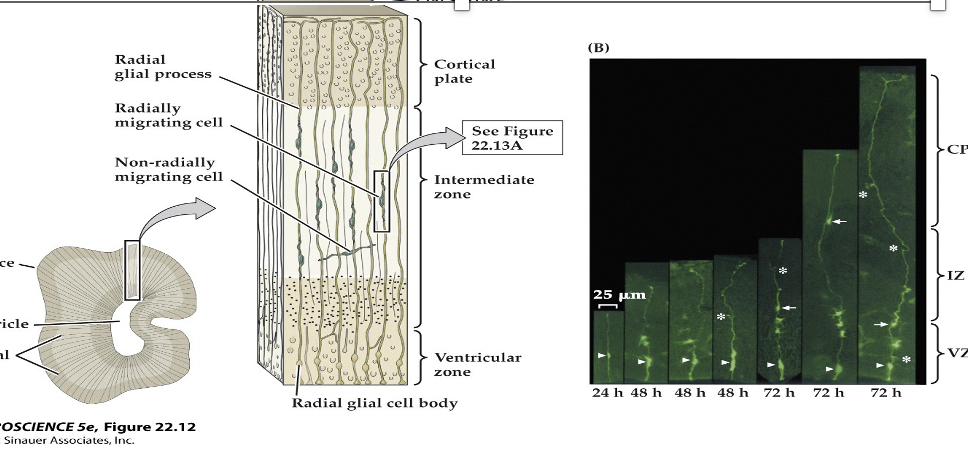
(A) Section through the developing forebrain showing radial glial processes from the ventricular to the pial surfaces. Migrating neurons are intimately apposed to radial glial cells, which guide them to their final position in the cortex. Some cells take a nonradial migratory route, which can lead to wide dispersion of neurons derived from the same precursor.
(B) Time-lapse micrography showing radially migrating (arrows) and nonradially migrating (asterisks) neurons.
22.13 Radial glia guide migrating neurons
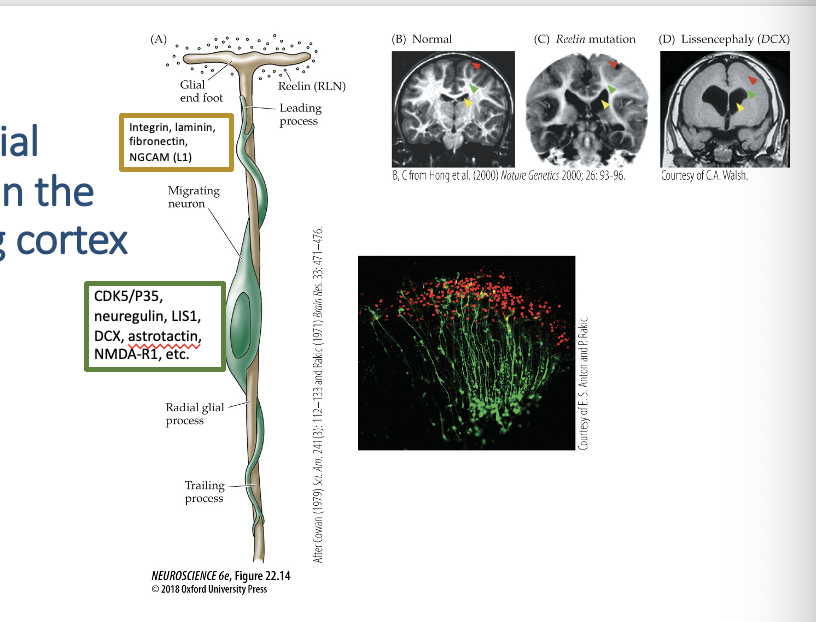
(A) A single neuroblast migrates on a radial glial process (based on serial reconstruction of EM sections as well as in vitro assays of migration). Cell adhesion and other signaling molecules or receptors found on the surface of either the neuron (green) or the radial glial process (tan) are indicated in the respective boxes.
The micrograph shows migrating neurons labeled with an antibody to neuregulin, a protein specific to migrating cortical neurons.
(B–D) Mutations in genes that influence neuronal migration cause malformations of the human cerebral cortex. In these MRI images, yellow arrows point to the lateral ventricle, green arrows indicate the subcortical white matter in the internal capsule (a “thoroughfare” for axons entering and exiting the cerebral cortex), and red arrows highlight the typical appearance of sulci and gyri.
(B) Typical cerebral cortex. (normal)
(C) Individual with a mutation of the gene encoding Reelin, a protein that influences radial neuronal migration in the cortex. The lateral ventricles are enlarged, subcortical white matter is diminished, and the pattern of sulci and gyri is disrupted.
(D) In an individual with a mutation in the DCX gene, the ventricles are dramatically enlarged, subcortical white matter is nearly absent, and sulci and gyri are completely absent. This dramatic cortical malformation is known clinically as lissencephaly (“smooth brain”).
22.14 Radial migration in the developing cortex
•Spina Bifida (spinal cord does not develop normally)
•Anencephaly (missing parts of the brain and skull)
_____– failure of the neural tube to close completely – insufficient intake of folic acid
______– failure of the neural tube to close at all
•Exogenous exposure to retinoids (teratogens)
•Accutane (acne treatment, 1980’s) – increase in spontaneous abortions and birth defects
•Retinoic acid – important for proper development of the nervous system; alters gene expression of SHH (Sonic Hedgehog)
•Mutations in human genes for SHH or related receptors are associated with serious disorders
SHH sonic hedgehog
is a signaling protein for brain hemisphere formation, cell proliferation, neural pattering
disorder linked to () are
_____- failure of forebrain to split into two hemisphere; can cause cyclopia (single eye)
_____- childhood cerebellar tumor; caused by uncontrolled granule cell precursor proliferation
_____- skin cancer associated with () pathways mutations
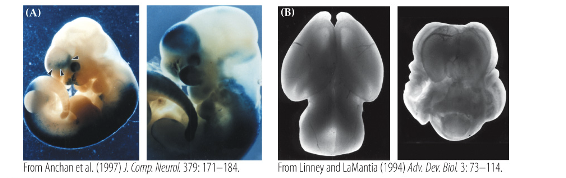
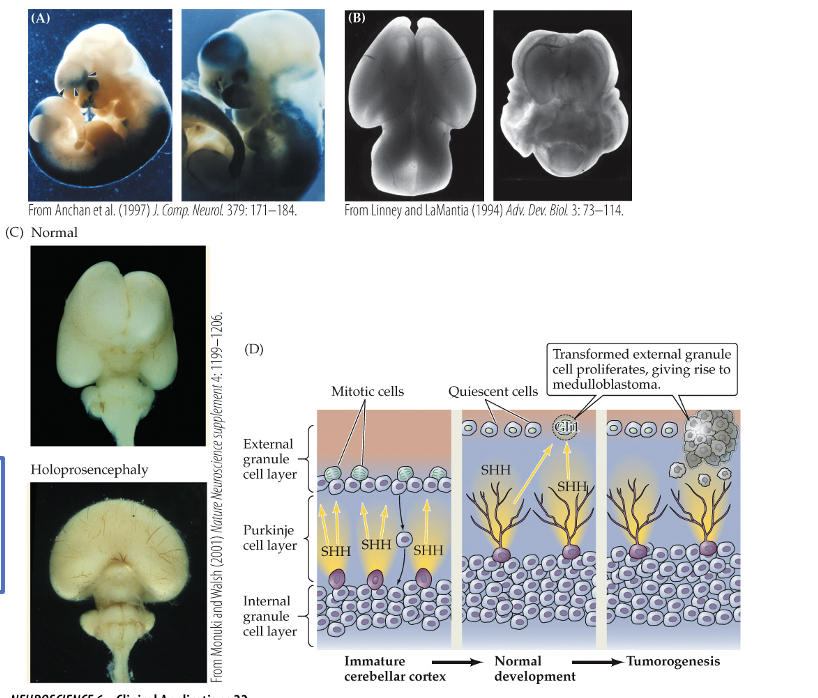
Holoprosencephaly
•disrupted regional differentiation of the forebrain, improper development of cerebral hemispheres (incomplete seration
•1 in 16,000 live births and most common malformation of the forebrain known
•Cyclopia – development of a single eye
•Range of severity from mild to still birth – responsible for 1 in 250 still births
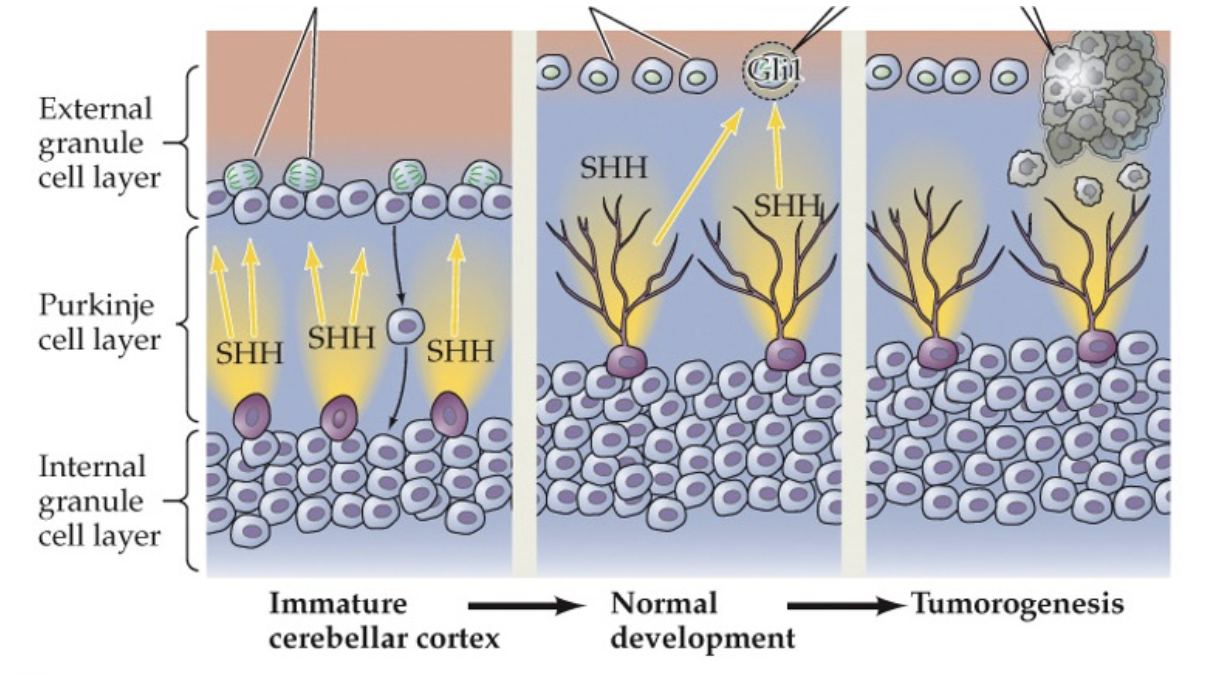
•Medulloblastoma
•Result of cancerous transformation of cerebellar granule neuron precursors (tumor)
•Most common childhood brain tumor; 60 % survival rate
•Rare – between 1 in 50,000/100,000 births
•Basal cell carcinoma
•Most prevalent form of skin cancer
(a) excess retinoic acid exposure during pregnancy causes abnormal gene activation and disrupts normal embryonic brain development
(b) retinoic acid exposure leads to severe forebrain malformations compare with the normal brain
(c ) normal brain develops two separate hemisphere, holoprosecephaly results from failed forebrain separation caused by defective SHH signaling
(d) SHH released y purkinje cells normally control granule cell precursor division, but uncontrolled SHH pathway activity causes SHSH pathway activity cause proliferation and medullaoblastoma formation.
Inductive Signals and Neurodevelopmental Disorders
BMP (Bone Morphogenetic Protein):
Influences the differentiation of the ectoderm.
Sonic Hedgehog (SHH):
Retinoic Acid (RA):
______- Secreted by the notochord and floorplate; critical for ventral patterning.
______ - A derivative of Vitamin A that regulates gene expression, including SHH.
cell proliferation
cell migration
process outgrowth
_____- process by which cells divide to produce more cells through mitosis. In neural development, it increases the number of neural precursor cells.
_____ - the movement of newly formed cell from their birthplace to their final location in the nervous system
_____ - The extension of axons and dendrites from neuron so they can form connections with target cells and establish neural circuits.
Embryonic Stem Cell
Neural Stem Cell
pluripotent
can becomes almost any cell type in the body
found in early embryo
less specialized
mutipotent
can only become nervous system cells
found in developing or adult nervous systems
Neural stem cell transportation into the adult brain is unsuccessful because
the adult brain environment lacks developmental guidance signals
damages tissue may contain inhibitory molecules
transplanted cells may not migrate properly
cells mat fail to form correct synaptic connections
limited trophic support reduces connections
The adult CNS is much less permissive for growth and circuit formation than the developing brain.
Prosencephalon (forebrain),
Mesencephalon (midbrain)
Rhombencephalon (hindbrain).
A. Telencephalon and Diencephalon Derivation:
These are derived from the Prosencephalon.
B. Metencephalon and Myelencephalon Derivation:
These are derived from the Rhombencephalon.
C. Telencephalon Mature Structures:
The mature structures derived from the telencephalon include the cerebral cortex, hippocampus, and basal ganglia.
1) List the three primary brain vesicles or divisions that appear first during development. As neural development progresses,
these three primary brain vesicles divide further.
A. From which primary brain vesicle are the telencephalon and diencephalon derived?
B. From which primary brain vesicle are the metencephalon and myelencephalon derived?
C. What mature brain structures are derived from the telencephalon?
In panel D of figure 22.5 on slide 18, the binding of BMP to its receptor triggers intracellular effects in the target cell that
ultimately lead to SMAD binding to DNA. What is the outcome of this process, and why is it significant? Additionally,
what is the result of SMAD binding to the DNA?
BMP binding activates SMAD proteins, which enter the nucleus and bind DNA to regulate gene transcription. This controls cell differentiation and tissue patterning during neural development
neural induction. While BMPs generally specify the ectoderm to become epidermis, the inhibition of this pathway by signals like Noggin or Chordin allows the ectoderm to become neuroectoderm.
Result of SMAD Binding: Once BMP binds to its receptor (a receptor serine kinase), it phosphorylates SMAD proteins. These SMADs then translocate to the nucleus and bind to DNA to regulate gene expression, which ultimately changes the target cell's identity, shape, and motility.
Refer to Figure 22.11 on slide 21 to answer the following questions:
A. Which signal induces neuronal precursors to differentiate into neurons?
B. Identify the signal that prompts oligodendrocyte precursors to develop into oligodendrocytes.
C. Indicate the signal that inhibits neuronal precursors from becoming neurons.
A. Signal inducing neurons: bHLH gene products induce neuronal precursors to differentiate into neurons.
B. Signal inducing oligodendrocytes: Olig1/2 and Nkx2.1 are the signals that prompt oligodendrocyte precursors to develop into oligodendrocytes.
C. Signal inhibiting neurons: Notch signaling inhibits neuronal precursors from becoming neurons, often biasing them toward a glial fate instead.
altering transcription of specific genes
All of the molecular signals shown in this diagram induce neuronal precursor cells to differentiate by _________.
medulloblastoma
the figure above demonstrates the pathogenic process that occurs in
neuroblasts; progenitors.
After asymmetric divison, a progenitor cell and neuroblast are formed; ______will begin to differentiate into neurons, while ___ have the ability to divide again
cell differentiation.
Neural stem cells will express specific genes, extend processes and develop into mature neurons we call this process ______
gastrulation.
the inaitation of nueronal devleopment is dependant on

neural plate.
during nuralation the ___forms as a result of a thickening of the neural ectoderm

diencephalon.
all of the following are initial subdivision of the neuronal tube except

notochord.
Which panel in the figure above is in the earliest stage of neurulation? Which letter correctly identifies the transient structure that provides signals to the developing neural tube?
neuronal precursors.
Which cell type expresses bHLH gene products?
telencephalon.
Diencephalon: Gives rise to the thalamus, hypothalamus, and optic cups (retina).
Metencephalon: Gives rise to the cerebellum and pons.
Myelencephalon: Gives rise to the medulla.
the hippocampus is derived formed from the