2.5.1 - 2.5.3 properties, substitution reactions, shapes of complexes
1/41
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
42 Terms
d-block element
highest energy electron in a d sublevel
when d-block atoms form ions, which sublevel is filled and emptied first?
4s fills and empties before 3d
which period 4 d-block elements do not follow the trend for electronic configuration?
why?
Cr is [Ar] 4s1 3d5
Cu is [Ar] 4s1 3d10
they have only one electron in their 4s sublevel before electrons fill up the 3d sublevel.
this allows them to have a hall-full or completely full 3d sublevel, which is more stable
what is a transition metal?
which elements are transition metals?
an element that has an incomplete d sublevel in its atoms or ions
elements Ti - Cu
why isn’t Sc a transition metal?
Sc electronic configuration: [Ar] 3d1 4s2
its only stable oxidation state, Sc3+, results in an empty 3d orbital, 3d0
doesn’t form stable ions with an incomplete d sublevel so doesn’t meet the criteria
why isn’t Zn a transition metal?
Zn electronic configuration: [Ar] 3d10 4s2
its only stable oxidation state, Zn2+, results in an empty 4s orbital, 4s0
doesn’t form stable ions with an incomplete d sublevel so doesn’t meet the criteria
characteristic properties of transition metals
formation of complexes
formation of coloured ions
variable oxidation states
catalytic activity
how do the characteristic properties of transition metals arise?
arise due to the incomplete d sub level
Sc and Zn form what colour compounds and what colour solutions?
white compounds
colourless solutions
ligand
a molecule or ion with that forms a co-ordinate bond with a transition metal by donating a lone pair of electrons
a complex
a cental metal atom or ion surrounded by ligands
co-ordination number
the number of co-ordinate bonds to the central metal atom or ion
Lewis acid vs Brønsted–Lowry acid
Lewis: electron pair acceptor (electrophile)
Brønsted–Lowry: proton donor
Lewis base vs Brønsted–Lowry base
Lewis: electron pair donor (nucleophile)
Brønsted–Lowry: proton acceptor
Lewis acid in a complex
Lewis base in a complex
acid → electron pair acceptor → metal
base → electron pair donor → ligand
monodenate ligands
examples
ligands which form one co-ordinate bond to a metal ion
H2O
NH3
Cl-
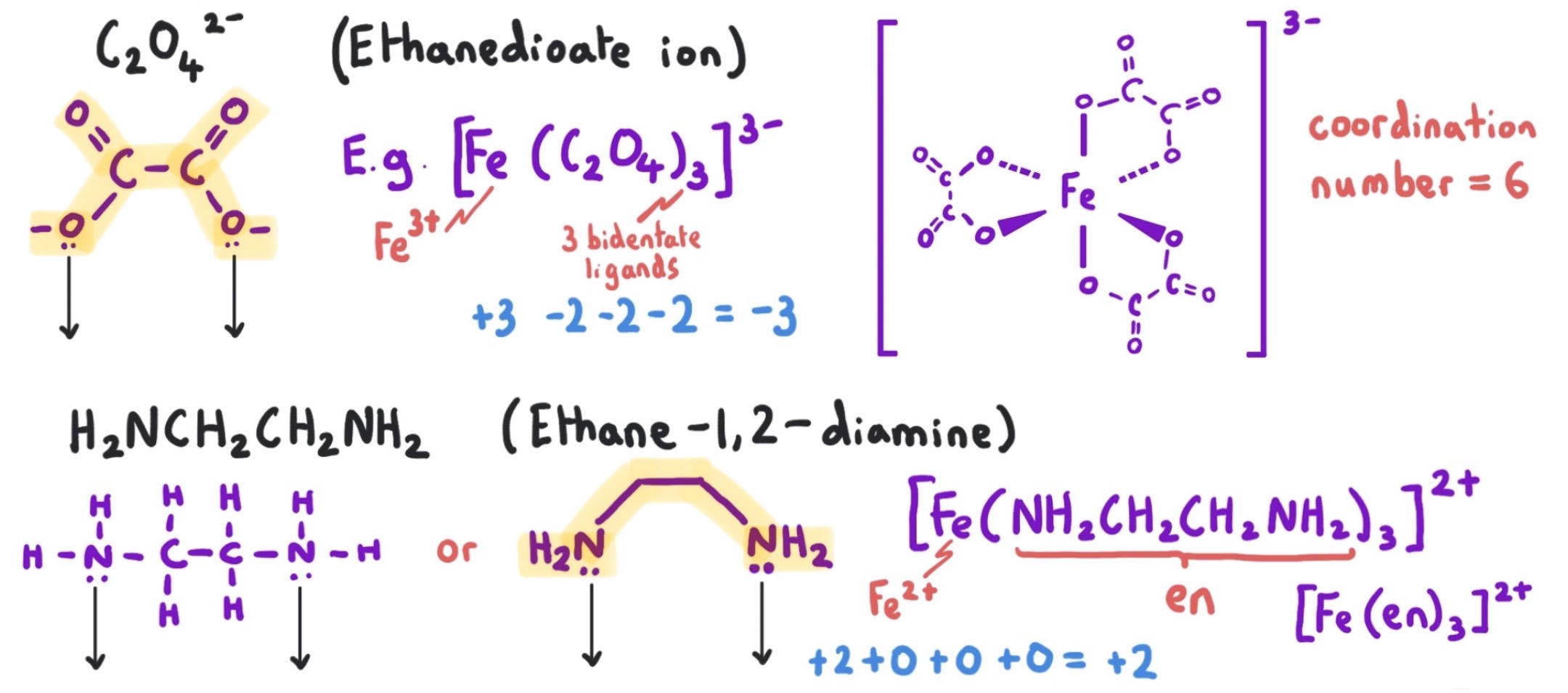
bidentate ligands
examples
ligands which form two co-ordinate bonds to a metal ion
1,2-diaminoethane: NH2CH2CH2NH2 or en
ethanedioate ion: C2O42-
mulitdentate ligands
examples
example of the formula of a complex
ligands which form many co-ordinate bonds to a metal ion
EDTA4–, forms 6 co-ordinate bonds
[CuEDTA]2-
![<ul><li><p>ligands which form many co-ordinate bonds to a metal ion</p></li><li><p>EDTA<sup>4–</sup>, forms 6 co-ordinate bonds</p></li><li><p>[CuEDTA]<sup>2-</sup> </p></li></ul><p></p>](https://assets.knowt.com/user-attachments/9830e93e-b1b2-48f8-8a8b-c0b66d2cae46.jpg)
how does the size of a ligand affect the shape of a complex?
smaller ligand: higher co-ordination number, octahedral complexes.
larger ligand: smaller co-ordination number, tetrahedral, square planar and linear complexes
compare and contrast ligands H2O, NH3 and Cl-
all are monodentate, as their geometry prevents multiple lps from bonding to the same metal atom/ion
H2O and NH3 are small, similar in size and neutral
Cl- is larger and charged
counter ions
ions that bond to the complex ion
e.g. NO3- can bond to [Fe(H2O)6]2+
this forms [Fe(H2O)6](NO3)2
what shape complex do Ag+ ions typically form?
what shape complex do Pt2+ ions typically form?
give reasons why
Ag+ forms linear complexes
Pt2+ forms square planar complexes
shape more stable due to electron configuration of Ag+ / Pt2+ ions
electron pairs repel as far apart as possible
formula of Tollens’ reagent
shape of complex
conditions for aldehyde test
what is oxidised, what is reduced
observation for positive result
[Ag(NH3)2]+
linear
test: [Ag(NH3)2]+ complex is in alkaline conditions, then warm water bath with aldehyde
aldehyde gets oxidised to carboxylate ion, [Ag(NH3)2]+ gets reduced to metallic silver
colourless to silver mirror
equation of positive result for Tollens’ reagent test
RCHO + 2 [Ag(NH3)2]+ + 3 OH- →
RCOO- + 2 Ag + 4 NH3 + 2 H2O
reaction when a silver halide dissolves in aqueous NH3
Tollens’ is produced
e.g. AgBr (s) + 2 NH3 (aq) → [Ag(NH3)2]+ (aq) + Br- (aq)
AgCl dissolves in dilute and conc NH3
AgBr dissolves in conc NH3 only
AgI doesn’t dissolve in NH3
types of stereoisomerism in transition metal complexes
cis-trans isomerism
optical isomerism
what type of isomerism is cis-trans isomerism?
when does it occur in complexes?
E-Z isomerism (geometric isomerism)
displayed in octahedral and square planar complexes with monodentate ligands, where 2 identical monodentate ligands differ in their positions in space relative to each other
cis-isomer vs trans-isomer
cis = 2 identical ligands are next to each other (sisters)
trans = 2 identical ligands are opposite each other
example of cis-trans isomerism in an octahedral complex
solid copper hydroxide, [Cu(H2O)4(OH)2]
![<p>solid copper hydroxide, [Cu(H<sub>2</sub>O)<sub>4</sub>(OH)<sub>2</sub>]</p>](https://assets.knowt.com/user-attachments/b39388b2-fc86-4a12-ba32-975d9fda6ac5.jpg)
example of cis-trans isomerism in an square planar complex
platin, [Pt(NH3)2Cl2]
![<p>platin, [Pt(NH<sub>3</sub>)<sub>2</sub>Cl<sub>2</sub>]</p>](https://assets.knowt.com/user-attachments/c907fa44-7cb4-4679-a00e-191e5f70d1a8.jpg)
why can cis-platin be used as an anti-cancer drug but trans-platin can’t?
trans-platin has the wrong spatial arrangement of its Cl ligands
what type of isomerism is optical isomerism?
when does it occur in complexes?
a form of stereoisomerism
displayed by octahedral complexes with bidentate ligands
what are optical isomers?
what’s another name for optical isomers?
non-superimposable mirror images
enantiomers
example of optical isomerism in a complex
describe the complex
hexaaminecobalt (III) ion, [Co(NH3)6]3+
octahedral complex with bidentate ligands
![<p>hexaaminecobalt (III) ion, [Co(NH<sub>3</sub>)<sub>6</sub>]<sup>3+</sup> </p><p>octahedral complex with bidentate ligands</p>](https://assets.knowt.com/user-attachments/7c5317e2-aa8b-413b-abdb-2033d23e67cc.jpg)
ligand substitution
one ligand is replaced by another
ligand substitution with NH3 and H2O
exchange of NH3 and H2O occurs without change of co-ordination number as they are similar sizes
example: hexa aqua ions + conc NH3
complete: [Co(H2O)6]2+ + 6 NH3 → [Co(NH3)6]2+ + 6 H2O
partial: [Cu(H2O)6]2+ + 4 NH3 → [Cu(NH3)4(H2O)2]2+ + 4 H2O
ligand substitution with Cl-
exchange of H2O by Cl- can cause a change of co-ordination number as Cl- is larger, so fewer ligands can fit around the central metal atom
example: hexa aqua ions + conc HCl
shape goes from octahedral to tetrahedral, co-ordination number goes from 6 to 4
[Cu(H2O)6]2+ + 4 Cl- → [CuCl4]2- + 6 H2O
blue to yellow-green solution
[Co(H2O)6]2+ + 4 Cl- → [CoCl4]2- + 6 H2O
pink to blue solution
[Fe(H2O)6]3+ + 4 Cl- → [FeCl4]1- + 6 H2O
yellow to yellow solution
the chelate effect
bidentate and multidentate ligands (readily) replace monodentate ligands from complexes
coordination number does not change
so same number of coordinate bonds broken as made
bonds have similar enthalpy
so ΔH negligible
ligand substitution involves increase in moles (nproducts side > nreactant side)
so increase in disorder, so positive ΔS
ΔG = ΔH - TΔS
ΔG = 0 - positive = negative, so substitution is feasible
using free energy, how will we know if we have a stable product?
forward reaction has negative ΔG so feasible
therefore reverse reaction has positive ΔG so will not readily occur
so product will not readily turn back into reactants
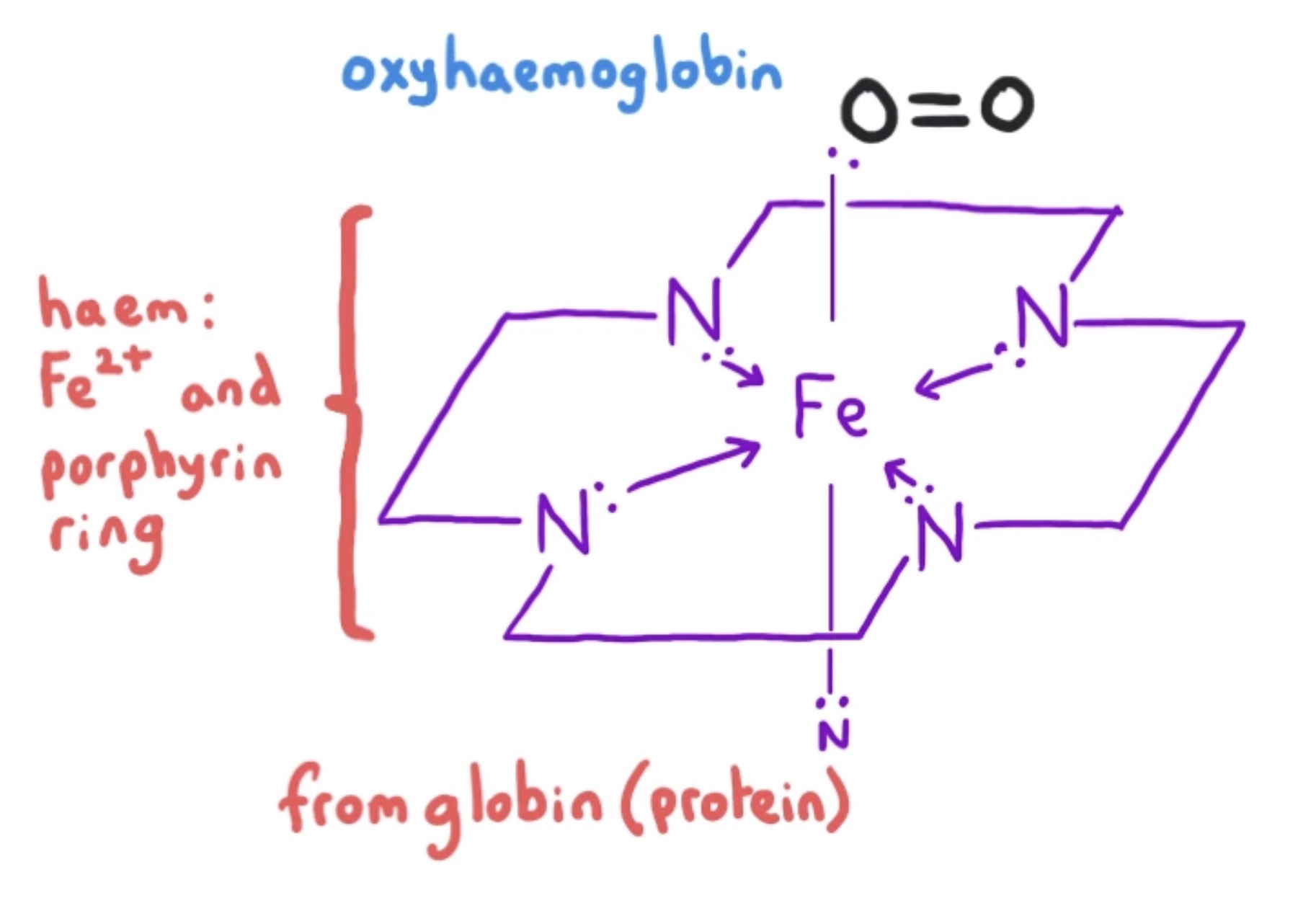
oxyhaemoglobin structure
octahedral complex of iron (II)
horizontal plane
haem group: Fe2+ and porphyrin ring
4 co-ordinate bonds from N atoms to Fe2+
vertical plane
below: 1 co-ordinate bond from N atom in globin to Fe2+
above: 1 co-ordinate bond from O2 to Fe2+
how does the function of haemoglobin involve ligand substitution?
haemoglobin forms oxyhaemoglobin when O2 forms a coordinate bond to Fe2+ in haemoglobin
this enables oxygen to be transported in the blood
O2 can be substituted for H2O in a reversible ligand substitution reaction, forming deoxyhaemoglobin
this enables O2 to be released and used for aerobic respiration
what makes CO toxic?
it replaces O2 co-ordinately bonded to Fe(II) in haemoglobin
forms carboxyhaemoglobin, which is a very stable complex
CO is more strongly bonded, so the ligand substitution is not easily reversible
prevents O2 from binding