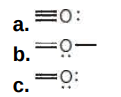Chapter 1
1/99
Earn XP
Description and Tags
Topics include: The Periodic Table, Bonding, Lewis Structures, Isomers, Exceptions to the octet rule, Resonance, Determining molecular shape, Drawing Organic Structures, Hybridizations, Ethane, Ethylene, Acetylene, Bond Length and Bond strength, Electronegativity and bond polarity, Polarity of Molecules, Oxybenzone - A representative organic molecule
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
100 Terms

False
Common Features of Organic Compounds
True or False
All compound that contain the element carbon are organic compounds.

True
Common Features of Organic Compounds
True or False
All organic compounds contain carbon atoms and most contain hydrogen atoms.
True
Common Features of Organic Compounds
True or False
All the carbon atoms have four bonds. A stable carbon atom is said to be tetravalent.
heteroatom
Common Features of Organic Compounds
In organic compounds, other elements (aside from carbon and hydrogen) may also be present. Any atom that is not carbon or hydrogen is called a ________.
N, S, O, P, Halogens
Common Features of Organic Compounds
In organic compounds, other elements (aside from carbon and hydrogen) may also be present. Any atom that is not carbon or hydrogen is called a heteroatom.
Common heteroatoms include __, __, __, __, and the ________.
chains, rings
Common Features of Organic Compounds
Some compounds have _______ of atoms and some compounds have ______.
Nucleus
All matter is composed of the same building blocks called atoms. There are two main components of an atom. The nucleus and the electron cloud.
Where is most of the mass of an atom contained?
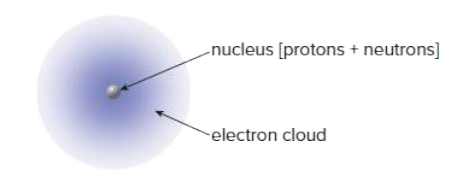
electron cloud
All matter is composed of the same building blocks called atoms. There are two main components of an atom. The nucleus and the electron cloud.
What comprises most of the volume of an atom?
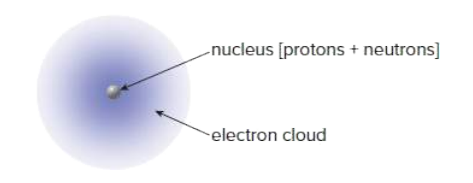
Atomic Number
This quantity relates to the number of protons in the nucleus of an element or neutral atom.
Cation
A ______ is positively charged ion and has fewer electrons than protons.
Anion
An ______ is negatively charged ion and has more electrons than protons.
Isotopes
The number of neutrons in the nucleus of a particular element can vary. ________are two atoms of the same element having a different number of neutrons.
Mass number
This quantity relates to the number of protons and neutrons in the nucleus.
Atomic weight
The ____________ of a particular element is the weighted average of the mass of all its isotopes, reported in atomic mass units (amu).
size
Elements in the same row (period) of the periodic table are similar in ______.
electronic and chemical properties
Elements in the same column (group) of the periodic table are similar in ____________.
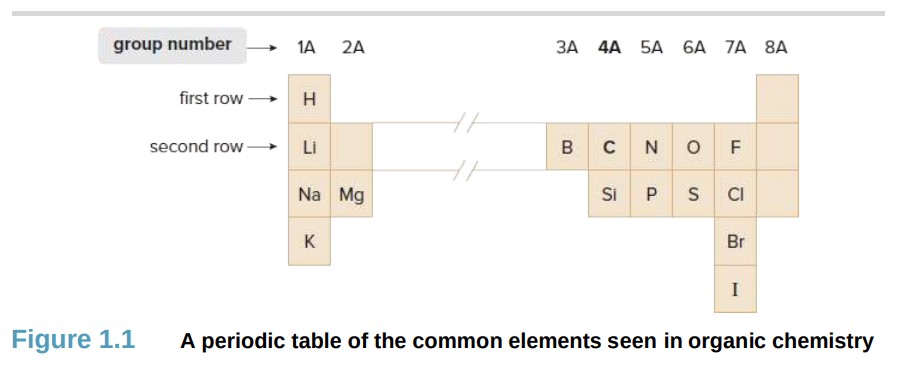
H, Li, Na, K, Mg, B, C, N, O, Si, P, S, F, Cl, Br, I
What are the 16 common elements seen in organic chemistry?
closest
Across each row of the periodic table, electrons are added to a particular shell of orbitals around the nucleus. The shells are numbered 1, 2, 3, and so on. Adding electrons to the first shell forms the first row. Adding electrons to the second shell forms the second row.
Electrons are first added to the shells ________ to the nucleus.
Electron density
Each shell of an atom contains a certain number of orbitals.
An orbital is a region of space that is high in _____________.
s, p, d, f
There are four different kinds of orbitals. Each type of orbital has a particular shape. What are these 4 kinds of orbitals?
lower
An s orbital has a sphere of electron density. It is ________ in energy than other orbitals of the same shell, because electrons are kept closer to the positively charged nucleus.
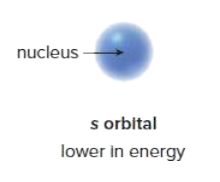
higher
A p orbital has a dumbbell shape. It contains a node of electron density at the nucleus.
A node means there is no electron density in this region.
A p orbital is________ in energy than an s orbital (in the same shell) because its electron density is farther away from the nucleus.
s orbital
What type of orbital is filled with electrons before a p orbital in the same shell.
valence electrons
The outermost electrons are called ____________.
participate in chemical reactions
The valence electrons are more loosely held than the electrons closer to the nucleus, and as such, they _____________.
4,6
The group number of a second-row element reveals its number of valence electrons.
For example, carbon in group 4A has _____ valence electrons, and oxygen in group 6A has ____.
Bonding
________ is the joining of two atoms in a stable arrangement.
complete
One general rule governs the bonding process.
Through bonding, atoms attain a _________ outer shell of valence electrons.
noble gas
Restatement of the general rule that governs the bonding process.
Through bonding, atoms gain, lose, or share electrons to attain the electronic configuration of the ________ closest to them in the periodic table.
Octet rule
It is the general rule governing the bonding process for second row elements. Through bonding, second-row elements attain a complete outer shell of eight valence electrons.
Ionic and Covalent
What are the two kinds of bonding?
Ionic bond
This type of bonding results from the transfer of electrons from one element to another.
Covalent bonds
This type of bonding results from from the sharing of electrons between two nuclei.
noble gas configuration
Atoms readily form ionic bonds when they can attain a _________ by gaining or losing just one or two electrons.
NaCl and KI are ionic compounds.
metal and a non-metal
The type of bonding is determined by the location of an element in the periodic table involved. Ionic bonds form between a __________ through electron transfer
non-metals and non-metals
The type of bonding is determined by the location of an element in the periodic table involved. Covalent bonds form between __________ through electron sharing.
molecule
A compound may have either ionic or covalent bonds. A ________ has only covalent bonds.
covalent bond
A _________ is a two electron bond, and a compound with this bonds is called a molecule.
Covalent
Is this compound ionic or covalent?
F2
Ionic
Is this compound ionic or covalent?
LiBr
Covalent
Is this compound ionic or covalent?
CH3CH3
Both
Is this compound ionic or covalent?
NaNH2
Both
Is this compound ionic or covalent?
NaOCH3
3
How many bonds does Al form?
Note: Atoms with one, two, three, or four valence electrons form one, two, three, or four bonds, respectively, in neutral molecules. Al is in Group 13.
3
How many bonds does N form?
Note: Atoms with five or more valence electrons form enough bonds to give an octet. In this case, the predicted number of bonds = 8 − the number of valence electrons. N is in Group 15.
1
How many bonds does F form?
Note: Atoms with five or more valence electrons form enough bonds to give an octet. In this case, the predicted number of bonds = 8 − the number of valence electrons. F is in Group 17.
2, 3, 1, 4
How many covalent bonds are predicted for the atom:
a. O
b. Al
c. Br
d. Si
Seperate your answer by a comma. Example: 1, 2, 3,
Lewis structures
These are electron dot representations for molecules
ok
Three rules are used for drawing Lewis structures
Draw only the valence electrons.
Give every second-row element no more than eight electrons.
Give each hydrogen two electrons.
Just type ‘ok’
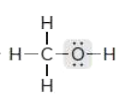
ok
Drawing a Lewis Structure
CH4O
type ‘ok’ to check answer.
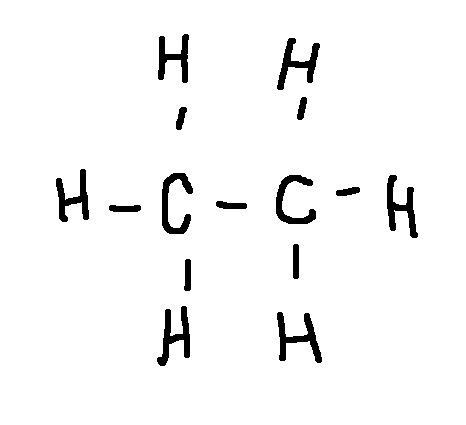
ok
Drawing a Lewis Structure
CH3CH3
type ‘ok’ to check answer.
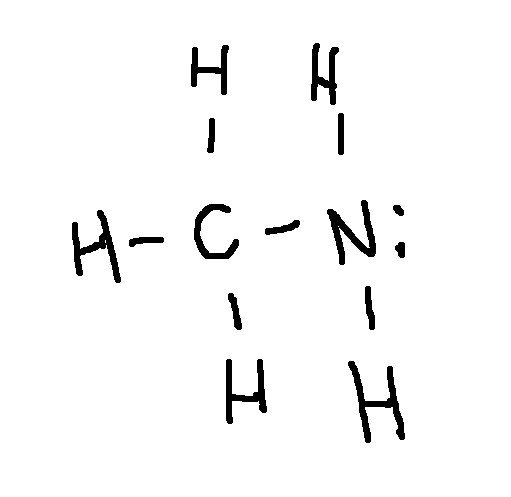
ok
Drawing a Lewis Structure
CH5N
type ‘ok’ to check answer.
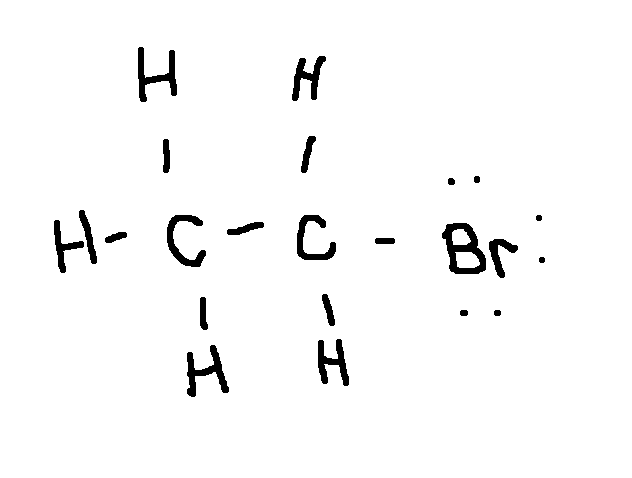
ok
Drawing a Lewis Structure
C2H5Br
type ‘ok’ to check answer.
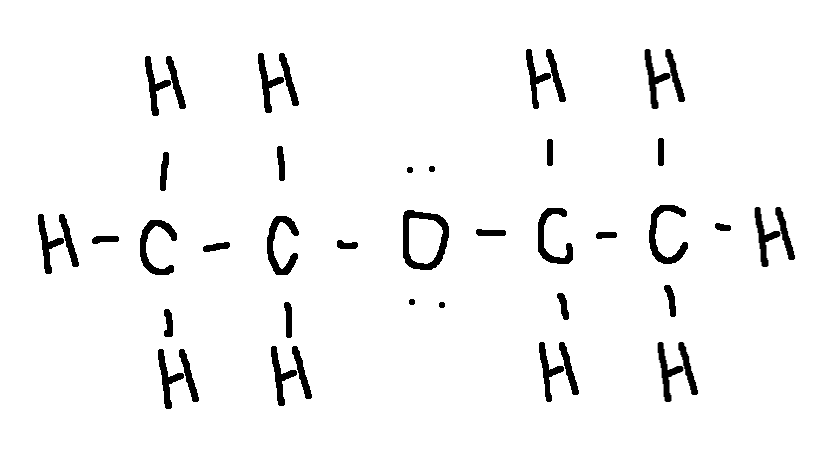
ok
Drawing a Lewis Structure
(CH3CH2)2O
diethyl ether, the first general anesthetic used in medical procedures
type ‘ok’ to check answer.
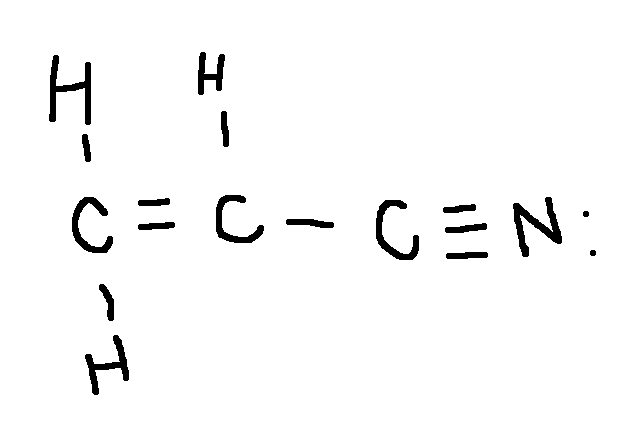
ok
Drawing a Lewis Structure
CH2CHCN
acrylonitrile, starting material used to manufacture synthetic Orlon fibers
type ‘ok’ to check answer.
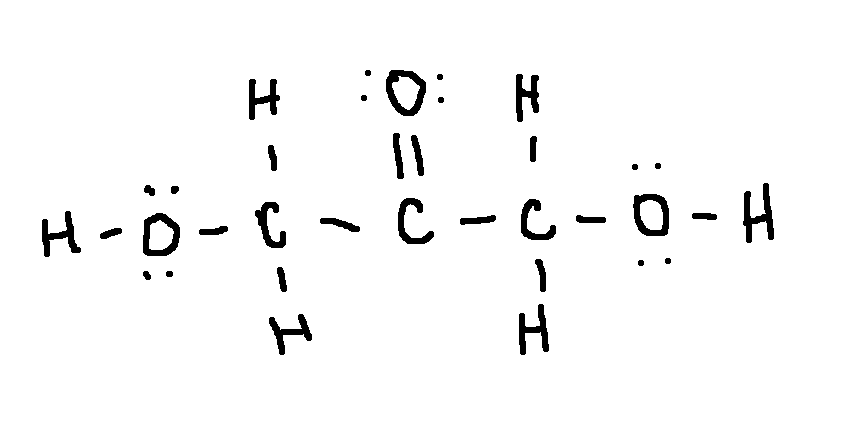
ok
Drawing a Lewis Structure
(HOCH2)2CO
dihydroxyacetone, , an ingredient in sunless tanning products
type ‘ok’ to check answer.
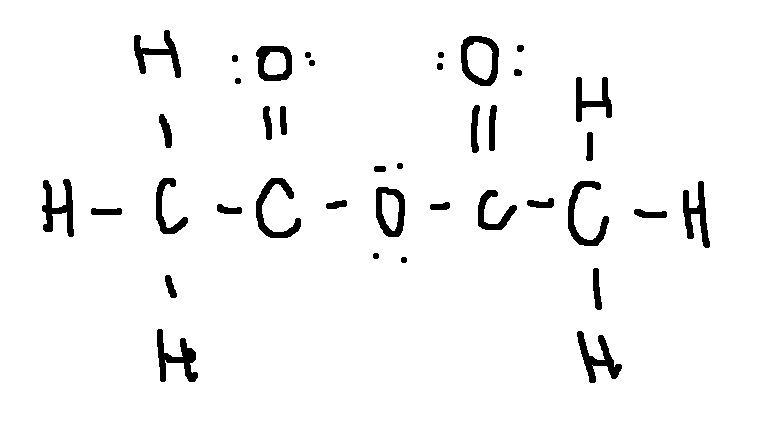
ok
Drawing a Lewis Structure
(CH3CO)2O
acetic anhydride, a reagent used to synthesize aspirin
type ‘ok’ to check answer.
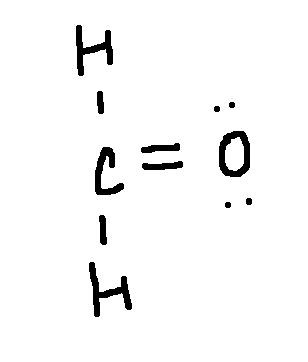
ok
Drawing a Lewis Structure
H2CO
type ‘ok’ to check answer.
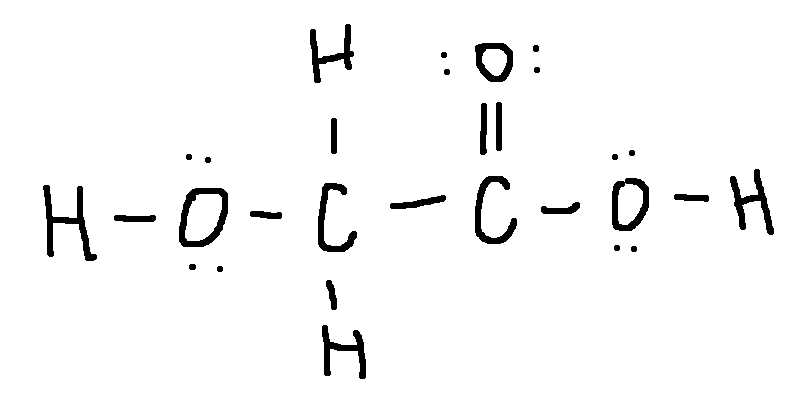
ok
Drawing a Lewis Structure
HOCH2CO2H
type ‘ok’ to check answer.
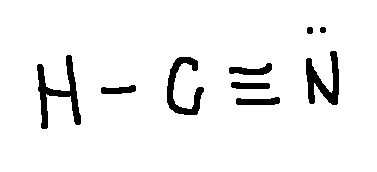
ok
Drawing a Lewis Structure
HCN
type ‘ok’ to check answer.
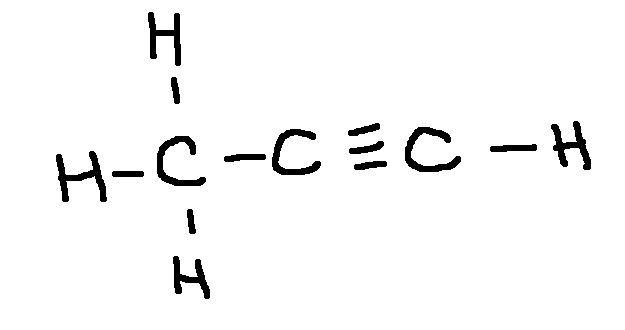
ok
Drawing a Lewis Structure
C3H4
type ‘ok’ to check answer.
Formal Charge
It is the charge assigned to individual atoms in a Lewis structure.
# of valence e - ( # of unshared e + # of bond)
What is the formula for calculating the formal charge of an atom.
bond, lone pair
Formal charge is calculated as follows:
The number of electrons “owned” by an atom is determined by its number of _____ and _______.

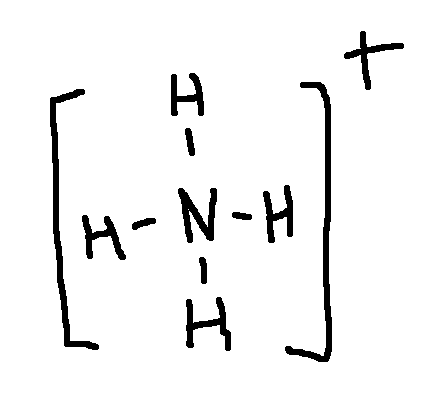
N: +1
Calculate the formal charge on each second-row atom.
Write answer in this format:
N: answer
CH3 Carbon: 0
C triple bonded to C: -1
N: +1
Calculate the formal charge on each second-row atom.
Write answer in this format:
CH3 Carbon: answer
Terminal C: answer
N: answer
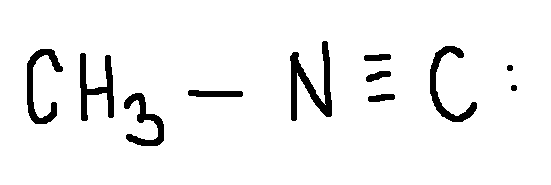
left O: 0
cental O: +1
right O: -1
Calculate the formal charge on each second-row atom.
Write answer in this format:
left O: answer
central O: answer
right O: answer
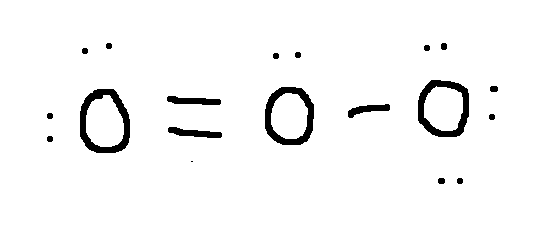
CH3 Carbon: 0
N: +1
double-bonded O: 0
single-bonded O: -1
Calculate the formal charge on each second-row atom.
Write answer in this format:
CH3 Carbon: answer
N: answer
double-bonded O: answer
single-bonded O: answer
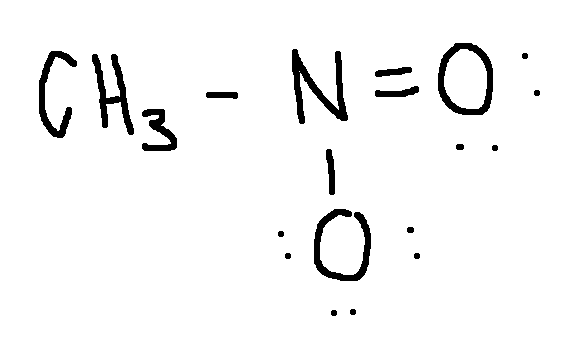
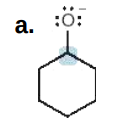
+1
Give the formal charge on the highlighted carbon in each species. All H's and electrons on the highlighted carbon are drawn in.
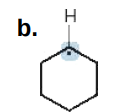
0
Give the formal charge on the highlighted carbon in each species. All H's and electrons on the highlighted carbon are drawn in.
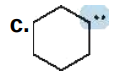
0
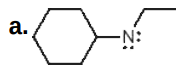
Give the formal charge on the highlighted carbon in each species. All H's and electrons on the highlighted carbon are drawn in.
+1
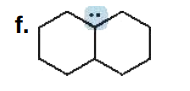
Give the formal charge on the highlighted carbon in each species. All H's and electrons on the highlighted carbon are drawn in.
0
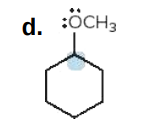
Give the formal charge on the highlighted carbon in each species. All H's and electrons on the highlighted carbon are drawn in.
-1
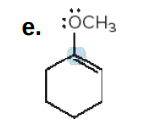
Give the formal charge on the highlighted carbon in each species. All H's and electrons on the highlighted carbon are drawn in.
N: -1
Assign formal charges to each N and O atom in the given molecules. All lone pairs have been drawn in.
Write answer in this format:
N: answer
left N: -1
central N: +1
right N: -1
Assign formal charges to each N and O atom in the given molecules. All lone pairs have been drawn in.
Write answer in this format:
left N: answer
central N: answer
right N: answer
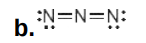
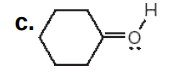
O: +1
Assign formal charges to each N and O atom in the given molecules. All lone pairs have been drawn in.
Write answer in this format:
O: answer
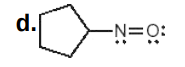
N: 0
O: 0
Assign formal charges to each N and O atom in the given molecules. All lone pairs have been drawn in.
Write answer in this format:
N: answer
O: answer
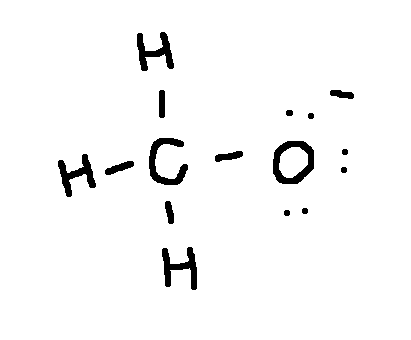
ok
Draw a Lewis structure for each ion:
CH3O-
type ‘ok’ to check answer.
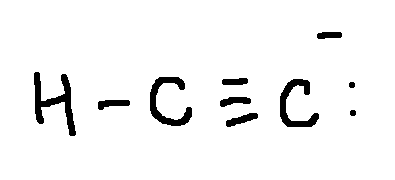
ok
Draw a Lewis structure for each ion:
HC2-
type ‘ok’ to check answer.
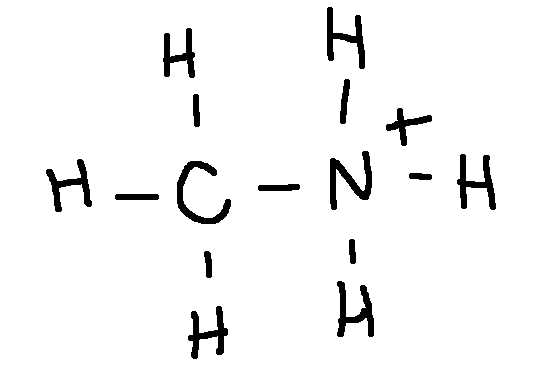
ok
Draw a Lewis structure for each ion:
(CH3NH3)+
type ‘ok’ to check answer.
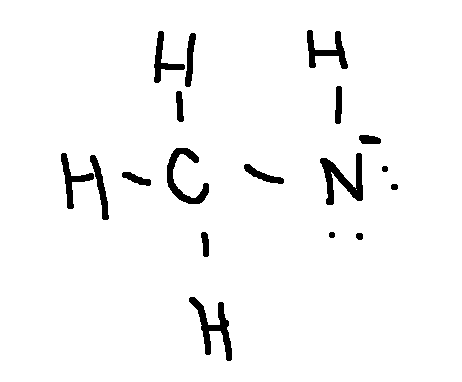
ok
Draw a Lewis structure for each ion:
(CH3NH)−
type ‘ok’ to check answer.
What is the formal charge on the O atom in each of the following species that contains a multiple bond to O?
Write answer in this format