3.1.1.3 - Electron Configuration
1/4
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
5 Terms
Define ionisation energy
What is the general trend along period 3 for ionisation energy and why does this occur?
Elements increase in ionisation energy due to:
Greater nuclear charge
Smaller atomic radius
What are the two elements that deviate from the general trend along period 3?
Aluminium
Sulfur
Why do the two elements deviate from the general trend along period 3?
Al - Aluminium's outer electron is in a 3p orbital rather than a 3s. The 3p electron is slightly more distant from the nucleus than the 3s therefore requires less energy to remove
S - Sulfur’s outer electron is being removed from a pair in an orbital. The repulsion between the two electrons in the same orbital means that the electron is easier to remove
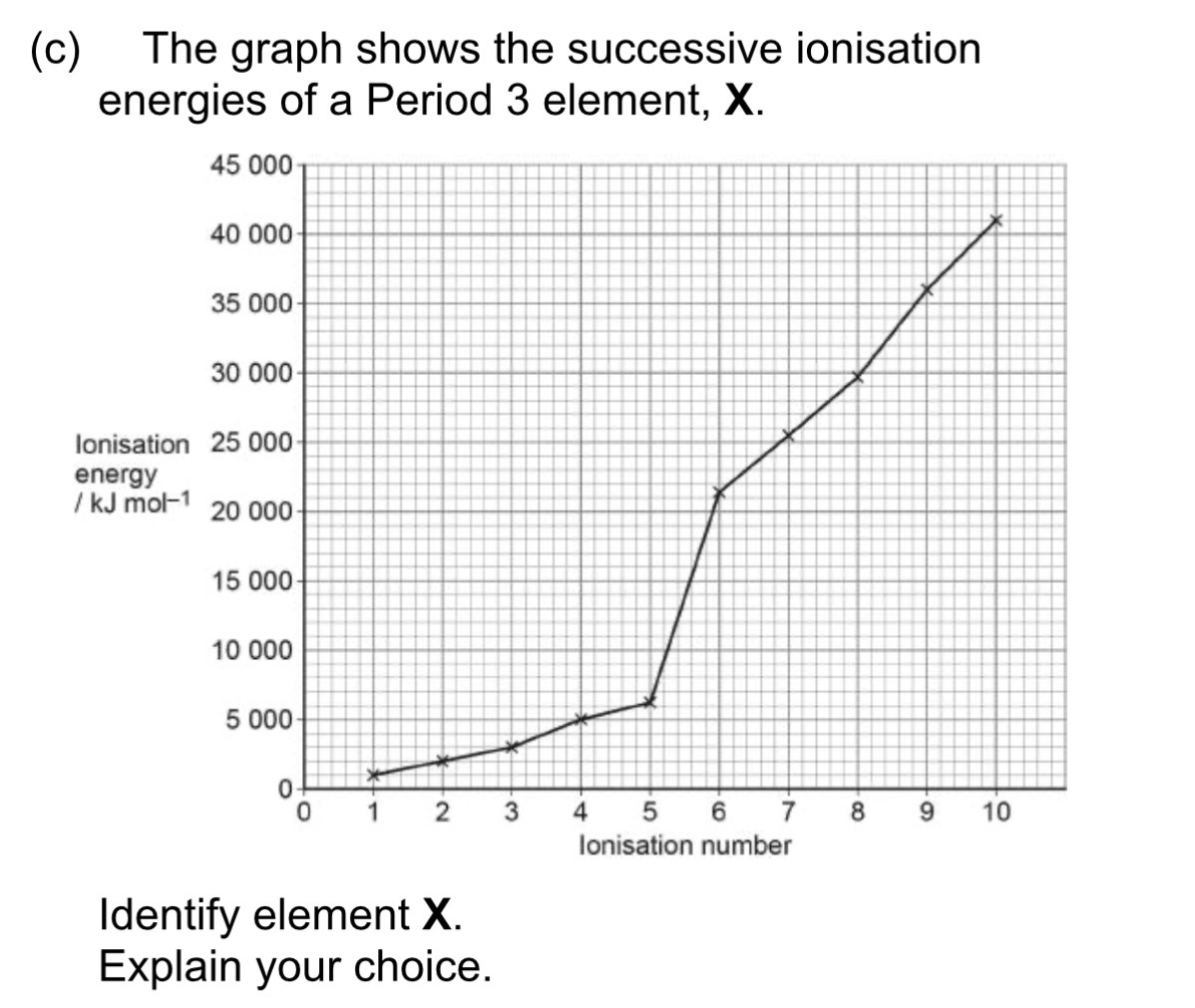
Element - Phosphorus (1)
Explanation - Large jump in ionisation energy for the 6th ionisation energy (1) due to the electron being removed from a shell closer to the nucleus therefore requiring more energy to remove (1)